Abstract
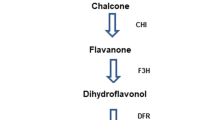
Plant cell culture is a good way to produce anthocyanins owing to its outstanding advantages of short cycles and no limitation on natural environments. However, this method has not been widely used due to the lack of elite cell lines, low yield, and relatively high cost. In the present paper, suspension culture technology of red Cyclocarya paliurus cells was developed to produce anthocyanins, and the effects of hormones on anthocyanin biosynthesis in the cultured cells were investigated through omics. Under the optimized culture conditions and hormones combination, total anthocyanins yield reached 20.81 mg/L. A total of 23 compounds were identified and quantified in the cultured cells by targeted metabolomic analysis of anthocyanins. Hormones showed significant effects on anthocyanins accumulation, and the content of cyanidin-3-O-galactoside and cyanidin-3-O-glucoside increased 1.92 and 1.68 times than that of the control group respectively. Compared with the control group, the optimized hormone combination upregulated the expression levels of key genes in the anthocyanin biosynthesis pathway (flavonoid 3′-hydroxylase (F3′H), dihydroflavonol 4-reductase (DFR), leucocyanidin reductase (LAR), leucoanthocyanidin dioxygenase (LDOX), and anthocyanin-3-O-glucosyltransferase (3GT)) and Transcription factors (TFs) (MYB5, MYB86, ERF003, ERF024, and bZIP44), meanwhile downregulated the expression levels of pathway genes (flavonoid 3′,5′-hydroxylase (F3′5′H) and anthocyanin reductase (ANR)) and TFs (MYB4, MYC2, and bHLH137). Integrative analysis of metabolome and transcriptome indicated that the optimized hormones combination changed TFs’ expression, which further led to the expression change of anthocyanin biosynthesis pathway genes and eventually resulted in a significant increment of anthocyanins biosynthesis in the cultured cells.
Graphical abstract

Key message
Suspension culture of red Cyclocarya paliurus cells was developed to produce anthocyanins and the promotion mechanism of hormone on anthocyanin biosynthesis was interpreted by transcriptome and metabolome.






Similar content being viewed by others
Data availability
The datasets generated during the current study are not publicly available as Cyclocarya paliurus does not yet have publicly available genomic data, all of our subsequent studies rely on existing data but are available from the corresponding author on reasonable request.
Abbreviations
- MS:
-
Murashige and Skoog medium
- 1/2MS:
-
1/2 Murashige and Skoog medium
- B5:
-
Gamborg B5 medium
- KT:
-
Kinetin
- 2,4-D:
-
2,4-Dichlorophenoxy acetic acid
- NAA:
-
1-Naphthylacetic acid
- TDZ:
-
1-Phenyl-3-(1,2,3-thiadiazo-5-yl)urea (thidiazuron)
- IBA:
-
Indole-3-butytric acid
- HPLC:
-
High performance liquid chromatography
- UPLC:
-
Ultra performance liquid chromatography
- ESI-MS:
-
MS Electrospray ionization-mass spectrometry
- LC-MS/MS:
-
Liquid chromatograph-mass spectrometry
References
An JP, Yao JF, Xu RR, You CX, Wang XF, Hao YJ (2018) Apple bZIP transcription factor MdbZIP44 regulates abscisic acid-promoted anthocyanin accumulation. Plant Cell Environ 41(11):2678–2692. https://doi.org/10.1111/pce.13393
Appelhagen I, Wulff-Vester AK, Wendell M, Hvoslef-Eide A-K, Russell J, Oertel A, Martens S, Mock H-P, Martin C, Matros A (2018) Colour bio-factories: towards scale-up production of anthocyanins in plant cell cultures. Metab Eng 48:218–232. https://doi.org/10.1016/j.ymben.2018.06.004
Celli GB, Tan C, Selig MJ (2019) Anthocyanidins and anthocyanins. Encyclopedia of Food Chemistry. Elsevier, Amsterdam, pp 218–223
Chan L, Koay S, Boey P, Bhatt A (2010) Effects of abiotic stress on biomass and anthocyanin production in cell cultures of Melastoma malabathricum. Biol Res 43(1):127–135. https://doi.org/10.4067/S0716-97602010000100014
Davies KM, Deroles SC (2014) Prospects for the use of plant cell cultures in food biotechnology. Curr Opin Biotechnol 26:133–140. doi:https://doi.org/10.1016/j.copbio.2013.12.010
Deng GM, Zhang S, Yang QS, Gao HJ, Hu CH (2021) MaMYB4, an R2R3-MYB repressor transcription factor, negatively regulates the biosynthesis of anthocyanin in banana. Front Plant Sci 11:600704. https://doi.org/10.3389/fpls.2020.600704
Eibl R, Meier P, Stutz I, Schildberger D, Hühn T, Eibl D (2018) Plant cell culture technology in the cosmetics and food industries: current state and future trends. Appl Microbiol Biotechnol 102(20):8661–8675. doi:https://doi.org/10.1007/s00253-018-9279-8
Farjaminezhad R, Zare N, ZAKARIA RA, Farjaminezhad M (2013) Establishment and optimization of cell growth in suspension culture of Papaver bracteatum: a biotechnology approach for Thebaine production. Turk J Biol 37(6):689–697. https://doi.org/10.3906/biy-1304-54
Gagné S, Cluzet S, Mérillon J, Gény L (2011) ABA initiates anthocyanin production in grape cell cultures. J Plant Growth Regul 30(1):1–10. https://doi.org/10.1007/s00344-010-9165-9
He Z, Lv F, Gan Y, Gu J, Que T (2018) Anticancer effects of Cyclocarya paliurus polysaccharide (cpp) on thyroid carcinoma in vitro and in vivo. Int J Polym Sci 2018:1–7. https://doi.org/10.1155/2018/2768120
Huang D, Ming R, Yao S, Li L, Huang R, Tan Y (2021) Identification of anthocyanins in the fruits of Kadsura coccinea using UPLC-MS/MS-based metabolomics. Biochem Syst Ecol 98:104324. https://doi.org/10.1016/j.bse.2021.104324
Jiang T, Zhang M, Wen C, Xie X, Tian W, Wen S, Lu R, Liu L (2020) Integrated metabolomic and transcriptomic analysis of the anthocyanin regulatory networks in Salvia miltiorrhiza Bge. flowers. BMC Plant Biol 20(1):1–13. https://doi.org/10.1186/s12870-020-02553-7
Jun J, Xiao X, Rao X, Dixon R (2018) Proanthocyanidin subunit composition determined by functionally diverged dioxygenases. Nat Plants 4(12):1034–1043. https://doi.org/10.1038/s41477-018-0292-9
Kim JH, Kim HY, Jin CH (2019) Mechanistic investigation of anthocyanidin derivatives as α-glucosidase inhibitors. Bioorg Chem 87:803–809. doi:https://doi.org/10.1016/j.bioorg.2019.01.033
Kodama M, Brinch-Pedersen H, Sharma S, Holme IB, Joernsgaard B, Dzhanfezova T, Amby DB, Vieira FG, Liu S, Gilbert M (2018) Identification of transcription factor genes involved in anthocyanin biosynthesis in carrot (Daucus carota L.) using RNA-Seq. BMC Genom 19(1):1–13. https://doi.org/10.1186/s12864-018-5135-6
Koh ES, Lim JH, Kim MY, Chung S, Shin SJ, Choi BS, Kim HW, Hwang SY, Kim SW, Park CW (2015) Anthocyanin-rich Seoritae extract ameliorates renal lipotoxicity via activation of AMP-activated protein kinase in diabetic mice. J Transl Med 13(1):1–12. https://doi.org/10.1186/s12967-015-0563-4
Konczak I, Terahara N, Yoshimoto M, Nakatani M, Yoshinaga M, Yamakawa O (2005) Regulating the composition of anthocyanins and phenolic acids in a sweetpotato cell culture towards production of polyphenolic complex with enhanced physiological activity. Trends Food Sci Technol 16(9):377–388. doi:https://doi.org/10.1016/j.tifs.2005.02.007
Lai B, Du L-N, Hu B, Wang D, Huang X-M, Zhao J-T, Wang H-C, Hu G-b (2019) Characterization of a novel litchi R2R3-MYB transcription factor that involves in anthocyanin biosynthesis and tissue acidification. BMC Plant Biol 19(1):1–13. doi:https://doi.org/10.1186/s12870-019-1658-5
Lee EJ, Mobin M, Hahn EJ, Paek KY (2006) Effects of sucrose, inoculum density, auxins, and aeration volume on ceil growth of Gymnema sylvestre. J Plant Biol 49(6):427–431. https://doi.org/10.1007/BF03031122
Li L, Zhang CR (2006) Production of puerarin and isoflavones in cell suspension cultures of Pueraria lobata (Willd.): effects of medium supplementation with casein hydrolysate and coconut milk. J Environ Biol 27(1):21–26. https://doi.org/10.2112/05A-0018.1
Li Y, Shan X, Zhou L, Gao R, Yang S, Wang S, Wang L, Gao X (2019) The R2R3-MYB factor FhMYB5 from freesia hybrida contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis. Front Plant Sci. 5:6. https://doi.org/10.3389/fpls.2018.01935
Li H, Yang Z, Zeng Q, Wang S, Luo Y, Huang Y, Xin Y, He N (2020) Abnormal expression of bHLH3 disrupts a flavonoid homeostasis network, causing differences in pigment composition among mulberry fruits. Horticult Res 7(1):1–19. https://doi.org/10.1038/s41438-020-0302-8
Li YP, Tang DB, Wang XQ, Wang M, Yin ZP (2021) Development of origanum vulgare cell suspension culture to produce polyphenols and the stimulation effect of salicylic acid elicitation and phenylalanine feeding. Biotechnol Bioprocess Eng 26(3):456–467. https://doi.org/10.1007/s12257-020-0193-4
Linh TM, Mai NC, Hoe PT, Ngoc NT, Van NT (2021) Development of a cell suspension culture system for promoting alkaloid and vinca alkaloid biosynthesis using endophytic fungi isolated from local Catharanthus roseus. Plants 10(4):672. https://doi.org/10.3390/plants10040672
Liu W, Deng S, Zhou D, Huang Y, Li C, Hao L, Zhang G, Su S, Xu X, Yang R (2020) 3,4-Seco-dammarane triterpenoid saponins with anti-inflammatory activity isolated from the leaves of Cyclocarya paliurus. J Agric Food Chem 68(7):2041–2053. https://doi.org/10.1021/acs.jafc.9b06898
Liu Z, Mohsin A, Wang Z, Zhu X, Zhuang Y, Cao L, Guo M, Yin Z (2021) Enhanced biosynthesis of chlorogenic acid and its derivatives in methyl-jasmonate-treated gardenia jasminoides cells: a study on metabolic and transcriptional responses of cells. Front Bioeng Biotechnol 8:1418. https://doi.org/10.3389/fbioe.2020.604957
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆CT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):1–21. https://doi.org/10.1186/s13059-014-0550-8
Martins V, Garcia A, Costa C, Sottomayor M, Gerós H (2018) Calcium- and hormone-driven regulation of secondary metabolism and cell wall enzymes in grape berry cells. J Plant Physiol 231:57–67. https://doi.org/10.1016/j.jplph.2018.08.011
Mathur A, Mathur AK, Gangwar A, Yadav S, Verma P, Sangwan RS (2010) Anthocyanin production in a callus line of Panax sikkimensis Ban. In Vitro Cell Dev Biol-Plant 46(1):13–21. https://doi.org/10.1007/s11627-009-9253-3
McCabe PF, Levine A, Meijer PJ, Tapon NA, Pennell RI (1997) A programmed cell death pathway activated in carrot cells cultured at low cell density. Plant J 12(2):267–280. https://doi.org/10.1046/j.1365-313X.1997.12020267.x
Nakata M, Mitsuda N, Herde M, Koo A, Moreno JE, Suzuki K, Howe GA, Ohme-Takagi M (2013) A bHLH-type transcription factor, ABA-INDUCIBLE BHLH-TYPE TRANSCRIPTION FACTOR/JA-ASSOCIATED MYC2-LIKE1, acts as a repressor to negatively regulate jasmonate signaling in arabidopsis. Plant Cell 25(5):1641–1656. https://doi.org/10.1105/tpc.113.111112
Ni J, Bai S, Zhao Y, Qian M, Tao R, Yin L, Gao L, Teng Y (2019) Ethylene response factors Pp4ERF24 and Pp12ERF96 regulate blue light-induced anthocyanin biosynthesis in ‘Red Zaosu’pear fruits by interacting with MYB114. Plant Mol Biol 99(1–2):67–78. doi:https://doi.org/10.1007/s11103-018-0802-1
Oraei M, Panahirad S, Zaare-Nahandi F, Gohari G (2019) Pre‐véraison treatment of salicylic acid to enhance anthocyanin content of grape (Vitis vinifera L.) berries. J Sci Food Agric 99(13):5946–5952. https://doi.org/10.1002/jsfa.9869
Owens D, Alerding A, Crosby K, Bandara A, Westwood J, Winkel B (2008) Functional analysis of a predicted flavonol synthase gene family in Arabidopsis. Plant Physiol 147(3):1046–1061. https://doi.org/10.1104/pp.108.117457
Premathilake AT, Ni J, Shen J, Bai S, Teng Y (2020) Transcriptome analysis provides new insights into the transcriptional regulation of methyl jasmonate-induced flavonoid biosynthesis in pear calli. BMC Plant Biol 20(1):1–14. https://doi.org/10.1186/s12870-020-02606-x
Qineng Lu QY, Huawen Zou (2006) Effects of cerium on accumulation of anthocyanins and expression of anthocyanin biosynthetic genes in potato cell tissue cultures. J Rare Earths 24(4):479–484. https://doi.org/10.1016/S1002-0721(06)60147-6
Radha Munagala FA, Jeyaprakash Jeyabalan AK, Agrawal AM, Mudd AH, Kyakulaga, Inder P, Singh MVV, Ramesh C, Gupta (2017) Exosomal formulation of anthocyanidins against multiple cancer types. Cancer Lett 393:94–102. https://doi.org/10.1016/j.canlet.2017.02.004
Rajasekhar E, Edwards M, Wilson S, Street H, THE INFLUENCE OF SHAKING RATE ON THE GROWTH OF SUSPENSION CULTUEES (1971) Studies on the growth in culture of plant cells: XI. J Exp Bot 22(1):107–117. https://doi.org/10.1093/jxb/22.1.107
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3(6):1101–1108. https://doi.org/10.1038/nprot.2008.73
Shimizu Y, Maeda K, Kato M, Shimomura K (2010) Methyl jasmonate induces anthocyanin accumulation in Gynura bicolor cultured roots. In Vitro Cellular & Developmental Biology-Plant 46(5):460–465. doi:https://doi.org/10.1007/s11627-010-9294-7
Silva S, Costa E, Calhau C, Morais R, Pintado M (2017) Anthocyanin extraction from plant tissues: a review. Crit Rev Food Sci Nutr 57(14):3072–3083. https://doi.org/10.1080/10408398.2015.1087963
Simoes-Gurgel C, da Silva Cordeiro L, de Castro TC, Callado CH, Albarello N, Mansur E (2011) Establishment of anthocyanin-producing cell suspension cultures of Cleome rosea Vahl ex DC.(Capparaceae). Plant Cell Tissue Org Cult 106(3):537–545. https://doi.org/10.1007/s11240-011-9945-3
Thorpe TA (2007) History of plant tissue culture. Mol Biotechnol 37(2):169–180. https://doi.org/10.1007/s12033-007-0031-3
Tsay H, Huang H (1998) Somatic embryo formation and germination from immature embryo-derived suspension-cultured cells of Angelica sinensis (Oliv.) Diels. Plant Cell Rep 17(9):670–674. https://doi.org/10.1007/s002990050463
Wang Q, Jiang C, Fa Ng S, Wang J, Ji Y, Shang X, Ni Y, Yin Z, Zhang J (2013) Antihyperglycemic, antihyperlipidemic and antioxidant effects of ethanol and aqueous extracts of Cyclocarya paliurus leaves in type 2 diabetic rats. J Ethnopharmacol 150(3):1119–1127. https://doi.org/10.1016/j.jep.2013.10.040
Wei Z, Furusaki S (1999) Production of anthocyanins by plant cell cultures. Biotechnol Bioprocess Eng 4(4):231–252. https://doi.org/10.1007/BF02933747
Wei X, Ju Y, Ma T, Zhang J, Sun X (2020) New perspectives on the biosynthesis, transportation, astringency perception and detection methods of grape proanthocyanidins. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2020.1777527
Wilson S, Roberts S (2012) Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol J 10(3):249–268. doi:https://doi.org/10.1111/j.1467-7652.2011.00664.x
Wu S, Zu Y, Wu M (2003) High yield production of salidroside in the suspension culture of Rhodiola sachalinensis. J Biotechnol 106(1):33–43. https://doi.org/10.1016/j.jbiotec.2003.07.009
Wu T, Shen M, Yu Q, Chen Y, Chen X, Yang J, Huang L, Guo X, Xie J (2021) Cyclocarya paliurus polysaccharide improves metabolic function of gut microbiota by regulating short-chain fatty acids and gut microbiota composition. Food Res Int 141:110119. https://doi.org/10.1016/j.foodres.2021.110119
Xiao L, Cao S, Shang X, Xie X, Zeng W, Lu L, Kong Q, Yan H (2021) Metabolomic and transcriptomic profiling reveals distinct nutritional properties of cassavas with different flesh colors. Food Chem 2:100016. https://doi.org/10.1016/j.fochms.2021.100016
Xie JH, Xin L, Shen MY, Nie SP, Hui Z, Li C, Gong DM, Xie MY (2013) Purification, physicochemical characterisation and anticancer activity of a polysaccharide from Cyclocarya paliurus leaves. Food Chem 136(3–4):1453–1460. https://doi.org/10.1016/j.foodchem.2012.09.078
Xie J, Dong C, Nie S, Li F, Wang Z, Shen M, Xie M (2015) Extraction, chemical composition and antioxidant activity of flavonoids from Cyclocarya paliurus (Batal.) Iljinskaja leaves. Food Chem 186:97–105. https://doi.org/10.1016/j.foodchem.2014.06.106
Yang B, He S, Liu Y, Liu B, Fang Y (2020) Transcriptomics integrated with metabolomics reveals the effect of regulated deficit irrigation on anthocyanin biosynthesis in cabernet sauvignon grape berries. Food Chem 314:126170. https://doi.org/10.1016/j.foodchem.2020.126170
Yao X, Lin Z, Jiang C, Gao M, Wang Q, Yao N, Ma Y, Li Y, Fang S, Shang X (2015) Cyclocarya paliurus prevents high fat diet induced hyperlipidemia and obesity in Sprague-Dawley rats. Can Physiol Pharmacol 93(8):677–686. https://doi.org/10.1139/cjpp-2014-0477
Yao G, Ming M, Allan AC, Gu C, Li L, Wu X, Wang R, Chang Y, Qi K, Zhang S (2017) Map-based cloning of the pear gene MYB 114 identifies an interaction with other transcription factors to coordinately regulate fruit anthocyanin biosynthesis. Plant J 92(3):437–451. https://doi.org/10.1111/tpj.13666
Yin Z, Shangguan X, Chen J, Qiang Z, Li D (2013) Growth and triterpenic acid accumulation of Cyclocarya paliurus cell suspension cultures. Biotechnol Bioprocess Eng 18(3):606–614. https://doi.org/10.1007/s12257-012-0751-5
Zhang W, Curtin C, Kikuchi M, Franco C (2002) Integration of jasmonic acid and light irradiation for enhancement of anthocyanin biosynthesis in Vitis vinifera suspension cultures. Plant Sci 162(3):459–468. https://doi.org/10.1016/S0168-9452(01)00586-6
Zhang Y, Li W, Dou Y, Zhang J, Jiang G, Miao L, Han G, Liu Y, He L, Zhang Z (2015) Transcript quantification by RNA-seq reveals differentially expressed genes in the red and yellow fruits of Fragaria vesca. PLoS ONE 10(12):e0144356. https://doi.org/10.1371/journal.pone.0144356
Zhao L, Jiang X-L, Qian Y-M, Wang P-Q, Xie D-Y, Gao L-P, Xia T (2017) Metabolic characterization of the anthocyanidin reductase pathway involved in the biosynthesis of Flavan-3-ols in elite shuchazao tea (Camellia sinensis) cultivar in the field. Molecules 22(12):2241. https://doi.org/10.3390/molecules22122241
Zhao W, Tang D, Yuan E, Wang M, Yin Z (2020) Inducement and cultivation of novel red Cyclocarya paliurus callus and its unique morphological and metabolic characteristics. Ind Crops Prod 147:112266. https://doi.org/10.1016/j.indcrop.2020.112266
Zheng J, An Y, Wang L (2018) 24-Epibrassinolide enhances 5-ALA-induced anthocyanin and flavonol accumulation in calli of ‘Fuji’apple flesh. Plant Cell Tissue Organ Cult 134(2):319–330. https://doi.org/10.1007/s11240-018-1418-5
Zhu F (2018) Anthocyanins in cereals: composition and health effects. Food Res Int 109:232–249. https://doi.org/10.1016/j.foodres.2018.04.015
Zurai Da AR, Nazreena OA, Izzati K, Aziz A (2014) Establishment and optimization growth of shoot buds-derived callus and suspension cell cultures of Kaempferia parviflora. Am J Plant Sci 5(18):2693. https://doi.org/10.4236/ajps.2014.518284
Acknowledgements
This study was financially supported by National Natural Science Foundation of China (No. 31960515) and Jiangxi Provincial Natural Science Foundation (No. 20192BAB204004). We thank Wuhan MetWare Biotechnology Co., Ltd. (Wuhan, China), for their support during the metabolome and transcriptome data analysis.
Author information
Authors and Affiliations
Contributions
YL: Methodology, Validation, Investigation, Data curation, Writing - original draft, Writing - review & editing. QL: Data analysis. DT: Conceptualization, Methodology. YC: Investigation. JZ: Investigation. WZ: Methodology & Data analysis. JC: Conceptualization, Methodology, Supervision. QZ: Writing - review & editing. ZY: Conceptualization, Writing - review & editing, Visualization, Project administration, Funding acquisition. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Patricia Marconi.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Liang, Q., Tang, D. et al. Development of suspension culture technology and hormone effects on anthocyanin biosynthesis for red Cyclocarya paliurus cells. Plant Cell Tiss Organ Cult 149, 175–195 (2022). https://doi.org/10.1007/s11240-021-02215-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-021-02215-y