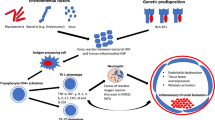
Abstract
Deep vein thrombosis (DVT) is a common complication in hematologic malignancies and immunologic disorders that coagulation and inflammatory factors play a crucial role in its occurrence. The content used in this article has been obtained by PubMed database and Google Scholar search engine of English-language articles (1980–2019) using the “Deep vein thrombosis,” “Hematologic malignancies,” “Immunologic disorders” and “Treatment.” Increased levels of coagulation factors, the presence of genetic disorders, or the use of thrombotic drugs that stimulate coagulation processes are risk factors for the development of DVT in patients with hematologic malignancies. Inflammatory and auto-anti-inflammatory factors, along with coagulant factors, play an essential role in the formation of venous thrombosis in patients with immunological disorders by increasing the recruitment of inflammatory cells and adhesion molecules. Therefore, anti-coagulants in hematologic malignancies and immunosuppressants in immune disorders can reduce the risk of developing DVT by reducing thrombotic and inflammatory activity. Considering the increased risk of DVT due to impaired coagulation and inflammation processes, analysis of coagulation and inflammatory factors have prognostic values in patients with immunologic deficiencies and hematologic malignancies. Evaluation of these factors as diagnostic and prognostic biomarkers in the prediction of thrombotic events could be beneficial in implementing effective treatment strategies for DVT.

Similar content being viewed by others
Abbreviations
- MM:
-
Multiple myeloma
- PNH:
-
Paroxysmal nocturnal hemoglobinuria
- MPNs:
-
Myeloproliferative neoplasms
- DM:
-
Diabetes mellitus
- RA:
-
Rheumatoid arthritis
- SLE:
-
Systemic lupus erythematosus
- LMWH:
-
Low molecular weight heparin
- UFH:
-
Unfractionated heparin
- TNF-α:
-
Tumor necrosis factor-alpha
- NSAIDs:
-
Nonsteroidal anti-inflammatory drugs
- U-PAR:
-
Urokinase-type plasminogen activator receptor
- PAI-1:
-
Plasminogen activator inhibitor-1
- TKA:
-
Total knee arthroplasty
- CRP:
-
C-reactive protein
- NETs:
-
Neutrophil extracellular traps
- PIG-A:
-
Phosphatidylinositol glycan-class A
- G-CSF:
-
Granulocyte-colony stimulating factor
- GM-CSF:
-
Granulocyte macrophage colony-stimulating factor
- APS:
-
Anti-phospholipid syndrome
- TFPI:
-
Tissue factor pathway inhibitor
References
Cushman M, Tsai AW, White RH, Heckbert SR, Rosamond WD, Enright P et al (2004) Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med 117(1):19–25
Ramagopalan SV, Wotton CJ, Handel AE, Yeates D, Goldacre MJ (2011) Risk of venous thromboembolism in people admitted to hospital with selected immune-mediated diseases: record-linkage study. BMC Med 9(1):1
Di Nisio M, van Es N, Büller HR (2016) Deep vein thrombosis and pulmonary embolism. Lancet 388(10063):3060–3073
Kyrle PA, Eichinger S (2005) Deep vein thrombosis. Lancet 365(9465):1163–1174
Budnik I, Brill A (2018) Immune factors in deep vein thrombosis initiation. Trends Immunol. https://doi.org/10.1016/j.it.2018.04.010
van Tilburg NH, Rosendaal FR, Bertina RM (2000) Thrombin activatable fibrinolysis inhibitor and the risk for deep vein thrombosis. Blood 95(9):2855–2859
Tang J, Zhu W, Mei X, Zhang Z (2018) Plasminogen activator inhibitor-1: a risk factor for deep vein thrombosis after total hip arthroplasty. J Orthop Surg Res 13(1):8
Eichinger S, Weltermann A, Mannhalter C, Minar E, Bialonczyk C, Hirschl M et al (2002) The risk of recurrent venous thromboembolism in heterozygous carriers of factor V Leiden and a first spontaneous venous thromboembolism. Arch Intern Med 162(20):2357–2360
Tamaki H, Khasnis A (2015) Venous thromboembolism in systemic autoimmune diseases: a narrative review with emphasis on primary systemic vasculitides. Vasc Med 20(4):369–376
Agirbasli M, Inanc N, Baykan O, Direskeneli H (2006) The effects of TNF alpha inhibition on plasma fibrinolytic balance in patients with chronic inflammatory rheumatical disorders. Clin Exp Rheumatol 24(5):580–583
Elice F, Rodeghiero F (2012) Hematologic malignancies and thrombosis. Thromb Res 129(3):360–366
Yusuf HR, Hooper WC, Beckman MG, Zhang QC, Tsai J, Ortel TL (2014) Risk of venous thromboembolism among hospitalizations of adults with selected autoimmune diseases. J Thromb Thrombolysis 38(3):306–313
Tsai AW, Cushman M, Rosamond WD, Heckbert SR, Tracy RP, Aleksic N et al (2002) Coagulation factors, inflammation markers, and venous thromboembolism: the longitudinal investigation of thromboembolism etiology (LITE). Am J Med 113(8):636–642
Martinelli I, Battaglioli T, Razzari C, Mannucci P (2007) Type and location of venous thromboembolism in patients with factor V Leiden or prothrombin G20210A and in those with no thrombophilia. J Thromb Haemost 5(1):98–101
Croles FN, Borjas-Howard J, Nasserinejad K, Leebeek FW, Meijer K (2018) Risk of venous thrombosis in antithrombin deficiency: a systematic review and Bayesian meta-analysis. Semin Thromb Hemost. https://doi.org/10.1055/s-0038-1625983
Cantu C, Alonso E, Jara A, Martínez L, Ríos C, de ML Fernandez et al (2004) Hyperhomocysteinemia, low folate and vitamin B12 concentrations, and methylene tetrahydrofolate reductase mutation in cerebral venous thrombosis. Stroke 35(8):1790–1794
Radulescu VC, D’Orazio JA (2016) Venous thromboembolic disease in children and adolescents. In: Thrombosis and embolism: from research to clinical practice. Springer, Cham, p 149–165
von Brühl M-L, Stark K, Steinhart A, Chandraratne S, Konrad I, Lorenz M et al (2012) Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J Exp Med 209(4):819–835
Brill A, Fuchs TA, Chauhan AK, Yang JJ, De Meyer SF, Köllnberger M et al (2011) von Willebrand factor-mediated platelet adhesion is critical for deep vein thrombosis in mouse models. Blood 117(4):1400–1407
Brill A, Yesilaltay A, De Meyer SF, Kisucka J, Fuchs TA, Kocher O et al (2012) Extrahepatic high-density lipoprotein receptor SR-BI and apoA-I protect against deep vein thrombosis in mice. Arterioscler Thromb Vasc Biol 32(8):1841–1847
Deguchi H, Pecheniuk NM, Elias DJ, Averell PM, Griffin JH (2005) High-density lipoprotein deficiency and dyslipoproteinemia associated with venous thrombosis in men. Circulation 112(6):893–899
Falanga A, Marchetti M (2014) Thrombosis in myeloproliferative neoplasms. Semin Thromb Hemost. https://doi.org/10.1055/s-0034-1370794
Hill A, Kelly RJ, Hillmen P (2013) Thrombosis in paroxysmal nocturnal hemoglobinuria. Blood 121(25):4985–4996
Kristinsson SY (2010) Thrombosis in multiple myeloma. ASH Educ Program Book 2010(1):437–444
Colombo R, Gallipoli P, Castelli R (2014) Thrombosis and hemostatic abnormalities in hematological malignancies. Clin Lymphoma Myeloma Leuk 14(6):441–450
Franchini M (2015) Thromboembolic risk in hematological malignancies. Clin Chem Lab Med 53(8):1139–1147
Castelli R, Ferrari B, Cortelezzi A, Guariglia A (2010) Thromboembolic complications in malignant haematological disorders. Curr Vasc Pharmacol 8(4):482–494
Carrabba G, Riva M, Conte V, Di Cristofori A, Caroli M, Locatelli M et al (2018) Risk of post-operative venous thromboembolism in patients with meningioma. J Neurooncol 138(2):401–406
Hoefnagel D, Kwee LE, van Putten EH, Kros JM, Dirven CM, Dammers R (2014) The incidence of postoperative thromboembolic complications following surgical resection of intracranial meningioma. A retrospective study of a large single center patient cohort. Clin Neurol Neurosurg 123:150–154
Kristinsson SY, Fears TR, Gridley G, Turesson I, Mellqvist U-H, Björkholm M et al (2008) Deep vein thrombosis after monoclonal gammopathy of undetermined significance and multiple myeloma. Blood 112(9):3582–3586
Palumbo A, Anderson K (2011) Multiple myeloma. N Engl J Med 364(11):1046–1060
Carr ME Jr, Dent RM, Carr SL (1996) Abnormal fibrin structure and inhibition of fibrinolysis in patients with multiple myeloma. J Lab Clin Med 128(1):83–88
Auwerda JJ, Sonneveld P, de Maat MP, Leebeek FW (2007) Prothrombotic coagulation abnormalities in patients with newly diagnosed multiple myeloma. Haematologica 92(2):279–280
Elice F, Fink L, Tricot G, Barlogie B, Zangari M (2006) Acquired resistance to activated protein C (aAPCR) in multiple myeloma is a transitory abnormality associated with an increased risk of venous thromboembolism. Br J Haematol 134(4):399–405
Leebeek FW (2016) Update of thrombosis in multiple myeloma. Thromb Res 140(Suppl 1):S76–S80
Bagratuni T, Kastritis E, Politou M, Roussou M, Kostouros E, Gavriatopoulou M et al (2013) Clinical and genetic factors associated with venous thromboembolism in myeloma patients treated with lenalidomide-based regimens. Am J Hematol 88(9):765–770
van Marion AM, Auwerda JJ, Minnema MC, van Oosterom R, Adelmeijer J, de Groot PG et al (2005) Hypofibrinolysis during induction treatment of multiple myeloma may increase the risk of venous thrombosis. Thromb Haemost 94(12):1341–1343
Ghosh N, Ye X, Ferguson A, Huff CA, Borrello I (2011) Bortezomib and thalidomide, a steroid free regimen in newly diagnosed patients with multiple myeloma. Br J Haematol 152(5):593–599
Rosse W (2000) Paroxysmal nocturnal hemoglobinuria. In: Hematology: basic principles and practice, 3rd edn. Churchill Livingstone, New York, p 331–342
Risitano AM (2012) Paroxysmal nocturnal hemoglobinuria and other complement-mediated hematological disorders. Immunobiology 217(11):1080–1087
Risitano AM (2013) Paroxysmal nocturnal hemoglobinuria and the complement system: recent insights and novel anticomplement strategies. Complement therapeutics. Adv Exp Med Biol 735:155–172
Lewis DA, Nyska A, Potti A, Hoke HA, Klemp KF, Ward SM et al (2006) Hemostatic activation in a chemically induced rat model of severe hemolysis and thrombosis. Thromb Res 118(6):747–753
Studt J-D, Hovinga JAK, Antoine G, Hermann M, Rieger M, Scheiflinger F et al (2005) Fatal congenital thrombotic thrombocytopenic purpura with apparent ADAMTS13 inhibitor: in vitro inhibition of ADAMTS13 activity by hemoglobin. Blood 105(2):542–544
Haybar H, Khodadi E, Kavianpour M, Saki N (2018) Mutations and common polymorphisms in ADAMTS13 and vWF genes responsible for increasing risk of thrombosis. Cardiovasc Haematol Disord Drug Targets (Formerly Curr Drug Targets Cardiovasc Hematol Disord) 18(3):176–181
Catani MV, Bernassola F, Rossi A, Melino G (1998) Inhibition of clotting factor XIII activity by nitric oxide. Biochem Biophys Res Commun 249(1):275–278
Shao J, Miyata T, Yamada K, Hanafusa N, Wada T, Gordon KL et al (2001) Protective role of nitric oxide in a model of thrombotic microangiopathy in rats. J Am Soc Nephrol 12(10):2088–2097
Ploug M, Plesner T, Ronne E, Ellis V, Hoyer-Hansen G, Hansen NE et al (1992) The receptor for urokinase-type plasminogen activator is deficient on peripheral blood leukocytes in patients with paroxysmal nocturnal hemoglobinuria. Blood 79(6):1447–1455
Sloand EM, Pfannes L, Scheinberg P, More K, Wu CO, Horne M et al (2008) Increased soluble urokinase plasminogen activator receptor (suPAR) is associated with thrombosis and inhibition of plasmin generation in paroxysmal nocturnal hemoglobinuria (PNH) patients. Exp Hematol 36(12):1616–1624
Maroney S, Cunningham A, Ferrel J, Hu R, Haberichter S, Mansbach C et al (2006) A GPI-anchored co-receptor for tissue factor pathway inhibitor controls its intracellular trafficking and cell surface expression. J Thromb Haemost 4(5):1114–1124
Maroney SA, Mast AE (2008) Expression of tissue factor pathway inhibitor by endothelial cells and platelets. Transfus Apher Sci 38(1):9–14
Tefferi A, Elliott M (2007) Thrombosis in myeloproliferative disorders: prevalence, prognostic factors, and the role of leukocytes and JAK2V617F. Semin Thromb Hemost. https://doi.org/10.1055/s-2007-976165
Colaizzo D, Tiscia GL, Bafunno V, Amitrano L, Vergura P, Lupone MR et al (2011) Sex modulation of the occurrence of jak2 v617f mutation in patients with splanchnic venous thrombosis. Thromb Res 128(3):233–236
Akpan IJ, Stein BL (2018) Splanchnic vein thrombosis in the myeloproliferative neoplasms. Curr Hematol Malig Rep 13(3):183–190
Smalberg JH, Arends LR, Valla DC, Kiladjian J-J, Janssen HL, Leebeek FW (2012) Myeloproliferative neoplasms in Budd–Chiari syndrome and portal vein thrombosis: a meta-analysis. Blood 120(25):4921–4928
Bagheropur S, Ehsanpour A, Birgani MT, Saki N (2018) JAK2V617F allele burden: innovative concept in monitoring of myeloproliferative neoplasms. Mag Eur Med Oncol 11(2):152–157
Villani L, Bergamaschi G, Primignani M, Rosti V, Carolei A, Poletto V et al (2012) JAK2 46/1 haplotype predisposes to splanchnic vein thrombosis-associated BCR-ABL negative classic myeloproliferative neoplasms. Leuk Res 36(1):e7–e9
Spaander M, Hoekstra J, Hansen B, Van Buuren H, Leebeek F, Janssen H (2013) Anticoagulant therapy in patients with non-cirrhotic portal vein thrombosis: effect on new thrombotic events and gastrointestinal bleeding. J Thromb Haemost 11(3):452–459
Shahrabi S, Behzad MM, Jaseb K, Saki N (2018) Thrombocytopenia in leukemia: pathogenesis and prognosis. Histol Histopathol 33(9):895–908
Maleknia M, Valizadeh A, Pezeshki SMS, Saki N (2019) Immunomodulation in leukemia: cellular aspects of anti-leukemic properties. Clin Transl Oncol Off Publ Fed Span Oncol Soc Natl Cancer Inst Mex. https://doi.org/10.1007/s12094-019-02132-9
Kwaan HC, Cull EH (2014) The coagulopathy in acute promyelocytic leukaemia—what have we learned in the past twenty years. Best Pract Res Clin Haematol 27(1):11–18
Koch A, Meesters MI, Scheller B, Boer C, Zacharowski K (2013) Systemic endotoxin activity correlates with clot formation: an observational study in patients with early systemic inflammation and sepsis. Crit Care 17(5):R198
Shahrabi S, Maleknia M, Tavakolifar Y, Zayeri DZ, Saki N (2019) Neutropenia and leukemia development: genetic risk factors and prognosis. Leuk Lymphoma. https://doi.org/10.1080/10428194.2019.1630622
LeBlanc R, Roy J, Demers C, Vu L, Cantin G (1999) A prospective study of G-CSF effects on hemostasis in allogeneic blood stem cell donors. Bone Marrow Transplant 23(10):991
Topcuoglu P, Arat M, Dalva K, Özcan M (2004) Administration of granulocyte-colony-stimulating factor for allogeneic hematopoietic cell collection may induce the tissue factor-dependent pathway in healthy donors. Bone Marrow Transplant 33(2):171
De Stefano V, Sora F, Rossi E, Chiusolo P, Laurenti L, Fianchi L et al (2005) The risk of thrombosis in patients with acute leukemia: occurrence of thrombosis at diagnosis and during treatment. J Thromb Haemost 3(9):1985–1992
Rajaei E, Jalali MT, Shahrabi S, Asnafi AA, Pezeshki SMS (2019) HLAs in autoimmune diseases: dependable diagnostic biomarkers? Curr Rheumatol Rev. https://doi.org/10.2174/1573397115666190115143226
Amin Asnafi A, Jalali MT, Pezeshki SMS, Jaseb K, Saki N (2019) The association between human leukocyte antigens and ITP, TTP, and HIT. J Pediatr Hematol Oncol 41(2):81–86
Chung W-S, Lin C-L, Kao C-H (2015) Diabetes increases the risk of deep-vein thrombosis and pulmonary embolism. Thromb Haemost 114(10):812–818
Chung WS, Lin CL, Chang SN, Lu CC, Kao CH (2014) Systemic lupus erythematosus increases the risks of deep vein thrombosis and pulmonary embolism: a nationwide cohort study. J Thromb Haemost 12(4):452–458
Tomasson G, Monach PA, Merkel PA (2009) Thromboembolic disease in vasculitis. Curr Opin Rheumatol 21(1):41
Chung W-S, Peng C-L, Lin C-L, Chang Y-J, Chen Y-F, Chiang JY et al (2014) Rheumatoid arthritis increases the risk of deep vein thrombosis and pulmonary thromboembolism: a nationwide cohort study. Ann Rheum Dis 73(10):1774–1780
Esmon CT (2005) The interactions between inflammation and coagulation. Br J Haematol 131(4):417–430
Slungaard A, Vercellotti GM, Tran T, Gleich GJ, Key NS (1993) Eosinophil cationic granule proteins impair thrombomodulin function. A potential mechanism for thromboembolism in hypereosinophilic heart disease. J Clin Investig 91(4):1721–1730
Khanna HK, Stevens AC (2017) Diabetic myonecrosis: a rare complication of diabetes mellitus mimicking deep vein thrombosis. Am J Case Rep 18:38
Stein PD, Goldman J, Matta F, Yaekoub AY (2009) Diabetes mellitus and risk of venous thromboembolism. Am J Med Sci 337(4):259–264
Grant P (2007) Diabetes mellitus as a prothrombotic condition. J Intern Med 262(2):157–172
Ferreiro JL, Gómez-Hospital JA, Angiolillo DJ (2010) Platelet abnormalities in diabetes mellitus. Diabetes Vasc Dis Res 7(4):251–259
Avogaro A, Albiero M, Menegazzo L, de Kreutzenberg S, Fadini GP (2011) Endothelial dysfunction in diabetes: the role of reparatory mechanisms. Diabetes Care 34(Supplement 2):S285–S290
Stavenuiter F, Bouwens EA, Mosnier LO (2013) Down-regulation of the clotting cascade by the protein C pathway. Hematol Educ/Congr Eur Hematol Assoc Eur Hematol Assoc Congr Educ Program NIH Public Access 7:365–374
Esmon CT (2012) Protein C anticoagulant system—anti-inflammatory effects. Semin Immunopathol. https://doi.org/10.1007/s00281-011-0284-6
Charles MA, Morange P, Eschwège E, André P, Vague P, Juhan-Vague I (1998) Effect of weight change and metformin on fibrinolysis and the von Willebrand factor in obese nondiabetic subjects: the BIGPRO1 Study. Biguanides and the prevention of the risk of obesity. Diabetes Care 21(11):1967–1972
Standeven KF, Ariëns RA, Whitaker P, Ashcroft AE, Weisel JW, Grant PJ (2002) The effect of dimethylbiguanide on thrombin activity, FXIII activation, fibrin polymerization, and fibrin clot formation. Diabetes 51(1):189–197
Bailey CJ (2008) Metformin: effects on micro and macrovascular complications in type 2 diabetes. Cardiovasc Drugs Ther 22(3):215–224
Lu D-Y, Huang C-C, Huang P-H, Chung C-M, Lin S-J, Chen J-W et al (2014) Metformin use in patients with type 2 diabetes mellitus is associated with reduced risk of deep vein thrombosis: a non-randomized, pair-matched cohort study. BMC Cardiovasc Disord 14(1):187
Zhao Z, Wang S, Ma W, Kong G, Zhang S, Tang Y et al (2014) Diabetes mellitus increases the incidence of deep vein thrombosis after total knee arthroplasty. Arch Orthop Trauma Surg 134(1):79–83
Gandhi R, Razak F, Tso P, Davey JR, Mahomed NN (2009) Metabolic syndrome and the incidence of symptomatic deep vein thrombosis following total knee arthroplasty. J Rheumatol 36(10):2298–2301
Bhasin R, Ghobrial I (2013) Diabetic myonecrosis: a diagnostic challenge in patients with long-standing diabetes. J Community Hosp Intern Med Perspect 3(1):20494
Mukherjee S, Aggarwal A, Rastogi A, Bhansali A, Prakash M, Vaiphei K et al (2015) Spontaneous diabetic myonecrosis: report of four cases from a tertiary care institute. Endocrinol Diabetes Metab Case Rep. https://doi.org/10.1530/EDM-15-0003
Baeten D, Peene I, Union A, Meheus L, Sebbag M, Serre G et al (2001) Specific presence of intracellular citrullinated proteins in rheumatoid arthritis synovium: relevance to antifilaggrin autoantibodies. Arthritis Rheum 44(10):2255–2262
Holmqvist ME, Neovius M, Eriksson J, Mantel Ä, Wållberg-Jonsson S, Jacobsson LT et al (2012) Risk of venous thromboembolism in patients with rheumatoid arthritis and association with disease duration and hospitalization. JAMA 308(13):1350–1356
Mukubo Y, Kawamata M (2004) Perioperative hypercoagulability in patients with rheumatoid arthritis: sonoclot study. J Anesth 18(1):62–64
Lee JK, Choi C-H (2012) Total knee arthroplasty in rheumatoid arthritis. Knee Surg Relat Res 24(1):1
Lee D-K, Kim H-J, Lee D-H (2016) Incidence of deep vein thrombosis and venous thromboembolism following TKA in rheumatoid arthritis versus osteoarthritis: a meta-analysis. PLoS ONE 11(12):e0166844
Bazzan M, Vaccarino A, Marletto F (2015) Systemic lupus erythematosus and thrombosis. Thromb J 13(1):16
Xu J, Lupu F, Esmon CT (2010) Inflammation, innate immunity and blood coagulation. Hämostaseologie 30(01):5–9
Brill A, Fuchs T, Savchenko A, Thomas G, Martinod K, De Meyer S et al (2012) Neutrophil extracellular traps promote deep vein thrombosis in mice. J Thromb Haemost 10(1):136–144
Male C, Chait P, Ginsberg JS, Hanna K, Andrew M, Halton J et al (2002) Comparison of venography and ultrasound for the diagnosis of asymptomatic deep vein thrombosis in the upper body in children. Thromb Haemost 87(04):593–598
Maufus M, Elias A, Barrellier M-T, Pernod G (2018) Diagnosis of deep vein thrombosis recurrence: ultrasound criteria. Thromb Res 161:78–83
Wells PS, Anderson DR, Rodger M, Forgie M, Kearon C, Dreyer J et al (2003) Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med 349(13):1227–1235
Wang X, Sundquist K, Elf JL, Strandberg K, Svensson PJ, Hedelius A et al (2016) Diagnostic potential of plasma microRNA signatures in patients with deep-vein thrombosis. Thromb Haemost 116(08):328–336
Streiff MB, Agnelli G, Connors JM, Crowther M, Eichinger S, Lopes R et al (2016) Guidance for the treatment of deep vein thrombosis and pulmonary embolism. J Thromb Thrombolysis 41(1):32–67
Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M et al (2015) Targeting CD38 with daratumumab monotherapy in multiple myeloma. N Engl J Med 373(13):1207–1219
de Latour RP, Mary JY, Salanoubat C, Terriou L, Etienne G, Mohty M et al (2008) Paroxysmal nocturnal hemoglobinuria: natural history of disease subcategories. Blood 112(8):3099–3106
Hillmen P, Young NS, Schubert J, Brodsky RA, Socié G, Muus P et al (2006) The complement inhibitor eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med 355(12):1233–1243
Griffin M, Munir T (2017) Management of thrombosis in paroxysmal nocturnal hemoglobinuria: a clinician’s guide. Ther Adv Hematol 8(3):119–126
De Stefano V, Finazzi G, Barbui T (2018) Antithrombotic therapy for venous thromboembolism in myeloproliferative neoplasms. Blood Cancer J 8(7):65
Napolitano M, Valore L, Malato A, Saccullo G, Vetro C, Mitra ME et al (2016) Management of venous thromboembolism in patients with acute leukemia at high bleeding risk: a multi-center study. Leuk Lymphoma 57(1):116–119
Emmi G, Silvestri E, Squatrito D, Amedei A, Niccolai E, D’Elios MM et al (2015) Thrombosis in vasculitis: from pathogenesis to treatment. Thromb J 13(1):15
Arida A, Fragiadaki K, Giavri E, Sfikakis PP (2011) Anti-TNF agents for Behcet’s disease: analysis of published data on 369 patients. Semin Arthritis Rheum. https://doi.org/10.1016/j.semarthrit.2010.09.002
Al-Ani B (2013) Simvastatin inhibits neutrophil degranulation induced by anti-neutrophil cytoplasm auto-antibodies and N-formyl-methionine-leucine-phenylalanine (fMLP) peptide. Saudi Med J 34(5):477–483
Kaiser R, Cleveland CM, Criswell LA (2009) Risk and protective factors for thrombosis in systemic lupus erythematosus: results from a large, multi-ethnic cohort. Ann Rheum Dis 68(2):238–241
Acknowledgements
We wish to thank all our colleagues in Allied Health Sciences School, Ahvaz Jundishapur University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
MM conceived the manuscript and revised it. HA, RGH, MSH, and MB wrote the manuscript and prepared tables and figures.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alipanahzadeh, H., Ghulamreza, R., Shokouhian, M. et al. Deep vein thrombosis: a less noticed complication in hematologic malignancies and immunologic disorders. J Thromb Thrombolysis 50, 318–329 (2020). https://doi.org/10.1007/s11239-019-02005-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-019-02005-6