Abstract
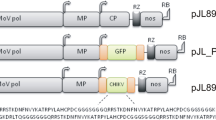
To optimize the efficiency of expression of foreign proteins using Potato virus X (PVX) -- based vector, the gene for the coat protein (CP) of other virus (Potato virus A, PVA) was cloned into the vector, propagated in E. coli and subsequently inoculated or agroinfected into the host plants. Host range studies showed that the best host plant is N. benthamiana. By means of RT PCR the presence and the stability of the construct were tested. Both ELISA and Western blot analysis were applicable for expressed protein detection. Expression level of PVA CP achieved approximately 5--10 per mille of total soluble proteins. The results demonstrated that agroinfection is the most suitable method for the propagation of our model gene using PVX--based vectors.
Similar content being viewed by others
References
Al-Kaff NS, Covey SN, Kreike MM, Page AM, Pinder R & Dale PJ (1998) Transcriptional and posttranscriptional plant gene silencing in response to a pathogen. Science 279: 2113–2115
Angell SM & Baulcombe DC (1997) Consistent gene silencing in transgenic plants expressing a replicating potato virus X RNA. EMBO J. 16: 3675–3684
Clarck MF & Adams AN (1977) Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol. 34: 475–483
Čeřovská N, Moravec T & Velemínský J (2002) Polyclonal antibodies to a recombinant coat protein of Potato virus A. Acta Virol. 46: 147–151
Čeřovská N, Petrzik K, Moravec T & Mraz I (1998) Potato virus A detection by reverse transcription polymerase chain reaction. Acta Virol. 42: 83–85
Filigarová M, Čeřovská N, Franěk F & Dědič P (1994) Reactivity of monoclonal antibodies to potato virus A in various types of enzyme linked immunosorbent assay. Potato Res. 37: 135–147
Fischer R, Vaquero-Martin C, Sack M, Drossard J, Emans N & Commandeur U (1999) Toward molecular farming in the future: transient protein expression in plants. Biotechnol. Appl. Biochem. 30: 113–116
Hirano H & Watanabe T (1990) Microsequencing of proteins electrotransferred onto immobilizing matrices from polyacrylamide gel electrophoresis: application to an insoluble protein. Electrophoresis 11: 573–580
Hobbs SL, Kpodar P & DeLong CM (1990) The effect of TDNA copy number, position and methylation on reporter gene expression in tobacco transformants. Plant Mol. Biol. 15: 851–864
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Liu L & Lomonossoff G (2002) Agroinfection as a rapid method for propagating Cowpeea mosaic virus-based constructs. J. Virol. Meth. 105: 343–348
Mayer MP (1995) A new set of useful cloning and expression vectors derived from pBlueScript. Gene 163: 41–46
O'Brien GJ, Bryant CJ, Voogd Ch, Greenberg HB, Gardner RC & Bellamy AR (2000) Rotavisrus VP6 expressed by PVX vectors in Nicotiana benthamiana coats PVX rods and also assembles into viruslike particles. Virology 270: 444–453
Puurand U, Makinen K, Paulin L & Saarma M (1994) The nucleotide sequence of potato virus A genomic RNA and its sequence similarities with other potyviruses. J. Gen. Virol. 75: 457–461
Ratcliff F, Martin-Hernandez AM & Baulcombe DC (2001) Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J. 25: 237–245
Sambrook J, Fritsch EF & Maniatis T<nt>(eds)</nt> (1989) Molecular Cloning. A Laboratory Manual, 2nd edn. Cold Spring Harbor, NY
Subr Z, Moravec T & Cerovská N (1996) Monoclonal antibodies against potato virus A – immunoblot analysis. Acta Virol. 40: 289–292
Yusibov V, Shiprasad S, Turpen TH, Dawson W & Koprowski H (1999) Curr. Top. Microbiol. Immunol. 240: 81–94
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Čeřovská, N., Pečenková, T., Moravec, T. et al. Transient expression of heterologous model gene in plants using Potato virus X-based vector. Plant Cell, Tissue and Organ Culture 79, 147–152 (2004). https://doi.org/10.1007/s11240-004-0654-z
Issue Date:
DOI: https://doi.org/10.1007/s11240-004-0654-z