Abstract
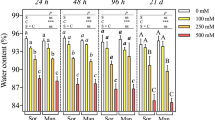
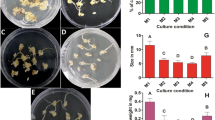
In plants, sucrose is the principal transported carbon compound. Sucrose and/or glucose and fructose are relevant in the regulation of meristematic cell proliferation. The aim was to define the sugar balance and the relationship between the osmoregulation processes and the cell division patterns in callus of Populus alba in response to different sucrose medium concentrations. Callus proliferation and metabolism were assessed through biometric measures, non-structural soluble carbohydrates quantification, osmotic potential determination, as well as by quantification of mRNA accumulation of B-type cyclin genes. Calli were cultured on proliferation medium without sucrose or supplemented with 10, 20 or 30 g l−1 of sucrose. A large amount of non-structural soluble carbohydrates was used to restore the osmotic balance between callus and medium; upon reaching the equilibrium, carbon was then used for cell division. The initial investment of carbon for osmoregulation processes can explain the differences in the lag phase duration in response to the decrease of medium sucrose concentration. However, calli cultured on medium added with 30 g l−1 of sucrose used carbon both to adjust their osmotic potential and to restore the growth whilst the calli on sucrose free medium entered in a quiescent state. The growth rates compared to the transcript accumulation trends suggested that a threshold effect, rather than a quantitative regulation model, governed the relation between CycB gene transcription and cell division. The findings showed that poplar calli used the carbon following a “hierarchical” model based on their physiological state and the sugar concentration available in the medium.






Similar content being viewed by others
References
Asemota O, Eke CR, Odewale JO (2007) Date palm (Phoenix dactylifera L.) in vitro morphogenesis in response to growth regulators, sucrose and nitrogen. Afr J Biotechnol 6(20):2353–2357
Berta M, Giovannelli A, Sebastiani F, Camussi A, Racchi ML (2010) Transcriptome changes in the cambial region of poplar (Populus alba L.) in response to water deficit. Plant Biol 12(2):341–354. doi:10.1111/j.1438-8677.2009.00320.x
Beritognolo I, Harfouche A, Brilli F, Prosperini F, Gaudet M, Brosché M, Salani F, Kuzminsky E, Auvinen P, Paulin L, Kangasjärvi J, Loreto F, Valentini R, Mugnozza GS, Sabatti M (2011) Comparative study of transcriptional and physiological responses to salinity stress in two contrasting Populus alba L. genotypes. Tree Physiol 31(12):1335–1355. doi:10.1093/treephys/tpr083
Blanc G, Lardet L, Martin A, Jacob JL, Carron MP (2002) Differential carbohydrate metabolism conducts morphogenesis in embryogenic callus of Hevea brasiliensis (Müll. Arg.). J Exp Bot 53(373):1453–1462. doi:10.1093/jexbot/53.373.1453
Brown DCW, Leung DWM, Thorpe TA (1979) Osmotic requirement for shoot formation in tobacco callus. Physiol Plant 46(1):36–41. doi:10.1111/j.1399-3054.1979.tb03182.x
Brunner AM, Yakovlev IA, Strauss SH (2004) Validating internal controls for quantitative plant gene expression studies. BMC Plant Biol 4:14. doi:10.1186/1471-2229-4-14
Botha FC, O’Kennedy MM (1998) Carbohydrate utilization by cell suspension cultures of Phaseolus vulgaris. Physiol Plant 102:429–436. doi:10.1034/j.1399-3054.1998.1020311
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Report 11(2):113–116. doi:10.1007/BF02670468
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103(4):551–560. doi:10.1093/aob/mcn125
Confalonieri M, Balestrazzi A, Bisoffi S, Carbonera D (2003) In vitro culture and genetic engineering of Populus spp.: synergy for forest tree improvement. Plant Cell Tissue Organ Cult 72(2):109–138. doi:10.1023/A:1022265504775
Confalonieri M, Belenghi B, Balestrazzi A, Negri S, Facciotto G, Schenone G, Delledonne M (2000) Transformation of elite white poplar (Populus alba L.) cv. ‘Villafranca’ and evaluation of herbicide resistance. Plant Cell Rep 19(10):978–982. doi:10.1007/s002990000230
Delledonne M, Allegro G, Belenghi B, Balestrazzi A, Picco F, Levine A, Zelasco S, Calligari P, Confalonieri M (2001) Transformation of white poplar (Populus alba L.) with a novel Arabidopsis thaliana cysteine proteinase inhibitor gene and analysis of insect pest resistance. Mol Breed 7(1):35–42. doi:10.1023/A:1009605001253
Demeter Z, Kanalasa P, Máthéa C, Csekeb K, Szöllösi E, Hamvasa M, Jámbrika K, Kissa Z, Mészáros I (2014) Osmotic stress responses of individual white oak (Quercus section, Quercus subgenus) genotypes cultured in vitro. J Plant Physiol 171:16–24. doi:10.1016/j.jplph.2013.09.013
De Veylder L, Joubes J, Inzè D (2003) Plant cell cycle transitions. Curr Opin Plant Biol 6(6):536–543. doi:10.1016/j.pbi.2003.09.001
Dong Q, Zhao Y, Jiang H, He H, Zhu S, Cheng B, Xiang Y (2011) Genome-wide identification and characterization of the cyclin gene family in Populus trichocarpa. Plant Cell Tissue Organ Cult 107:55–67. doi:10.1007/s11240-011-9957-z
Eckstein A, Zieba P, Gabrys H (2012) Sugar and light effects on the condition of photosynthetic apparatus of Arabidopsis thaliana cultured in vitro. J Plant Growth Regul 31(1):90–101. doi:10.1007/s00344-011-9222-z
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucl Acids Res 32(5):1792–1797. doi:10.1093/nar/gkh340
Fowler MR, Eyre S, Scott NW, Slater A, Elliott MC (1998) The plant cell cycle in context. Mol Biotechnol 10:123–153. doi:10.1093/jxb/eru188
Giovannelli A, Emiliani E, Traversi ML, Deslauriers A, Rossi S (2011) Sampling cambial region and mature xylem for non structural carbohydrates and starch analyses. Dendrochronologia 29:177–182. doi:10.1016/j.dendro.2011.01.001
Harashima H, Dissmeyer N, Schnittger A (2013) Cell cycle control across the eukaryotic kingdom. Trends Cell Biol 23(7):345–356. doi:10.1016/j.tcb.2013.03.002
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8(2):R19. doi:10.1186/gb-2007-8-2-r19
Howard A, Pelc SR (1986) Synthesis of deoxyribonucleic acid in normal and irradiated cells and its relation to chromosome breakage. Int J Radiat Biol 49(2):207–218. doi:10.1080/09553008514552501
Kirkan MB, Gardner WR, Gelroff GC (1972) Regulation of cell division and cell enlargement by turgor pressure. Plant Physiol 49(6):961–962
Khayri JM, Bahrani AM (2002) Callus growth and proline accumulation in response to sorbitol and sucrose-induced osmotic stress in rice. Biol Plant 45(4):609–611. doi:10.1023/A:1022380827034
Javed F, Ikram S (2008) Effect of sucrose induced osmotic stress on callus growth and biochemical aspects of two wheat genotypes. Pak J Bot 40(4):1487–1495
Jayaraman S, Daud NH, Halis R, Mohamed R (2014) Effects of plant growth regulators, carbon sources and pH values on callus induction in Aquilaria malaccensis leaf explants and characteristics of the resultant calli. J For Res 25(3):535–540. doi:10.1007/s11676-014-0492-8
Iannucci A, Russo M, Arena L, Fonzo N, Martiniello P (2002) Water deficit effects on osmotic adjustment and solute accumulation in leaves of annual clovers. Eur J Agron 16(2):111–122. doi:10.1016/S1161-0301(01)00121-6
Lambardi M, Fabbri A, Caccavale A (2000) Cryopreservation of white poplar (Populus alba L.) by vitrification of in vitro-grown shoot tips. Plant Cell Rep 19(3):213–218. doi:10.1007/s002990050001
Luisi A, Giovannelli A, Traversi ML, Anichini M, Sorce C (2014) Hormonal response to water deficit in cambial tissue of Populus alba L. J Plant Growth Regul 33(3):489–498. doi:10.1007/s00344-013-9401-1
Marron N, Ricciotti L, Bastien C, Beritognolo I, Gaudet M, Paolucci I, Fabbrini F, Salani F, Dillen SY, Ceulemans R, Pinel MPC, Taylor G, Scarascia-Mugnozza G, Sabattia M (2010) Plasticity of growth and biomass production of an intraspecific Populus alba family grown at three sites across Europe during three growing seasons. Can J Res 40:1887–1903. doi:10.1139/X10-113
Matsumoto T, Okunishi K, Nishida K, Noguchi M, Tamaki E (1971) Studies on the culture conditions of higher plant cells in suspension culture. Part II. Effect of nutritional factors on the growth. Agric Biol Chem 35(4):543–551. doi:10.1271/bbb1961.35.543
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15(3):473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Mustafa NR, De Winter W, Van Iren F, Veerporter R (2011) Initiation, growth and cryopreservation of plant cell suspension cultures. Nat Protoc 6(6):715–742. doi:10.1038/nprot.2010.144
Nieuwland J, Scofield S, Murray JAH (2009) Control of division and differentiation of plant stem cells and their derivatives. Elsevier Semin Cell Dev Biol 20:1134–1142. doi:10.1016/j.semcdb.2009.09.011
Nuutila AM, Kurtén U, Kauppinen V (1991) Optimization of sucrose and inorganic nitrogen concentrations for somatic embryogenesis of birch (Betula pendula Roth.) callus cultures: a statistical approach. Plant Cell Tissue Organ Cult 24(2):73–77. doi:10.1007/BF00039733
Obata-Sasamoto H, Thorpe TA (1982) Cell wall invertase activity in cultured tobacco tissues. Plant Cell Tissue Organ Cult 2:3–9. doi:10.1007/BF00033546
Pallara G, Giovannelli A, Traversi ML, Camussi A, Racchi ML (2012) Effect of water deficit on expression of stress-related genes in the cambial region of two contrasting poplar clones. J Plant Growth Regul 31:102–112. doi:10.1007/s00344-011-9223
Pan MJ, Staden J (1999) Effect of activated charcoal, autoclaving and culture media on sucrose hydrolysis. Plant Growth Regul 29:135–141. doi:10.1023/A:1006238300890
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45. doi:10.1093/nar/29.9.e45
Richard C, Lescot M, Inzè D, De Veylder L (2002) Effect of auxin, cytokinin and sucrose on cell cycle gene expression in Arabidopsis thaliana cell suspension cultures. Plant Cell Tissue Organ Cult 69:167–176. doi:10.1023/A:1015241709145
Riou-Khamlichi C, Menges M, Healy JMS, Murray JAH (2000) Sugar control of the plant cell cycle: differential regulation of Arabidopsis D-type cyclin gene expression. Mol Cell Biol 20(3):4513–4521
Rolland F, Moore B, Sheen J (2002) Sugar sensing and signaling in plants. Plant Cell 14(1):185–205. doi:10.1105/tpc.010455
Shin KS, Chakrabarty D, Ko JY, Han SS, Paek KY (2003) Sucrose utilization and mineral nutrient uptake during hairy root growth of red beet (Beta vulgaris L.) in liquid culture. Plant Growth Regul 39:187–193. doi:10.1023/A:1022525308389
Stanzel M, Sjolund RD, Komor E (1988) Transport of glucose, fructose and sucrose by Streptanthus tortuosus suspension cells. I. Uptake at low sugar concentration. Planta 174:201–209. doi:10.1007/BF00394773
Stals H, Inzè D (2001) When plant cells decide to divide. Trends Plant Sci 6(8):359–364. doi:10.1016/S1360-1385(01)02016-7
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. doi:10.1093/molbev/msr121
Tisserat B (1982) Factors involved in the production of plantlets from date palm callus cultures. Euphytica 31:201–214. doi:10.1007/BF00028323
Valliyodan B, Nguyen HT (2006) Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr Opin Biol 9(2):189–195. doi:10.1016/j.pbi.2006.01.019
Acknowledgments
Financial support was provided by MIUR (Progetto Premiale 2012 “Aqua”) and Regione Toscana (POR CRO FSE 2012-2014, research project “Sysbiofor”). The authors would like to extend their sincere thanks to Mrs Monica Anichini for the histological observations and B.Sc. students Ms Elena Palmieri and Ms Noemi Bonadies for their technical support. The authors would like to thank the anonymous reviewers for their valuable comments and suggestions to improve the quality of the paper.
Author contribution statement
AG and GE conceived and designed the research. ST, ADC, GE and MLT performed all the biometric, biochemical, and molecular analyses. ST, GE, AG, ADC and CC analysed data. All Authors drafted and approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Giovanni Emiliani and Silvia Traversari have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Emiliani, G., Traversari, S., De Carlo, A. et al. B-type cyclin modulation in response to carbon balance in callus of Populus alba . Plant Cell Tiss Organ Cult 124, 283–293 (2016). https://doi.org/10.1007/s11240-015-0892-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0892-2