Abstract
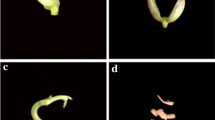
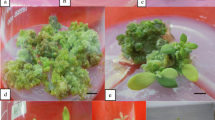
We report the first protocol for callus induction and shoot regeneration for Phaseolus lunatus L. cv. Wonder Bush and cv. Pole Sieva. We used different explants viz., epicotyls, cotyledons and hypocotyls. The medium used was MS basal medium with thidiazuron (0.5 mg l−1) and IAA (0.05 mg l−1) for the induction of callus followed by BAP (1.0 mg l−1) for the induction of shoots. Epicotyl explants showed the fastest response and the highest percentage of shoot regeneration. This protocol opens new biotechnological strategies to transfer economically important genes to this important crop species.

Similar content being viewed by others
Abbreviations
- TDZ:
-
Thidiazuron (1-phenyl-3-(1,2,3-thiadiazol-5-yl) urea)
- BAP:
-
6-Benzylaminopurine
- IAA:
-
Indole-3-acetic acid
- CIM:
-
Callus induction medium
- SIM:
-
Shoot induction medium
- SDM:
-
Shoot development medium
- MS:
-
Murashige and Skoog
References
Barik DP, Mohapatra U, Chand PK (2005) High frequency in vitro regeneration of Lathyrus sativus L. Biol Plant 49:637–639
Barik DP, Mohapatra U, Chand PK (2006) Direct shoot regeneration from epicotyl explants of grasspea (Lathyrus sativus). Aust J Bot 54:505–508
Barros LMG, Gama MICS, Goncalves CHRD, Barreto CC, Santana EF, Carneiro VTD (1997) Bean tissue culture for introduction of foreign genes. Pesqui Agropecu Bras 32:267–275
Cardenas-Avila ML, Verde-Star MJ, Foroughbakhch R, Gamez-Gonzalez H, Lozano-Martinez SJ, Nunez-Gonzalez MA, Hernandez-Pinero JL (2000) Variability of in vitro callus induction in four bean (Phaseolus vulgaris L.) varieties. Phyton 69:61–64
Christou P (1997) Biotechnology applied to grain legumes. Field Crops Res 53:83–97
De Clercq J, Zambre M, Van Montagu M, Dillen W, Angenon G (2002) An optimized Agrobacterium-mediated transformation procedure for Phaseolus acutifolius A. Gray. Plant Cell Rep 21:333–340
De Jaeger G, Scheffer S, Jacobs A, Zambre M, Zobell O, Goossens A, Depicker A, Angenon G (2002) Boosting heterologous protein production in transgenic dicotyledonous seeds using Phaseolus vulgaris regulatory sequences. Nat Biotechnol 20:1265–1268
Delgado-Sánchez P, Saucedo-Ruiz M, Guzman-Maldonado SH, Villordo-Pineda E, Gonzalez-Chavira M, Fraire-VelAzquez SF, costa-Gallegos JA, Mora-Aviles A (2006) An organogenic plant regeneration system for common bean (Phaseolus vulgaris L.). Plant Sci 170:822–827
Dillen W, DeClercq J, VanMontagu M, Angenon G (1996) Plant regeneration from callus in a range of Phaseolus acutifolius A. Gray genotypes. Plant Sci 118:81–88
Franklin CI, Trieu TN, Gonzales RA, Dixon RA (1991) Plant-regeneration from seedling explants of Green Bean (Phaseolus vulgaris L.) via organogenesis. Plant Cell Tissue Organ Cult 24:199–206
Kumar AS, Gamborg OL, Nabors MW (1988) Regeneration from long-term cell suspension cultures of tepary bean (Phaseolus acutifolius). Plant Cell Rep 7:322–325
Landi L, Mezzetti B (2006) TDZ, auxin and genotype effects on leaf organogenesis in Fragaria. Plant Cell Rep 25:281–288
Maffei ME, Mithofer A, Arimura GI, Uchtenhagen H, Bossi S, Bertea CM, Cucuzza LS, Novero M, Volpe V, Quadro S, Boland W (2006) Effects of feeding Spodoptera littoralis on lima bean leaves. III. Membrane depolarization and involvement of hydrogen peroxide. Plant Physiol 140:1022–1035
Maffei M, Mithofer A, Boland W (2007) Before gene expression: early events in plant-insect interaction. Trends Plant Sci 12:310–316
Malik KA, Saxena PK (1992) Somatic embryogenesis and shoot regeneration from intact seedlings of Phaseolus acutifolius A, P. aureus (L) Wilczek, P. coccineus L., and P. wrightii l L. Plant Cell Rep 11:163–168
Mohamed MF, Read PE, Coyne DP (1992) Plant-regeneration from in vitro culture of embryonic axis explants in common and Tepary Beans. J Am Soc Horticult Sci 117:332–336
Mohamed MF, Coyne DP, Read PE (1993) Shoot organogenesis in callus induced from pedicel explants of common Bean (Phaseolus-Vulgaris L). J Am Soc Horticult Sci 118:158–162
Mroginski E, Rey HY, Gonzalez AM, Mroginski LA (2004) Thidiazuron promotes in vitro plant regeneration of Arachis correntina (Leguminosae) via organogenesis. J Plant Growth Regul 23:129–134
Mundhara R, Rashid A (2006) Recalcitrant grain legume Vigna radiata, mung bean, made to regenerate on change of hormonal and cultural conditions. Plant Cell Tissue Organ Cult 85:265–270
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
San-Jose MC, Ballester A, Vieitez AM (2001) Effect of thidiazuron on multiple shoot induction and plant regeneration from cotyledonary nodes of chestnut. J Horticult Sci Biotechnol 76:588–595
Santalla M, Power JB, Davey MR (1998) Efficient in vitro shoot regeneration responses of Phaseolus vulgaris and P. coccineus. Euphytica 102:195–202
Somers DA, Samac DA, Olhoft PM (2003) Recent advances in legume transformation. Plant Physiol 131:892–899
Svetleva D, Velcheva M, Bhowmik G (2003) Biotechnology as a useful tool in common bean (Phaseolus vulgaris L.) improvement. Euphytica 131:189–200
Thomas TD, Philip B (2005) Thidiazuron-induced high-frequency shoot organogenesis from leaf-derived callus of a medicinal climber, Tylophora indica (Burm. F.) Merrill. In Vitro Cell Dev Biol Plant 41:124–128
Veltcheva M, Svetleva D, Petkova S, Perl A (2005) In vitro regeneration and genetic transformation of common bean (Phaseolus vulgaris L.) – Problems and progress. Sci Hortic 107:2–10
Zambre M, Geerts P, Maquet A, Van Montagu M, Dillen W, Angenon G (2001) Regeneration of fertile plants from callus in Phaseolus polyanthus Greenman (year bean). Ann Bot 88:371–377
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kanchiswamy, C.N., Maffei, M. Callus induction and shoot regeneration of Phaseolus lunatus L. cv. Wonder Bush and cv. Pole Sieva. Plant Cell Tiss Organ Cult 92, 239–242 (2008). https://doi.org/10.1007/s11240-007-9322-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-007-9322-4