Abstract
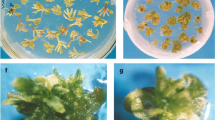
Echinops Asteraceae/Compositae is widespread throughout southern Europe and Asia. It is a native plant in Israel and usually found as a weed in wheat fields. Due to the need of the plants for vernalization, horticultural practice is to chill the crowns for 2 month before planting to achieve early flowering. A method for the propagation of Echinops through axillary buds and root cutting cultures is described in this manuscript. Best conditions for propagation were using MS salt medium supplemented with 3 sucrose and 1 mg l-1 IAA plus 0.5 mg l-11 BA under 16 h of cool fluorescent light. Root cuttings produced shoots all along the root but each growth regulator combination affected where roots will sprout shoots. Rooted plants were successfully acclimatized after propagating on the above medium and eventually flowered. Agrobacterium transformation of Echinops was successfully achieved and expression of GUS reporter gene was tested under the control of two constitutive promoters.
Similar content being viewed by others
Abbreviations
- BA:
-
6-Benzyladenine
- CTAB:
-
Hexadecyltrimethylammonium bromide
- EDTA:
-
Na2-Ethylenediaminetetraacitic dihydrate
- IAA:
-
Indole-3-acetic acid
- IBA:
-
3-Indole butyric acid
- MS:
-
Murashige and Skoog salt medium
- NAA:
-
1-Naphtalene acetic acid
- TDZ:
-
Thidiazuron
- WPM:
-
Woody Plant Medium
- β-ME:
-
β-Mercaptoethanol
References
Duzenli A and Cakan H (2001). Flora of Mount Musa (Hatay-Turkey). Turk. J. Bot. 25: 285–309
Famiani F, Ferradini N, Staffolani P and Standardi A (1994). Effect of leaf excision time and age, BA concentration and dark treatments on in vitro shoot regeneration of M.26 apple rootstock. J. Hortic. Sci. 69: 679–685
Garbarino JE, Oosumi T and Belknap WR (1995). Isolation of a polyubiquitin promoter and its expression in transgenic potato plants. Plant physiol 109: 1371–1378
Hahn P and Naser K (1979). Cultivation of herbaceous perennials for production of cut flowers in the open-species worth recommending for cut flower supply in the spring and summer months. Gartenbau Wissenschaf 26: 341–344
Iversen RR and Weiler TC (1994). Strategies to force flowering of six herbaceous garden perennials. Hort Technol. 4: 61–65
Jefferson RA (1987). Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol. Biol. Rep. 5: 387–405
Ke J, Kahn R, Johnson T, Somers DA and Das A (2001). High-efficiency gene transfer to recalcitrant plants by Agrobacrerium tumefaciens Plant Cell Rep. 20: 150–156
Lin HS, Jeu MJ and Jacobsen E (1998). Fromation of shoots from leaf axils of Alstroemeria: the effect of the position on the stem. Plant Cell, Tiss.-Org.-Cult. 52: 165–169
Ma B, Tatumoto I, Ogawa T, Morikawa T, Ma B (2002) Micro-propagation using leaf blades in genus Lycoris. Scientific report of the graduate school of agriculture and biological sciences, Osaka, Japan. 54: 1–5
McCown BH and Llyod C (1981). Woody Plant Medium (WPM): a revised mineral nutrient formulation for microculture of woody plant species. Hort. Sci. 16: 453
Murch SJ, Wierenga EJ, El-Demerdash MA and Saxena PK (2003). In vitro propagation of the Egyptian medicinal plant, Echinops spinosissimus Turra. Plant Cell Tis. Org. Cul. 74(1): 81–86
Pan ZG, Liu CZ., MUrch SJ, EL-Demerdash M and Saxena PK (2003). Plant regeneration from mesophyll protoplasts of the Egyptian medicinal plants Artemisia judaica L. and Echinops spinosissimus Turra. Plant Sci. 165(4): 681–687
Pedroso MC and Pais MS (1993). Direct embryo formation in leaves of Camellia japonica L. Plant Cell Rep. 12: 639–643
Rimbau V, Cerdan C, Vila R and Iglesias J (1999). Antiinflammatory activity of some extracts from plants used in the traditional medicine of North-African countries (II). Phytother. Res. 13: 128–132
Sathiyamoorthy P, Lugasi-Evgi H, Schlesinger P, Kedar I, Gopas J, Pollack Y and Golan-Goldhirsh A (1999). Screening for cytotoxic and antimalarial activities in desert plants of the Negev and bedouin market plant products. Pharm. Biol. 37: 188–195
Shah MJ, Subhan F, Tahir F and Alam W (1997). Male contraceptives from traditional drugs (plant based). Hamdard Medicus. 40: 34–36
Sheidai M, Nasirzadeh A and Kheradnam M (2000). Karyotypic study of Echinops (Asteraceae) in Fars province. Iran. Bot. J. Lin. Soc. 134: 453–463
Shibli RA, Ajlouni MMA and Obeidat AA (2000). Direct regeneration from wild pear (Pyrus syniaca) leaf explants. Adv. Hortic. Sci. 14: 12–18
Surridge C (2004). Plant development: the flowers that bloom in the spring. Nature 427: 112
Tel-zur N, Abbo S, Myslabodski D and Mizrahi Y (1999). Modified CTAB procedure for DNA isolation from epiphytic cacti of the genera Hylocereus and Selenicereus (Cactaceae). Plant Mol. Biol. Report. 17: 249–254
Wallerstein I (1983). The flowering of Echinops blancheanus Boiss. Hassadeh. (in Hebrew) 63: 992–993
Weiss D. and Ziv O (1999). Agrothechnical and physiological factors that are involved in the control of flowering in Echinops Dapi Maida (in Hebrew) 15: 63–64
Zhang Y and Zhang YH (1999). Effect of different explants and phyto-hormones on organogenesis in scarlet eggplant (Solanum aethiopicum L). J. Yunnan Agric. U. 14: 279–283
Author information
Authors and Affiliations
Corresponding author
Additional information
This is publication number 05/03 from Horticulture Institute ARO.
Rights and permissions
About this article
Cite this article
Dalia, E., Michele, Z., Xinlu, C. et al. Regeneration and Transformation of Echinops cv Veitsch Blue. Plant Cell Tiss Organ Cult 85, 1–9 (2006). https://doi.org/10.1007/s11240-004-2564-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-004-2564-5