Abstract
The recent development and approval of novel oral anticoagulants represents a significant success in the intelligent design of target-specific therapeutics. However, while these agents obviate many of the shortcomings of their predecessor (warfarin), they present novel challenges in pharmacologic management as well. Each was designed to have high oral bioavailability and high affinity for its target molecule, conveying significant anticoagulant effects. Yet, such unique drug–ligand binding, coupled with unfamiliar drug interactions and renal-based clearance, represent challenges to clinical management. The current review describes the development and pharmacokinetic properties of these agents, in the context of their clinical utility and pitfalls.


Similar content being viewed by others
References
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, Pogue J, Reilly PA, Themeles E, Varrone J, Wang S, Alings M, Xavier D, Zhu J, Diaz R, Lewis BS, Darius H, Diener HC, Joyner CD, Wallentin L (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361(12):1139–1151. doi:10.1056/NEJMoa0905561
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365(10):883–891. doi:10.1056/NEJMoa1009638
Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, Al-Khalidi HR, Ansell J, Atar D, Avezum A, Bahit MC, Diaz R, Easton JD, Ezekowitz JA, Flaker G, Garcia D, Geraldes M, Gersh BJ, Golitsyn S, Goto S, Hermosillo AG, Hohnloser SH, Horowitz J, Mohan P, Jansky P, Lewis BS, Lopez-Sendon JL, Pais P, Parkhomenko A, Verheugt FW, Zhu J, Wallentin L (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365(11):981–992. doi:10.1056/NEJMoa1107039
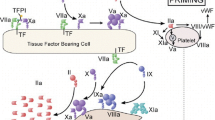
Davie EW, Fujikawa K, Kisiel W (1991) The coagulation cascade: initiation, maintenance, and regulation. Biochemistry 30(43):10363–10370
Perzborn E, Roehrig S, Straub A, Kubitza D, Misselwitz F (2011) The discovery and development of rivaroxaban, an oral, direct factor Xa inhibitor. Nat Rev Drug Discov 10(1):61–75. doi:10.1038/nrd3185
Weitz JI (2011) Factor Xa and thrombin as targets for new oral anticoagulants. Thromb Res 127(Suppl 2):S5–S12. doi:10.1016/s0049-3848(10)70147-x
Hauel NH, Nar H, Priepke H, Ries U, Stassen JM, Wienen W (2002) Structure-based design of novel potent nonpeptide thrombin inhibitors. J Med Chem 45(9):1757–1766
Rawlings ND, Barrett AJ (1993) Evolutionary families of peptidases. Biochem J 290(Pt 1):205–218
Krishnaswamy S (2005) Exosite-driven substrate specificity and function in coagulation. J Thromb Haemost 3(1):54–67. doi:10.1111/j.1538-7836.2004.01021.x
Perzborn E, Strassburger J, Wilmen A, Pohlmann J, Roehrig S, Schlemmer KH, Straub A (2005) In vitro and in vivo studies of the novel antithrombotic agent BAY 59-7939—an oral, direct factor Xa inhibitor. J Thromb Haemost 3(3):514–521. doi:10.1111/j.1538-7836.2005.01166.x
Wong PC, Pinto DJ, Zhang D (2011) Preclinical discovery of apixaban, a direct and orally bioavailable factor Xa inhibitor. J Thromb Thrombolysis 31(4):478–492. doi:10.1007/s11239-011-0551-3
Wilkens M, Krishnaswamy S (2002) The contribution of factor Xa to exosite-dependent substrate recognition by prothrombinase. J Biol Chem 277(11):9366–9374. doi:10.1074/jbc.M110848200
Corte JR, Fang T, Pinto DJ, Han W, Hu Z, Jiang XJ, Li YL, Gauuan JF, Hadden M, Orton D, Rendina AR, Luettgen JM, Wong PC, He K, Morin PE, Chang CH, Cheney DL, Knabb RM, Wexler RR, Lam PY (2008) Structure–activity relationships of anthranilamide-based factor Xa inhibitors containing piperidinone and pyridinone P4 moieties. Bioorg Med Chem Lett 18(9):2845–2849. doi:10.1016/j.bmcl.2008.03.092
Furugohri T, Shiozaki Y, Muramatsu S, Honda Y, Matsumoto C, Isobe K, Sugiyama N (2005) Different antithrombotic properties of factor Xa inhibitor and thrombin inhibitor in rat thrombosis models. Eur J Pharmacol 514(1):35–42. doi:10.1016/j.ejphar.2005.03.009
Eriksson BI, Quinlan DJ, Weitz JI (2009) Comparative pharmacodynamics and pharmacokinetics of oral direct thrombin and factor Xa inhibitors in development. Clin Pharmacokinet 48(1):1–22
Mega JL, Braunwald E, Wiviott SD, Bassand JP, Bhatt DL, Bode C, Burton P, Cohen M, Cook-Bruns N, Fox KA, Goto S, Murphy SA, Plotnikov AN, Schneider D, Sun X, Verheugt FW, Gibson CM (2012) Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med 366(1):9–19. doi:10.1056/NEJMoa1112277
Wong PC, Crain EJ, Xin B, Wexler RR, Lam PY, Pinto DJ, Luettgen JM, Knabb RM (2008) Apixaban, an oral, direct and highly selective factor Xa inhibitor: in vitro, antithrombotic and antihemostatic studies. J Thromb Haemost 6(5):820–829. doi:10.1111/j.1538-7836.2008.02939.x
Pinto DJ, Orwat MJ, Koch S, Rossi KA, Alexander RS, Smallwood A, Wong PC, Rendina AR, Luettgen JM, Knabb RM, He K, Xin B, Wexler RR, Lam PY (2007) Discovery of 1-(4-methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro-1H -pyrazolo[3,4-c]pyridine-3-carboxamide (apixaban, BMS-562247), a highly potent, selective, efficacious, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem 50(22):5339–5356. doi:10.1021/jm070245n
Alexander JH, Lopes RD, James S, Kilaru R, He Y, Mohan P, Bhatt DL, Goodman S, Verheugt FW, Flather M, Huber K, Liaw D, Husted SE, Lopez-Sendon J, De Caterina R, Jansky P, Darius H, Vinereanu D, Cornel JH, Cools F, Atar D, Leiva-Pons JL, Keltai M, Ogawa H, Pais P, Parkhomenko A, Ruzyllo W, Diaz R, White H, Ruda M, Geraldes M, Lawrence J, Harrington RA, Wallentin L (2011) Apixaban with antiplatelet therapy after acute coronary syndrome. N Engl J Med 365(8):699–708. doi:10.1056/NEJMoa1105819
Connolly SJ, Eikelboom J, Joyner C, Diener HC, Hart R, Golitsyn S, Flaker G, Avezum A, Hohnloser SH, Diaz R, Talajic M, Zhu J, Pais P, Budaj A, Parkhomenko A, Jansky P, Commerford P, Tan RS, Sim KH, Lewis BS, Van Mieghem W, Lip GY, Kim JH, Lanas-Zanetti F, Gonzalez-Hermosillo A, Dans AL, Munawar M, O’Donnell M, Lawrence J, Lewis G, Afzal R, Yusuf S (2011) Apixaban in patients with atrial fibrillation. N Engl J Med 364(9):806–817. doi:10.1056/NEJMoa1007432
Ruff CT, Giugliano RP, Antman EM, Crugnale SE, Bocanegra T, Mercuri M, Hanyok J, Patel I, Shi M, Salazar D, McCabe CH, Braunwald E (2010) Evaluation of the novel factor Xa inhibitor edoxaban compared with warfarin in patients with atrial fibrillation: design and rationale for the Effective aNticoaGulation with factor xA next GEneration in Atrial Fibrillation-Thrombolysis In Myocardial Infarction study 48 (ENGAGE AF-TIMI 48). Am Heart J 160(4):635–641. doi:10.1016/j.ahj.2010.06.042
Bathala MS, Masumoto H, Oguma T, He L, Lowrie C, Mendell J (2012) Pharmacokinetics, biotransformation, and mass balance of edoxaban, a selective, direct factor Xa inhibitor, in humans. Drug Metab Dispos 40(12):2250–2255. doi:10.1124/dmd.112.046888
Zhang P, Huang W, Wang L, Bao L, Jia ZJ, Bauer SM, Goldman EA, Probst GD, Song Y, Su T, Fan J, Wu Y, Li W, Woolfrey J, Sinha U, Wong PW, Edwards ST, Arfsten AE, Clizbe LA, Kanter J, Pandey A, Park G, Hutchaleelaha A, Lambing JL, Hollenbach SJ, Scarborough RM, Zhu BY (2009) Discovery of betrixaban (PRT054021), N-(5-chloropyridin-2-yl)-2-(4-(N,N-dimethylcarbamimidoyl)benzamido)-5-methoxybenz amide, a highly potent, selective, and orally efficacious factor Xa inhibitor. Bioorg Med Chem Lett 19(8):2179–2185. doi:10.1016/j.bmcl.2009.02.111
Connolly SJ, Eikelboom J, Dorian P, Hohnloser SH, Gretler DD, Sinha U, Ezekowitz MD (2013) Betrixaban compared with warfarin in patients with atrial fibrillation: results of a phase 2, randomized, dose-ranging study (Explore-Xa). Eur Heart J 34(20):1498–1505. doi:10.1093/eurheartj/eht039
Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M (2011) Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation 124(14):1573–1579. doi:10.1161/circulationaha.111.029017
Lu G, DeGuzman FR, Hollenbach SJ, Karbarz MJ, Abe K, Lee G, Luan P, Hutchaleelaha A, Inagaki M, Conley PB, Phillips DR, Sinha U (2013) A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat Med 19(4):446–451. doi:10.1038/nm.3102
Wang L, He K, Maxwell B, Grossman SJ, Tremaine LM, Humphreys WG, Zhang D (2011) Tissue distribution and elimination of [14C]apixaban in rats. Drug Metab Dispos 39(2):256–264. doi:10.1124/dmd.110.036442
Gnoth MJ, Buetehorn U, Muenster U, Schwarz T, Sandmann S (2011) In vitro and in vivo P-glycoprotein transport characteristics of rivaroxaban. J Pharmacol Exp Ther 338(1):372–380. doi:10.1124/jpet.111.180240
Weinz C, Buetehorn U, Daehler HP, Kohlsdorfer C, Pleiss U, Sandmann S, Schlemmer KH, Schwarz T, Steinke W (2005) Pharmacokinetics of BAY 59-7939—an oral, direct Factor Xa inhibitor—in rats and dogs. Xenobiotica 35(9):891–910. doi:10.1080/00498250500250493
Hart RG, Diener HC, Yang S, Connolly SJ, Wallentin L, Reilly PA, Ezekowitz MD, Yusuf S (2012) Intracranial hemorrhage in atrial fibrillation patients during anticoagulation with warfarin or dabigatran: the RE-LY trial. Stroke 43(6):1511–1517. doi:10.1161/strokeaha.112.650614
Monroe DM, Hoffman M, Oliver JA, Roberts HR (1997) Platelet activity of high-dose factor VIIa is independent of tissue factor. Br J Haematol 99(3):542–547
Siegal DM, Cuker A (2013) Reversal of novel oral anticoagulants in patients with major bleeding. J Thromb Thrombolysis. doi:10.1007/s11239-013-0885-0
Asmis LM, Alberio L, Angelillo-Scherrer A, Korte W, Mendez A, Reber G, Seifert B, Stricker H, Tsakiris DA, Wuillemin WA (2012) Rivaroxaban: quantification by anti-FXa assay and influence on coagulation tests: a study in 9 Swiss laboratories. Thromb Res 129(4):492–498. doi:10.1016/j.thromres.2011.06.031
Becker RC, Yang H, Barrett Y, Mohan P, Wang J, Wallentin L, Alexander JH (2011) Chromogenic laboratory assays to measure the factor Xa-inhibiting properties of apixaban—an oral, direct and selective factor Xa inhibitor. J Thromb Thrombolysis 32(2):183–187. doi:10.1007/s11239-011-0591-8
Roehrig S, Straub A, Pohlmann J, Lampe T, Pernerstorfer J, Schlemmer KH, Reinemer P, Perzborn E (2005) Discovery of the novel antithrombotic agent 5-chloro-N-({(5S)-2-oxo-3-[4-(3-oxomorpholin-4-yl)phenyl]-1,3-oxazolidin-5-yl}methyl)thiophene-2-carboxamide (BAY 59-7939): an oral, direct factor Xa inhibitor. J Med Chem 48(19):5900–5908. doi:10.1021/jm050101d
Furugohri T, Isobe K, Honda Y, Kamisato-Matsumoto C, Sugiyama N, Nagahara T, Morishima Y, Shibano T (2008) DU-176b, a potent and orally active factor Xa inhibitor: in vitro and in vivo pharmacological profiles. J Thromb Haemost 6(9):1542–1549. doi:10.1111/j.1538-7836.2008.03064.x
Ogata K, Mendell-Harary J, Tachibana M, Masumoto H, Oguma T, Kojima M, Kunitada S (2010) Clinical safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel factor Xa inhibitor edoxaban in healthy volunteers. J Clin Pharmacol 50(7):743–753. doi:10.1177/0091270009351883
Turpie AG (2008) New oral anticoagulants in atrial fibrillation. Eur Heart J 29(2):155–165. doi:10.1093/eurheartj/ehm575
Acknowledgments
Dr. Steinberg was funded by NIH T-32 training Grant #5 T32 HL 7101-37. The authors wish to acknowledge Ms. Seanna Horar for her graphic design input.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steinberg, B.A., Becker, R.C. Structure–function relationships of factor Xa inhibitors: implications for the practicing clinician. J Thromb Thrombolysis 37, 234–241 (2014). https://doi.org/10.1007/s11239-013-0991-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-013-0991-z