Abstract
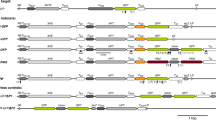
Double-stranded RNA interference can be used to silence gene expression in various organisms. Sustained RNAi-mediated gene silencing is typically triggered by hairpin RNAs (hpRNAs) generated by the transcription of inverted repeat (IR) DNA constructs. In this paper, we describe a transient expression assay that uses a green fluorescent protein marker gene (GFP) fused to the coat protein gene (CP) of papaya ring spot virus. This yields a nuclear and cytomembrane localized protein expression vector that can be used to monitor the silencing efficiency of inverted repeats. An ihpRNA was derived from a 215 nt IR segment of the 3′ end of the CP gene and recombined into the pHellsgate12 vector. We also recombined the 861 nt CP into the destination vectors pHellsgate12 and pWatergate using the GatewayTM technology. Nicotiana benthamiana protoplasts were co-transfected with (1) pGA, (2) pGA + pHellsgate12-CP IR, (3) pGA + pWatergate-CP IR, and (4) pGA + pHellsgate12-CP 215IR. Expression of the GFP fusion protein decreased by 77.7, 75.4, and 65.6% for the three constructs. Transfection with (2) and (3) yielded a silencing efficiency significantly higher than that of the (4) (P < 0.05), as measured by fluorescence intensity upon co-transfection, using the vector pGA-DsRed as an internal control. Depletions of 90.7, 91.5, and 87.5% of the CP transcript were observed by RT-PCR in protoplasts co-transfected with the (1) pGA, (2) pGA + pHellsgate12-CPIR, and (3) pGA + pWatergate-CPIR, (4) pGA + pHellsgate12-CP 215IR. Transient RNAi depletion of the target polypeptide was also detected by western blot analysis. Short RNA fragments complementary to the p6a and p7a antisense probes were detected by Ribonuclease Protection Assays.









Similar content being viewed by others
Abbreviations
- CP :
-
Coat protein
- dsRNA:
-
Double stranded RNA
- GFP:
-
Green fluorescence protein
- hpRNA:
-
Hairpin RNAs
- IR:
-
Inverse repeat
- NLS:
-
Nuclear localization signal
- PBS:
-
Phosphate buffer solution
- PTGS:
-
Post-transcriptional gene silencing
- RNAi:
-
RNA interference
- RPA:
-
Ribonuclease protection assays
- siRNA:
-
Small interfering RNA
References
Agarwal SK, Gao SH, Smith AV, Jin H (2007) A novel class of bacteria-induced small RNAs in Arabidosis. Genes Dev 21:3123–3134
An G (1986) Development of plant promoter expression vectors and their use for analysis synthase promoter in transformed tobacco cells. Plant Physiol 81:86–91
Bart R, Chern M, Park CJ, Bartley L, Ronalc PC (2006) A novel system for gene silencing using siRNAs in rice leaf and stem-derived protoplasts. Plant Methods 2:13. doi:10.1186/1746-4811-2-13
Baulcombe DC (2004) RNA silencing in plants. Nature 431:356–363
Bernstein E, Caudy AA, Hammond SM, Hannon G (2001) Role for abidentate ribonuclease in the initiation step of RNA interference. Nature 409:363–366
Bezanilla M, Aihong P, Quatrano RalphS (2003) RNA interference in the moss Physcomitrella patens. Plant Physiol 133:470–474
Brodersen P, Voinnet O (2006) The diversity of RNA silencing pathways in plants. Trends Genet 22:268–280
Brodersen P, Sakvarelidze-Achard L, Bruun-Rasmussen M, Dunoyer P, Yamamoto YY, Sieburth L, Voinnet O (2008) Widespread translational inhibition by plant miRNAs and siRNAs. Science 320:1185–1190
Chau BL, Lee KA (2007) Function and anatomy of plant siRNA pools derived from hairpin transgenes. Plant Methods 3:13. doi:10.1186/1746-4811-3-13
Chen YK, Lohuis D, Goldbach R, Prins M (2004) High frequence induction of RNA-mediated resistance against Cucumber mosaic virus using inverted repeat constructs. Mol Breeding 14:215–226
Cordula T, Angela S, Armin P, Wiebke W, Axel B, Agnes HW, Leticia SB, Peter L, Daniel M (2008) Off-target effects of siRNA specific for GFP. BMC Mol Biol 9:60. doi:10.1186/1471-2199-9-60
Fitch MM, Pang SZ, Slightom JL, Lius S (1994) Genetic Transformation in Carica papaya L (Papaya). In: Bajaj YPS (ed) Biotechnology in Agriculture and Forestry somatic embryogenesis and synthetic seed, vol 29. Springer, Berlin, pp 237–256
Hanako S, Yuki K, Kazunori G, Chikara M (2008) Degree of RNA silencing and the ability of a viral suppressor vary depending on the cell species in a protoplast system. J Gen Plant Pathol 74:326–330
He P, Shan L, Sheen J (2006) The use of protoplasts to study innate immune responses. In: Ronald PC (ed) From: methods in molecular biology, plant–pathogen interactions: methods and protocols, vol 354. Humana Press Inc., Totowa http://genetics.mgh.harvard.edu/sheenweb/reprints/Protoplast-Innate%20immunity-final.pdf
Huang Z, Han Y, Howell SH (2000) Formation of surface tubules and fluorescent foci in Arabidopsis thaliana protoplasts expressing a fusion between the green fluorescent protein and the cauliflower mosaic virus movement protein. Virology 271:58–64
Jiang L, Zhang FY, Zhang MT (2007) Detection of Papaya ringspot virus (PRSV) by cDNA probes labeled with Digoxgenin labelling and AlkPhos direct labelling. J Agric Biotechnology 15(3):496–502
Lennefors BL, Savenkov EI, Bensefelt J, Elisabeth WW, van Roggen P, Tuvesson S, Valkonen JPT, Gielen J (2006) DsRNA-mediated resistance to beet necrotic yellow vein virus infections in suger beet (Beta vulgaris L. ssp. Vulgaris). Mol Breeding 18(4):313–325
Ming R, Hou S, Feng Y, Yu Q, Dionne-Laporte A, Saw JH, Senin P, Wang W, Ly BV, Lewis KL, Salzberg SL, Feng L, Jones MR, Skelton RL, Murray JE, Chen C, Qian W, Shen J, Du P, Eustice M, Tong E, Tang H, Lyons E, Paull RE, Michael TP, Wall K, Rice DW, Albert H, Wang ML, Zhu YJ (2008) The draft genome of the transgenic tropical fruit tree papaya (Carica papaya Linnaeus). Nature 452:991–996
Porter BW, Aizawa KS, Zhu YJ, Christopher DA (2008) differentially expressed and new non-protein-coding genes from a Carica papaya root transcriptome survey. Plant Sci 174(1):38–50
Qi YJ, Zhong XH, Itaya A, Ding B (2004) Dissecting RNA silencing in protoplasts uncovers novel effects of viral suppressors on the silencing pathway at the cellular level. Nucleic Acids Res 32(22):e179
Sheen J (1995) Methods for mesophyll and bundle sheath cell separation. Methods Cell Biol 49:305–314
Sheen J (2001) Signal transduction in maize Arabidopsis mesophyll protoplasts. Plant Physiol 127:1466–1475
Smith NA, Singh SP, Wang MB (2000) Gene expression: total silencing by intron-spliced hairpin. Nature 407:319–320
Tougou M, Furutani N, Yamagishi N, Shizukawa Y, Takahata Y, Hidaka S (2006) Development of resistant transgenic soybeans with inverted repeat-coat protein genes of soybean dwarf virus. Plant Cell Rep 25:1213–1218
Tripathi S, Bau HJ, Chen LF, Yeh SD (2004) The ability of papaya ring spot virus strains overcoming the transgenic resistance of papaya conferred by the coat protein gene is not correlated with higher degrees of sequence divergence from the transgene. Eur J Plant Pathol 110(9):871–882
Von Arnim AG, Deng XW, Stacey MG (1998) Cloning vectors for the expression of green fluorescent protein fusion proteins in transgenic plants. Gene 221:35–43
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2(7):1565–1572
Zhai ZY, Sooksa-nguan T, Olena K (2008) Vatamaniuk functional analysis of genes in protoplasts by RNAi. Plant Physiol Preview. doi: 10.1104/pp.108.130260 (Published on November 12)
Zrachya A, Kumar PP, Ramakrishnan U, Levy Y, Loyter A, Arazi T, Lapidot M, Gafni Y (2007) Production of siRNA targeted against TYLCV coat protein transcripts leads to silencing of its expression and resistance to the virus. Transgenic Res 16(3):385–398
Acknowledgments
We thank CRIRO industry provided the destination vectors. We are grateful for the State Key Laboratory of Agricultural Microbiology of Huazhong Agriculture University provided the pGA482 vector and the equipments of phosphorimager and isotope label. Thanks for SRF (Student Researching Fund) project of Huazhong Agriculture University supported this work. This work was funded by the national natural science foundation of china projects (No. 30571277 and No. 30771477).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, L., Wang, J., Liu, Z. et al. Silencing induced by inverted repeat constructs in protoplasts of Nicotiana benthamiana . Plant Cell Tiss Organ Cult 100, 139–148 (2010). https://doi.org/10.1007/s11240-009-9629-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-009-9629-4