Abstract
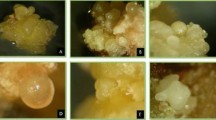
The current investigation revealed a highly proficient and reproducible method for comprehensive in vitro regeneration and structural differentiation of bulblets in Crinum malabaricum Lekhak & Yadav using meta-topolin [mT; 6-(3-hydroxybenzylamino) purine] [6-benzylaminopurine] for the first time. Cultures were established from twin scales explants using optimized sterilization method and growth regulators for regeneration of in vitro plantlets. It was observed that mT at 2.0 mg L−1 was the main factor involved in direct bulblets formation from the basal plate, and the highest mean number of bulblets per twin scale explant was 26 within 8 weeks. On maturation, the bulblets were rooted simultaneously on the mT containing medium. Regenerated plantlets were acclimatized with a 100% survival rate after 8 weeks. Light microscopic examination of bulblet scales/cataphylls was done to characterize the in vitro bulblets developed from mT and BAP containing media. The bulblet scales differed in quantitative traits of tissue systems, and the meta-topolin derived bulblets represented well differentiated dermal, ground and vascular tissues. The results obtained may provide a new tool to improve the production of pharmaceutically important compounds (viz. galanthamine) in bulblets and give the guarantee of survival of plants under natural conditions which can reaffirm the conservation measure of this critically endangered medicinal plant.
Key message
Meta-topolin enhanced in vitro production of bulblets with structurally stable cataphylls in Crinum malabaricum.




Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated during the current study.
References
Ahmad A, Anis M (2019) Meta-topolin improves in vitro morphogenesis, rhizogenesis and biochemical analysis in Pterocarpus marsupium Roxb.: a potential drug-yielding tree. J Plant Growth Regul 38:1007–1016
Aremu A, Dole¿al K, Van Staden J (2015) New cytokinin-like compounds as a tool to improve rooting and establishment of micropropagated plantlets. In: VI international symposium on production and establishment of micropropagated plants. 1155:497–504
Aremu AO, Bairu MW, Doležal K, Finnie JF, VanStaden J (2012) Topolins: a panacea to plant tissue culture challenges? Plant Cell Tiss Org Cult 108(1):1–16
Bairu MW, Stirk WA, Doležal K, Van Staden J (2007) Optimizing the micropropagation protocol for the endangered Aloe polyphylla: can meta-topolin and its derivatives serve as replacement for benzyladenine and zeatin? Plant Cell Tissue Organ Cult 90:15–23
Chahal S, Lekhak MM, Kaur H, Shekhawat MS, Dey A, Jha P, Pandey DK, Kumar V (2012) Unraveling the medicinal potential and conservation of Indian Crinum (Amaryllidaceae) species. S Afr J Bot 36:7–15
Chauhan RD, Taylor NJ (2018) Meta-topolin stimulates de novo shoot organogenesis and plant regeneration in cassava. Plant Cell Tiss Org Cult 132:219–224
Cheesman L, Finnie JF, Van Staden J (2010) Eucomis zambesiaca baker: factors affecting in vitro bulblet induction. S Afr J Bot 76:543–549
Churikoca OA, Barykina RP (2015) Microclonal propagation of some bulbous and cormous plants based on regeneration processes in morphological different explants. Wulfenia 22:21–32
Dragassaki M, Economou AS, Vlahos JC (2003) Bulblet formation in vitro and plantlet survival extra vitrum in Pancratium maritimum L. Acta Hort 616:347–352
Elayaraja D, Subramanyam K, Vasudevan V, Sathish S, Kasthurirengan S, Ganapathi A, Manickavasagam M (2019) Meta-topolin (mT) enhances the in vitro regeneration frequency of Sesamum indicus (L.). Biocatal Agric Biotech 21:101320. https://doi.org/10.1016/j.bcab.2019.101320
Erişen S, Kurt-Güra G, Servi H (2020) In vitro propagation of Salvia sclarea L. by meta-Topolin, and assessment of genetic stability and secondary metabolite profiling of micropropagated plants. Ind Crops Prod 157. https://doi.org/10.1016/j.indcrop.2020.112892
Fennell CW (2002) Crinum moorei: Propagation and secondary metabolite production in vitro. Ph.D. Thesis, University of Natal, Pietermaritzburg, SA
Ghane SG, Attar UA, Yadav PB, Lekhak MM (2018) Antioxidant, anti-diabetic, acetylcholinesterase inhibitory potential and estimation of alkaloids (lycorine and galanthamine) from Crinum species: an important source of anticancer and antiAlzheimer drug. Ind Crops Prod 125:168–177
Grzegorczyk-Karolak I, Hnatuszko-Konka K, Zarzycka M, Kuźma L (2020) The stimulatory effect of purine-type cytokinins on proliferation and polyphenolic compound accumulation in shoot culture of Salvia viridis. Biomol 10:178. https://doi.org/10.3390/biom10020178
Habartová K, Cahlíková L, Řezáčová M, Havelek R (2016) The biological activity of alkaloids from the Amaryllidaceae: from cholinesterases inhibition to anticancer activity. Nat Prod Commun 11(10):1587–1594
Haque M, Jahan S, Rahmatullah M (2014) Ethnomedicinal uses of Crinum asiaticum: a review. World J Pharm Pharm Sci 3:119–128
IUCN (2017) Threatened Species of India Listed in IUCN Red List. www.iucnredlist.org. Accessed 30 April 2021
Jagtap UB, Lekhak MM, Fulzele DP, Yadav SR, Bapat VA (2014) Analysis of selected Crinum species for galanthamine alkaloid: an anti-Alzheimer drug. Curr Sci 107(12):2008–2010
Jayaprakash K, Manokari M, Badhepuri MK, Cokul Raj M, Dey A, Shekhawat MS (2021) Influence of meta-topolin on in vitro propagation and foliar micro-morpho-anatomical developments of Oxystelma esculentum (L. f.) Sm. Plant Cell Tiss Org Cult. https://doi.org/10.1007/s11240-021-02126-y
Johansen DA (1940) Plant Microtechnique. McGraw-Hill, New York, p 523
Khodorova NV, Boitel-Conti M (2013) The role of temperature in the growth and flowering of geophytes. Plants 2(4):699–711
Kumar S, Kashyap M, Sharma DR (2005) In vitro regeneration and bulblet growth from lily bulbscale explants as affected by retardants, sucrose and irradiance. Biol Plant 49(4):629–632
Lekhak MM, Yadav SR (2011) Karyotype studies in two critically endangered and endemic Crinum species (Amaryllidaceae) from Northern-Western Ghats of India. Nucleus 54(2):25–30
Lekhak MM, Yadav SR (2012) Crinum malabaricum (Amaryllidaceae), a remarkable new aquatic species from Kerala, India and lectotypification of Crinum thaianum. Kew Bull 67:521–526
Manokari M, Mehta SR, Priyadharshini S, Badhepuri MK, Dulam S, Jayaprakash K, Cokul Raj M, Dey A, Rajput BS, Shekhawat MS (2021) Meta-Topolin mediated improved micropropagation, foliar micro-morphological traits, biochemical profiling, and assessment of genetic fidelity in Santalum album L. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2021.113931
Marco L, Carmo Carreiras M (2006) Galanthamine, a natural product for the treatment of Alzheimer’s disease. Recent Pat CNS Drug Discov 1:105–111
Marković M, Trifunović Momčilov M, Uzelac B, Jevremović S, Subotić A (2021) Bulb dormancy in vitro—Fritillaria meleagris: initiation, release and physiological parameters. Plants 10:902. https://doi.org/10.3390/plants10050902
Muraseva DS, Novikova TI (2018) Efficient protocol for in vitro propagation from bulb scale explants of Fritillaria ruthenica Wikstr. (Liliaceae), a rare ornamental species. Rend Lincei 29(2):491–497
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol plant 15(3):473–497
Naidoo D, Aremu AO, Van Staden J, Finnie JF (2017) In vitro plant regeneration and alleviation of physiological disorders in Scadoxus puniceus. S Afr J Bot 109:316–322
Nair JJ, Van Staden J (2021) The Amaryllidaceae, a chemically and biologically privileged plant family. S Afr J Bot 136:1–6
Nasircilar AG, Mirici S, Karagüzel ÜÖ, Eren Ö, Baktir I (2011) In vitro propagation of endemic and endangered Muscari mirum from different explant types. Turk J Bot 35(1):37–43
Nowakowska K, Pacholczak A (1994) Comparison of the effect of meta-Topolin and benzyladenine during Daphne mezereum L. micropropagation. Agronomy. https://doi.org/10.3390/agronomy10121994
Ozel CA, Khawar KM, Unal F (2015) Factors affecting efficient in vitro micropropagation of Muscari muscarimi medikus using twin bulb scale. Saudi J Biol Sci 22(2):132–138
Patel M, Patel H (2019) Crinum reddyi sp. nov. (Amaryllidaceae) from Gujarat, India. Nordic J Bot 37:1–5
Patil VS, Adsul AA, Gholave AR, Kambale SS, Lekhak MM, Yadav SR (2015) Prerequisite for conservation of threatened species of Crinum (Amaryllidaceae) from Peninsular India. ENVIS Newsl 20(1):9–10
POWO (2019) ‘’Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew
Priyadharshini S, Kannan N, Manokari M, Shekhawat MS (2020) In vitro regeneration using twin scales for restoration of critically endangered aquatic plant Crinum malabaricum Lekhak & Yadav: a promising source of galanthamine. Plant Cell Tiss Org Cult 141:593–604
Priyadharshini S, Manokari M, Shekhawat MS (2020) In vitro conservation strategies for the critically endangered Malabar river lily (Crinum malabaricum Lekhak & Yadav) using somatic embryogenesis and synthetic seed production. S Afr J Bot 135:172–180
Priyadharshini S, Manokari M, Shekhawat MS (2020b) Attenuation of morpho-anatomical disorders of micropropagated plantlets of Crinum malabaricum Lekhak & Yadav using seismo-mechanical stimulation. Braz J Bot 43:969–977
Punekar SA, Kavade SP, Datar MN, Lakshminarasimhan P, Rao PSN (2004) Crinum woodrowii Baker (Amaryllidaceae), hitherto assumed to be extinct, rediscovered after a century from Mahabaleshwar, India. Curr Sci 87:1049–1051
Rahimi Khonakdari M, Rezadoost H, Heydari R, Mirjalili MH (2020) Effect of photoperiod and plant growth regulators on in vitro mass bulblet proliferation of Narcissus tazzeta L. (Amaryllidaceae), a potential source of galantamine. Plant Cell Tiss Org Cult 142:187–199
Refaat J, Kamel MS, Ramadan MA, Ali AA (2013) Crinum; an endless source of bioactive principles: a review. Part V. Biological profile. Int J Pharm Sci Res 4:39–1252
Rice LJ, Finnie JF, Van Staden J (2011) In vitro bulblet production of Brunsvigia undulata from twin-scales. S Afr J Bot 77:305–312
Saliba S, Ptak A, Boisbrum M, Spina R, Dupire F, Laurain-Mattar D (2016) Stimulating effect of both 4’-O-methylnorbelladine feeding and temporary immersion conditions on galanthamine and lycorine production by Leucojum aestivum L. bulblets. Eng Life Sci 16(8):1–9
Santos TC, Gomes TM, Pinto BAS, Camara AL, Paes AMA (2018) Naturally occurring acetylcholinesterase inhibitors and their potential use for Alzheimer’s disease therapy. Front Pharm 9:1192
Sass JE (1940) Elements of botanical microtechnique, 222nd edn. McFraw-Hill Book Co., New York
Shekhawat JK, Rai MK, Shekhawat NS, Kataria V (2020) Synergism of m-topolin with auxin and cytokinin enhanced micropropagation of Maytenus emarginata. In Vitro Cell Dev Biol Plant 57:418–426
Shekhawat MS, Priyadharshini S, Phanikant J, Vijayakumar, Manokari M (2021) Meta-topolin and liquid medium enhanced in vitro regeneration in Scaevola taccada (Gaertn.) Roxb. In Vitro Cell Dev Biol Plant 57:296–306
Sikder L, Alim MI, Saieda B, Islam MT, Martorell M, Sharifi-Rad J, Khan SA (2021) Pharmacological activities of Crinum viviparum: a laboratory-based study. J Herbs Spices Med Plants 27(2):177–187
Slabbert MM, De Bruyn MH, Ferreira DI, Pretorius J (1993) Regeneration of bulblets from twin scales of Crinum macowanii in vitro. Plant Cell Tiss Org Cult 33:133–141
Stalinova MI, Molle ED, Yanev SG (2010) Galanthamine production by Leucojum aestivum cultures in vitro. Alkaloids Chem Biol 68:167–270
Strnad M (1997) The aromatic cytokinins. Physiol Plant 101:674–688
Strnad M, Hanus J, Vanek T, Kaminek M, Ballantine JA, Fussell B, Hanke DE (1997) Meta-topolin, a highly active aromatic cytokinin from poplar leaves (Populus × canadensis Moench, cv. Robusta). Phytochemistry 45:213–218
Thakur R, Sood A, Nagar PK, Pandey S, Sobti RC, Ahuja PS (2006) Regulation of growth of Lilium plantlets in liquid medium by application of paclobutrazol or ancymidol, for its amenability in a bioreactor system: growth parameters. Plant Cell Rep 25:382–391
Turkyilmaz Unal B (2021) The use of Meta-topolin in cell and tissue cultures for increasing production of secondary metabolites. In: Ahmad N, Strnad M (eds) Meta-topolin: A Growth regulator for Plant Biotechnology and Agriculture. Springer, Singapore, pp 253–263. https://doi.org/10.1007/978-981-15-9046-7_18
Uranbey S, Ipek A, Caliskan M, Dundar E, Cocu S, Basalma D, Guneylioglu H (2010) In vitro bulblet induction from bulb scales of endangered ornamental plant Muscari azureum. Biotechnol Biotechnol Equip 24(2):1843–1848
Wang L, Du Y, Rahman MM, Tang B, Fan L-J, Kilaru A (2018) Establishment of an efficient in vitro propagation system for Iris sanguinea. Sci Rep 8(1):1–10
Youssef NM, Shaaban SA, Ghareeb ZF, Taha LS (2019) In vitro bulb formation of direct and indirect regeneration of Lilium orientalis cv. “Starfighter” plants. Bull Natl Res Cent 43:211. https://doi.org/10.1186/s42269-019-0246-z
Acknowledgements
The authors are grateful to the Science and Engineering Research Board, Department of Science and Technology, Government of India (Sanction Number EMR/2016/007795) for providing financial support to undertake the work for in vitro conservation of this critically endangered medicinal plant.
Author information
Authors and Affiliations
Contributions
MM, MSS, and AD: Conceptualization, investigation, methodology. PS and CRM: Data compilation and hardening of the plants. MSS and MM: Writing of original draft and revision of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Informed consent
was obtained from all individual participants included in the study. Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Research involving human and animal rights
This research did not involve experiments with human or animal participants.
Additional information
Communicated by K. X. Tang.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manokari, M., Priyadharshini, S., Cokulraj, M. et al. Meta-topolin induced morphometric and structurally stable bulblets in Malabar River Lily (Amaryllidaceae). Plant Cell Tiss Organ Cult 148, 377–385 (2022). https://doi.org/10.1007/s11240-021-02195-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-021-02195-z