Abstract
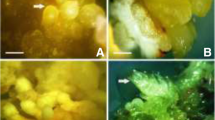
In the present study an efficient somatic embryogenesis method has been developed in Catharanthus roseus. Friable embryogenic callus was induced from hypocotyl of in vitro germinated seeds on Murashige and Skoog basal nutrient media supplemented with various auxins particularly 2,4-D (1.0 mg l−1). However, only NAA (1.0 mg l−1) produced somatic embryos in cultures. Embryo proliferation was even high on the same medium added with BAP. Cotyledonary somatic embryo germinated and converted into plantlets in BAP (0.5 mg l−1) added medium following a treatment with gibberellic acid (1.0 mg l−1) for maturation. Carbon sources and concentrations had a marked influence on maturation process. Plantlet conversion was better achieved when embryos were matured on 3% fructose or 3–6% maltose. The result discussed in this paper indicates that somatic embryos were produced in numbers and converted plantlets can be used as raw material, genetic modification to embryo precursor cell may improve alkaloid yield further.
Similar content being viewed by others
Abbreviations
- ANOVA:
-
analysis of variance
- BAP:
-
6-benzylaminopurine
- CPA:
-
chlorophenoxyacetic acid
- 2,4-D :
-
2,4-dichlorophenoxyacetic acid
- GA3 :
-
gibberellic acid
- IAA:
-
indole acetic acid
- MS:
-
Murashige and Skoog
- NAA:
-
naphthalene acetic acid
- 2,4,5-T:
-
2,4,5-trihydroxy-butyrophenone
References
Alemanno L, Berthouly M, Michaux-Ferriere N, (1997) A comparison between Theobroma cacao L. zygotic embryogenesis and somatic embryogenesis from floral explants In vitro Cell. Dev. Biol. Plant 33: 163–172
Blanc G, Michaux-Ferriere N, Teisson C, Lardet L, Carron MP, (1999) Effect of carbohydrate addition on the induction of somatic embryogenesis in Hevea brasiliensis Plant Cell Tiss. Org. cult. 59: 103–112
Corredoira E, Ballester A, Vieitez AM, (2003) Proliferation, maturation and germination of Castanea sativa Mill. somatic embryos originated from leaf explants Ann. Bot. 92: 129–136
Druart P, (1990) Improvement of somatic embryogenesis of cherry dwarf root stocks INMIL/GM, by use of different carbon sources Acta Horticult. 280: 125–129
Dudits D, Bogre L, Gyorgyey J, (1991) Molecular and cellular approaches to the analysis of plant embryo development from somatic cells in vitro J. Cell Sci. 99: 475–484
Fowler MW, (1983) Commercial application and economic aspects of mass plant cell culture In: Mantell SH, Smith H, (eds) Plant Biotechnology Cambridge University Press Cambridge pp. 3–38
Junaid A, Bhat MA, Mujib A, Sharma MP, (2004) Somatic embryogenesis study in Catharanthus roseus (L.) G.Don: an anticancerous plant In: Khan IA, Khanum A, (eds) Role of Biotechnology in Medicinal and Aromatic Plants. Vol. XI. Ukaaz Publication Hyderabad pp 98–106
Li XY, Feng H, Huang H, Murphy JB, Gbur Jr EE, (1998) Polyethylene glycol and maltose enhance somatic embryo maturation in loblolly pine (Pinus teada L.) In vitro Cell. Dev. Biol. Plant 34: 22–26
Lipavská H, Konrádová H, (2004) Somatic embryogenesis in conifers: the role of carbohydrate metabolism In Vitro Cell. Dev. Biol. Plant 40: 23–30
Michalczuk L, Cook TJ, Cohen JD (1992) Auxins levels at different stages of carrot somatic embryogenesis Phytochemistry 31: 1097–1103
Moreno PRH, Heijden Van der R, Verpoorte R, Vander Heijden R, (1995) Cell and tissue cultures of Catharanthus roseus, literatures survey II, updated from 1988–1993 Plant Cell Tiss. Org. Cult. 42: 1–25
Mujib A, Bandyopadhyay S, Jana BK, Ghosh PD, (1998) Direct and in vitro plant regeneration in Hippeastrum hybridum Plant Tiss. Cult. 8(1): 19–25
Mujib A, Ilah A, Gandotra N, Abdin MZ, (2003) In vitro application to improve alkaloids yield in Catharanthus roseus In: Govil JN, Kumar PA, Singh VK, (eds) Biotechnology and Genetic Engineering. Vol IV. Sci. Tech Publishing, Texas, pp 415–439
Murashige T, Skoog F, (1962) A revised medium of rapid growth and bioassay with tobacco tissue culture Physiol. Plant 15: 473–497
Norgaard JV, (1997) Somatic embryo maturation and plant regeneration in Abies nordmanniana LK Plant Sci. 124: 211–221
Nomura K, Komamine A, (1995) Physiological and biochemical aspects of somatic embryogenesis In: Thorpe TA, (eds) In Vitro Embryogenesis in Plants. Kluwer Academic Publishers, Dordrecht, pp 249–266
Pasternak TP, Prinsen E, Ayaydin F, Miskolezi P, Potters G, Asard H, Van Onckelen A, Dudits D, Feher A, (2002) The role of auxin, pH, and stress in the activation of embryogenic cell division in leaf protoplast derived cells of alfalfa Plant Physiol. 129: 1807–1819
Robichaud RL, Lessard VC, Merkle SA, (2004) Treatments affecting maturation and germination of American chestnut somatic embryos J. Plant Physiol. 161: 957–969
Rommens CM, Humara JM, Ye J, Yan H, Richael C, Zhang L, Perry R, Swords K, (2004) Crop improvement through modification of the plant’s own genome Plant Physiol. 135: 421–431
Rtienne H, Montoro P, Michaux-Ferriere N, Carron MP, (1993) Effect of desication medium osmolarity and abscisic acid on the maturation of Hivea brasiliensis somatic embryos J. Exp. Bot. 44: 1613–1619
Varga P, Badea EM, (1992) In vitro plant regeneration methods in alfalfa breeding Euphytica 59: 119–123
Walter C, (2004) Genetic engineering in conifer forestry: technical and social consideration In Vitro Cell. Dev. Biol. Plant 40: 434–441
Xing Z, Powell WA, Maynard CA, (1999) Development and germination of American chestnut somatic embryos Plant Cell Tiss. Org. Cult. 57: 47–55
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Junaid, A., Mujib, A., Bhat, M. et al. Somatic embryo proliferation, maturation and germination in Catharanthus roseus . Plant Cell Tiss Organ Cult 84, 325–332 (2006). https://doi.org/10.1007/s11240-005-9041-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-005-9041-7