Abstract
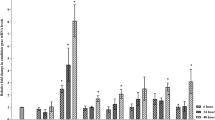
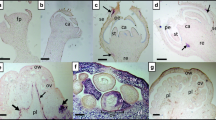
Whitefly (Bemisia tabaci) is a polyphagous insect that causes huge damage in several horticultural crops, including tomato, by sucking nutrients from the phloem and transmitting viruses. Whiteflies are particularly difficult to manage and the use of chemicals remains the common practice, which causes the development of insecticidal resistance. Thus, there is considerable interest in the introduction of resistance to whitefly by classical and molecular breeding. Here, we explored the concept of using an RNA interference construct to silence a v-ATPase gene in whiteflies interacting with transgenic tomato plants that express siRNA molecules corresponding to a fragment from the B. tabaci vATPase. PCR analyses revealed the presence of both ΔATPase and nptII transgenes in all transgenic lines. siRNA expressing lines were challenged against whitefly and revealed a mortality rate of 57.1% in transgenic line 4.4.1, while in the control the mortality was 7.6%. Mortality of 2nd instar nymphs was higher on the transgenic plants and the development of 3rd instar nymphs was slightly longer than on the control plants. Although the attraction of insects was not significantly different between treatments, the number of eggs laid by the insects on the transgenic plants was significantly lower, compared to the controls. RT-qPCR revealed a decreased expression level of endogenous v-ATPase gene in whiteflies feeding on transgenic plants. No unexpected effect was observed on the non-target insects Myzus persicae or Tuta absoluta. Results presented here may form the foundation for the generation of elite tomato varieties resistant to whitefly, a devastating insect pest.
Key message
Genetically modified tomato was generated to express a vATPase small interfering RNA. Transgenic plants were tolerant to whiteflies (Bemisia tabaci) and revealed no effect on Myzus persicae and Tuta absoluta.




Similar content being viewed by others
Data availability
The data generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- 35SCaMV :
-
35 S promoter from the cauliflower mosaic virus.
- AChE :
-
Acetylcholinesterase.
- AMV :
-
Alfalfa mosaic virus.
- Atms-i3 :
-
Intron 3 of the malate synthase gene from Arabidopsis thaliana.
- ATP :
-
Adenosine triphosphate.
- B5 :
-
Basal salt mixture containing micro and macro elements with vitamins as described by Gamborg et al. (1968)
- BAP :
-
6-Benzylaminopurine.
- cDNA :
-
Complementary DNA.
- dsRNA :
-
Double-stranded RNA.
- EcR :
-
Ecdysone receptor.
- EHA105 :
-
Elizabeth Hood Agrobacterium 105.
- hpRNA :
-
Hairpin RNA.
- IAA :
-
Indole-3-acetic acid.
- IBA :
-
Indole-3-butyric acid.
- MEAM1 :
-
Middle East Asia Minor – 1 = Biotype B.
- miRNA :
-
MicroRNA.
- mtCOI :
-
Mitochondrial cytochrome oxidase I.
- NPTII :
-
Neomycin Phosphotransferase II.
- PVP :
-
Polyvinylpyrrolidone.
- RNAi :
-
RNA interference.
- siRNA :
-
Small interfering RNAs.
- vATPase :
-
Vacuolar ATPase.
References
Abbasi A, Sufyan M, Arif MJ, Sahi TS (2020) Effect of silicon on oviposition preference and biology of Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae) feeding on Gossypium hirsutum (Linnaeus). Int J Pest Manag. https://doi.org/10.1080/09670874.2020.1802084
Andrade CM, Tinoco MLP, Rieth AF, Maia FCO, Aragão FJL (2016) Host-induced gene silencing in the necrotrophic fungal pathogen Sclerotinia sclerotiorum. Plant Pathol 65(4):626–632. https://doi.org/10.1111/ppa.12447
Aragão FJL, Nogueira EOPL, Tinoco MLP, Faria JC (2013) Molecular characterization of the first commercial transgenic common bean immune to the Bean golden mosaic virus. J Biotechnol 166(1–2):42–50. https://doi.org/10.1016/j.jbiotec.2013.04.009
Baldin ELL, Vendramim JD, Lourenção AL (2007) Interaction between resistant tomato genotypes and plant extracts on Bemisia tabaci (Genn.) biotype B. Sci. agric. (Piracicaba. Braz) 64(5):476–481. https://doi.org/10.1590/S0103-90162007000500004
Baum JA, Bogaert T, Clinton W, Heck GR, Feldmann P, Ilagan O, Johnson S, Plaetinck G, Munyikwa T, Pleau M, Vaughn T, Roberts J (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25:1322–1326. https://doi.org/10.1038/nbt1359
Bernays EA, Chapman RE (eds) (1994) Chemicals in Plants. In: Host-Plant Selection by Phytophagous Insects. Springer US, Boston, pp 14–60
Beyenbach KW, Wieczorek H (2006) The V-type H+ ATPase: molecular structure and function, physiological roles and regulation. J Exp Biol 209:577–589. https://doi.org/10.1242/jeb.02014
Bonfim K, Faria JC, Nogueira EOPL, Mendes EA, Aragão FJL (2007) RNAi-mediated resistance to Bean golden mosaic virus in genetically engineered common bean (Phaseolus vulgaris). Mol Plant-Microbe Interact 20(6):717–726. https://doi.org/10.1094/MPMI-20-6-0717
Byrne DN, Bellows Junior TS (1991) Whitefly biology. Annu Rev Entomol 36:431–457. https://doi.org/10.1146/annurev.en.36.010191.002243
Chilton MD, Currier TC, Farrand SK, Bendich AJ, Gordon MP, Nester EW (1974) Agrobacterium tumefaciens DNA and PS8 bacteriophage DNA not detected in crown gall tumors. Proc Natl Acad Sci USA 71(9):3672–3676. https://doi.org/10.1073/pnas.71.9.3672
Datla RS, Hammerlindl JK, Pelcher LE, Crosby WL, Selvaraj G (1991) A bifuntional fusion between beta-glucuronidase and neomycin phosphotransferase: a broad-spectrum marker enzyme for plants. Gene 101(2):239–246. https://doi.org/10.1016/0378-1119(91)90417-a
Dorais M, Ehret DL, Papadopoulos AP (2008) Tomato (Solanum lycopersicum) health components: from the seed to the consumer. Phytochem Rev 7:231–250. https://doi.org/10.1007/s11101-007-9085-x
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bull 19:11–15
Gordon KHJ, Waterhouse PM (2007) RNAi for insect-proof plants. Nat Biotechnol 25(11):1231–1232. https://doi.org/10.1038/nbt1107-1231
Greathead AH( (1986) Host plants. In: Cock) MJW (ed) Bemisia tabaci. A Literature Survey on the Cotton Whitefly with an Annotated Bibliography. C.A.B International Institute of Biological Control, Silwood Park, pp 17–26
Hammad EMA-F, Zournajian H, Talhouk S (2001) Efficacy of extracts of Melia azedarach L. callus, leaves and fruits against adults of the sweetpotato whitefly Bemisia tabaci (Hom. Aleyrodidae) J Appl Entomol 125(8):483–488. https://doi.org/10.1046/j.1439-0418.2001.00577.x
Head GP, Carroll MW, Evans SP, Rule DM, Willse AR, Clark TL, Storer NP, Flannagan RD, Samuel LW, Meinke LJ (2017) Evaluation of SmartStax and SmartStax PRO maize against western corn rootworm and northern corn rootworm: efficacy and resistance management. Pest Manage Sci 73(9):1883–1899. https://doi.org/10.1002/ps.4554
Ibrahim AB, Monteiro TR, Cabral GB, Aragão FJL (2017) RNAi-mediated resistance to whitefly (Bemisia tabaci) in genetically engineered lettuce (Lactuca sativa). Transgenic Res 26(5):613–624. https://doi.org/10.1007/s11248-017-0035-0
Jaenike J (1978) On optimal oviposition behavior in phytophagous insects. Theor Popul Biol 14(3):350–356. https://doi.org/10.1016/0040-5809(78)90012-6
Jafarbeigi F, Samih MA, Alaei H, Shirani H (2020) Induced tomato resistance against bemisia tabaci triggered by salicylic acid, β-aminobutyric acid, and trichoderma. Neotrop Entomol 49(3):456–467. https://doi.org/10.1007/s13744-020-00771-0
Jiao X, Xie W, Wang S, Wu Q, Zhou L, Pan H, Liu B, Zhang Y (2012) Host preference and nymph performance of B and Q putative species of Bemisia tabaci on three host plants. J Pest Sci 85:423–430. https://doi.org/10.1007/s10340-012-0441-2
Jones DR (2003) Plant viruses transmitted by whiteflies. Eur J Plant Pathol 109(3):195–219. https://doi.org/10.1023/A:1022846630513
Li J, Rao L, Xie H, Schreiner M, Chen L, Liu Y (2017) Morphology and glucosinolate profiles of chimeric Brassica and the responses of Bemisia tabaci in host selection, oviposition and development. J Integr Agric 16(9):2009–2018. https://doi.org/10.1016/S2095-3119(16)61617-9
Liu S, Jaouannet M, Dempsey D’MA, Imani J, Coustau C, Kogel K-H (2020) RNA-based technologies for insect control in plant production. Biotechnol Adv 39:107463. https://doi.org/10.1016/j.biotechadv.2019.107463
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Luan J-B, Ghanim M, Liu S-S, Czosnek H (2013) Silencing the ecdysone synthesis and signaling pathway genes disrupts nymphal development in the whitefly. Insect Biochem Mol Biol 43(8):740–746. https://doi.org/10.1016/j.ibmb.2013.05.012
Lück S, Kreszies T, Strickert M, Schweizer P, Kuhlmann M, Douchkov D (2019) siRNA-Finder (si-Fi) Software for RNAi-Target Design and Off-Target Prediction. Front Plant Sci 10:1023. https://doi.org/10.3389/fpls.2019.01023
Lundgren JG, Duan JJ (2013) RNAi-based insecticidal crops: potential effects on nontarget species. Bioscience 63(8):657–665. https://doi.org/10.1525/bio.2013.63.8.8
Luo Y, Chen Q, Luan J, Chung SH, Eck JV, Turgeon R, Douglas AE (2017) Towards an understanding of the molecular basis of effective RNAi against a global insect pest, the whitefly Bemisia tabaci. Insect Biochem Mol Biol 88:21e29. https://doi.org/10.1016/j.ibmb.2017.07.005
Malik HJ, Raza A, Amin I, Scheffler JA, Scheffler BE, Brown JK, Mansoor S (2016) RNAi-mediated mortality of the whitefly through transgenic expression of double-stranded RNA homologous to acetylcholinesterase and ecdysone receptor in tobacco plants. Sci Rep 6(1):38469. https://doi.org/10.1038/srep38469
Mao YB, Cai WJ, Wang JW, Hong GJ, Tao XY, Wang LJ, Huang YP, Chen XY (2007) Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25(11):1307–1313. https://doi.org/10.1038/nbt1352
Moshitzky P, Morin S (2014) Bemisia tabaci females from the Mediterranean (Q) species detect and avoid laying eggs in the presence of pyriproxyfen, a juvenile hormone analogue. Pest Manag Sci 70(10):1468–1476. https://doi.org/10.1002/ps.3698
Navas-Castillo J, Fiallo-Olivé E, Sánchez-Campos S (2011) Emerging virus diseases transmitted by whiteflies. Annu Rev Phytopathol 49(15):219–248. https://doi.org/10.1146/annurev-phyto-072910-095235
Obbard DJ, Gordon KHJ, Buck AH, Jiggins FM (2009) The evolution of RNAi as a defence against viruses and transposable elements. Phil Trans R Soc Lond B Biol Sci 364(1513):99–115. https://doi.org/10.1098/rstb.2008.0168
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20(9):709–723. https://doi.org/10.1016/S0261-2194(01)00108-9
Oriani MA, de G, Vendramim JD, (2010) Influence of trichomes on attractiveness and ovipositional preference of Bemisia tabaci (Genn) B biotype (Hemiptera: Aleyrodidae) on tomato genotypes. Neotrop Entomol 39(6):1002–1007. https://doi.org/10.1590/S1519-566-2010000600024
Perring TM, Stansly PA, Liu TX, Smith HA, Andreason SA (2018) Whiteflies: Biology, Ecology, and Management. In: Wakil W, Brust GE, Perring TM Sustainable Management of Arthropod Pests of Tomato. Academic Press - Elsevier Science 372: 73-110 https://doi.org/10.1016/B978-0-12-802441-6.00004-8.
Powell G, Tosh CR, Hardie J (2006) Host plant selection by aphids : Behavioral, evolutionary, and applied perspectives. Annu Rev Entomol 51:309–330. https://doi.org/10.1146/annurev.ento.51.110104.151107
Prabhaker N, Toscano NC, Henneberry TJ (1999) Comparison of neem, urea, and amitraz as oviposition suppressants and larvicides against Bemisia argentifolii (Homoptera: Aleyrodidae). J Econ Entomol 92(1):40–46. https://doi.org/10.1093/jee/92.1.40
Raza A, Malik HJ, Shafiq M, Amin I, Scheffler JA, Scheffler BE, Mansoor S (2016) RNA Interference based Approach to Down Regulate Osmoregulators of Whitefly (Bemisia tabaci): Potential Technology for the Control of Whitefly. PLoS One 11(4):e0153883. https://doi.org/10.1371/journal.pone.0153883
Shah MMR, Liu TX (2013) Feeding Experience of Bemisia tabaci (Hemiptera: Aleyrodidae) Affects Their Performance on Different Host Plants. PLoS One 8(10):e77368. https://doi.org/10.1371/journal.pone.0077368
Sun HJ, Uchii S, Watanabe S, Ezura H (2006) A highly efficient transformation protocol for Micro-Tom, a model cultivar for tomato functional genomics. Plant Cell Physiol 47(3):426–431. https://doi.org/10.1093/pcp/pci251
Swevers L, Smagghe G (2012) Use of RNAi for control of insect crop pests. In: Smagghe G, Diaz I (eds) Arthropod-plant interactions: novel insights and approaches for IPM, Progress in Biological Control 14, Netherlands, pp 177–197. https://doi.org/10.1007/978-94-007-3873-77
Tabuloc CA, Lewald KM, Conner WR, Lee Y, Lee EK, Cain AB, Godfrey KE, Arnó J, Agustí N, Perini CR, Guedes JC, Zalom FG, Chiu JC (2019) Sequencing of Tuta absoluta genome to develop SNP genotyping assays for species identification. J Pest Sci 92:1397–1407. https://doi.org/10.1007/s10340-019-01116-6
Thakur N, Upadhyay SK, Verma PC, Chandrashekar K, Tuli R, Singh PK (2014) Enhanced whitefly resistance in transgenic tobacco plants expressing double stranded RNA of v-ATPase A gene. PLoS One 9(3):e87235. https://doi.org/10.1371/journal.pone.0087235
Tinoco MLP, Dias BBA, Dall’Astta RC, Pamphile JA, Aragão FJL (2010) In vivo trans-specific gene silencing in fungal cells by in planta expression of a double-stranded RNA. BMC Biol 8(27):27. https://doi.org/10.1186/1741-7007-8-27
Ueda H, Kugimiya S, Tabata J, Kitamoto H, Mitsuhara I (2019) Accumulation of salicylic acid in tomato plant under biological stress affects oviposition preference of Bemisia tabaci. J Plant Interact 14(1):73–78. https://doi.org/10.1080/17429145.2018.1550220
Upadhyay SK, Chandrashekar K, Thakur N, Verma PC, Borgio JF, Singh PK, Tuli R (2011) RNA interference for the control of whiteflies (Bemisia tabaci) by oral route. J Biosci 36(1):153–161. https://doi.org/10.1007/s12038-011-9009-1
Vogel E, Santos D, Mingels L, Verdonckt T-W, Broeck JV (2019) RNA Interference in Insects: Protecting Beneficials and Controlling Pests. Front Physiol 9:1912. https://doi.org/10.3389/fphys.2018.01912
Xia J, Guo Z, Yang Z, Han H, Wang S, Xu H, Yang F, Wu Q, Xie W, Zhou X, Dermauw W, Turlings TCJ, Zhang Y (2021) Whitefly hijacks a plant detoxification gene that neutralizes plant toxins. Cell 184:1–13. https://doi.org/10.1016/j.cell.2021.02.014
Yan S, Ren B, Zeng B, Shen J (2020) Improving RNAi efficiency for pest control in crop species. Biotechniques 68(5):283–290. https://doi.org/10.2144/btn-2019-0171
Yang N-W, Li A-L, Wan F-H, Liu W-X, Johnson D (2010) Effects of plant essential oils on immature and adult sweetpotato whitefly, Bemisia tabaci biotype B. Crop Prot 29(10):1200–1207. https://doi.org/10.1016/j.cropro.2010.05.006
Yoo BC, Kragler F, Varkonyi Gasic E, Haywood V, Archer Evans S, Lee YM, Lough TJ, Lucas WJ (2004) A systemic small RNA signaling system in plants. Plant Cell 16:1979–2000 https://doi.org/10.1105/tpc.104.023614
Acknowledgements
We would like to thank Dr. Miguel Michereff Filho (Embrapa Hortaliças, Brasília, DF, Brazil) for providing the T. absoluta and Dr. Josias C. Faria (Embrapa Arroz e Feijão, Santo Antônio de Goiás, GO, Brazil) for providing the whitefly colony. This research was partially supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil, Grant Number 429161/2018-2).
Funding
Funding was provided by Empresa Brasileira de Pesquisa Agropecuária, Conselho Nacional de Desenvolvimento Científico e Tecnológico and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior.
Author information
Authors and Affiliations
Contributions
Investigation [CSRP, WRR, MCM, ALF, PVP, GBC]; Methodology [CSRP, WRR, MCM, FJLA]; Formal analysis [CSRP, PVP, FJLA]; Writing- Reviewing & Editing [CSRP, PVP, GBC, FJLA]; Conceptualization [FJLA]; Funding acquisition [FJLA], Supervision [FJLA]. All of the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no declarations of interest.
Additional information
Communicated by Sergio J. Ochatt.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pizetta, C.S.R., Ribeiro, W.R., Ferreira, A.L. et al. RNA interference-mediated tolerance to whitefly (Bemisia tabaci) in genetically engineered tomato. Plant Cell Tiss Organ Cult 148, 281–291 (2022). https://doi.org/10.1007/s11240-021-02185-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-021-02185-1