Abstract
Background
Obesity is common in patients undergoing percutaneous coronary intervention (PCI). Argatroban, a direct thrombin inhibitor, is used during PCI in patients with or at risk of heparin-induced thrombocytopenia (HIT) and also has been evaluated in conjunction with glycoprotein IIb/IIIa inhibition in nonHIT patients. We investigated the effect of body mass index (BMI), and specifically obesity (BMI > 30 kg/m2), on argatroban therapy during PCI.
Methods
From previously reported studies of argatroban therapy during PCI in patients with or at risk of HIT (ie, HIT group) or in conjunction with glycoprotein IIb/IIIa inhibition (ie, nonHIT group), we identified patients with sufficient data to determine BMI. After an initial bolus of 350 μg/kg (HIT group) or 300 or 250 μg/kg (nonHIT group), patients received continuous argatroban 25–30 μg/kg/min (adjusted to achieve ACTs of 300–450 s, HIT group) or 15 μg/kg/min (target ACTs of 275–325 s, nonHIT group) during PCI, with additional 150 μg/kg boluses allowed if needed. Regression analyses evaluated relationships between patient BMI and ACT response to initial bolus administration, mean infusion dose (HIT group only), and rate of ACT decline after PCI. Frequencies of additional bolus usage and clinical outcomes were compared between obese and nonobese patients.
Results
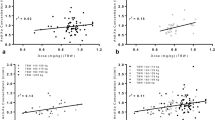
Our analysis population included 225 patients (85 obese) in total: 73 in the HIT group and 152 in the nonHIT group (300 μg/kg bolus, n = 101; 250 μg/kg bolus, n = 51), with BMIs of 16.3–50.9 kg/m2. No association was detected between BMI and the first ACT after bolus administration (median ACTs of 361, 298, and 289 s, respectively, following 350, 300, and 250 μg/kg bolus), mean infusion dose (24.2 ± 4.9 μg/kg/min overall in HIT group), or time to ACTs ≤160 s after argatroban cessation (median 4.4 h in HIT group and 1.7–2.1 h in nonHIT group). Fifteen (5 obese) patients in the HIT group and 36 (13 obese) in the nonHIT group required additional boluses, without differences by obesity versus nonobesity (P ≥ 0.35). Clinical outcomes did not differ (P ≥ 0.09) between obese and nonobese individuals: 4 (3 obese) patients in the HIT group and 4 (2 obese) in the nonHIT group had ischemic complications; 1 nonobese patient in the HIT group and 2 (1 obese) in the nonHIT group experienced major bleeding.
Conclusions
These findings support the use of actual body weight-adjusted (and ACT-targeted) argatroban therapy during PCI and suggest that dose adjustment for obesity (BMI up to 50.9 kg/m2) is unnecessary.



Similar content being viewed by others
References
Lewis BE, Matthai WH, Cohen M, Moses JW, Hursting MJ, Leya F (2002) Argatroban anticoagulation during percutaneous coronary intervention in patients with heparin-induced thrombocytopenia. Catheter Cardiovasc Interv 57:177–184
Argatroban Package Insert (2005) Research Triangle Park. GlaxoSmithKline, NC
Lewis BE, Wallis DE, Berkowitz SD et al (2001) Argatroban anticoagulant therapy in patients with heparin-induced thrombocytopenia. Circulation 103:1838–1843
Lewis BE, Wallis DE, Leya F, Hursting MJ, Kelton JG (2003) Argatroban anticoagulation in patients with heparin-induced thrombocytopenia. Arch Intern Med 163:1849–1856
Swan SK, Hursting MJ (2000) The pharmacokinetics and pharmacodynamics of argatroban: effects of age, gender, and hepatic or renal dysfunction. Pharmacotherapy 20:318–329
Cox DS, Kleiman NS, Boyle DA et al (2004) Pharmacokinetics and pharmacodynamics of argatroban in combination with a platelet glycoprotein Ilb/IIIa receptor antagonist in patients undergoing percutaneous coronary intervention. J Clin Pharmacol 44:981–990
Murray PT, Reddy BV, Grossman EJ et al (2004) A prospective study of three argatroban treatment regimens during hemodialysis in end-stage renal disease. Kidney Int 66:2446–2453
Guzzi LM, McCollum DA, Hursting MJ (2006) Effect of renal function on argatroban therapy in heparin-induced thrombocytopenia. J Thromb Thrombolysis 22:169–176
Jang IK, Lewis BE, Matthai WH, Kleiman NS (2004) Argatroban anticoagulation in conjunction with glycoprotein IIb/IIIa inhibition in patients undergoing percutaneous coronary intervention: an open-label, nonrandomized pilot study. J Thromb Thrombolysis 18:31–37
Rana JS, Mittleman MA, Ho KK, Cutlip DE (2005) Obesity and clinical restenosis after coronary stent placement. Am Heart J 150:821–826
Martin JL, Jia G, Martin SS et al (2006) The relationship of obesity to ischemic outcomes following coronary stent placement in contemporary practice. Catheter Cardiovasc Interv 67:563–570
Rice L, Hursting MJ, Baillie GM, McCollum DA (2007) Argatroban anticoagulation in obese versus non-obese patients: implications for treating heparin-induced thrombocytopenia. J Clin Pharmacol, in press
Cheymol G (2000) Effects of obesity on pharmacokinetics implications for drug therapy. Clin Pharmacokinet 39:215–231
Blouin RA, Warren GW (1999) Pharmacokinetic considerations in obesity. J Pharm Sci 88:1–7
Angulo P (2002) Nonalcoholic fatty liver disease. N Engl J Med 346:1221–1231
Rice L, Hursting MJ, McCollum DA (2006) Argatroban anticoagulation for heparin-induced thrombocytopenia in obese, versus nonobese, patients [abstract]. Blood 106(11 part 1):431a
Iqbal O, Ahmad S, Lewis BE, Walenga JM, Rangel Y, Fareed J (2002) Monitoring of argatroban in ARG310 study: potential recommendations for its use in interventional cardiology. Clin Appl Thromb Hemost 8:217–24
Shapiro NL, Durr EA, Krueger CD (2006) Prolonged anticoagulation after discontinuation of argatroban and warfarin therapy in an obese patient with heparin-induced thrombocytopenia. Pharmacotherapy 26:1806–1810
Erstad BL (2004) Dosing of medication in morbidly obese patients in the intensive care unit setting. Intensive Care Med 30:18–32
Baker MS, Skoyles RS, Shajar FM, Skinner H, Richens D, Mitchell IM (2005) Can lean body mass be used to reduce the dose of heparin and protamine for obese patients undergoing cardiopulmonary bypass? J Extra Corpor Technol 37:153–156
Lui HK (2000) Dosage, pharmacological effects and clinical outcomes for bivalirudin in percutaneous coronary intervention. J Invasive Cardiol 12(Suppl F):41F–51F
Lincoff AM, Bittl JA, Harrington RA et al (2003) Bivalirudin and provisional glycoprotein IIb/IIIa blockade compared with heparin and planned glycoprotein IIb/IIIa blockade during percutaneous coronary intervention. JAMA 289:853–863
Ellis SG, Elliott J, Horrigan M, Raymond RE, Howell G (1996) Low-normal or excessive body mass index: newly identified and powerful risk factors for death and other complications with percutaneous coronary intervention. Am J Cardiol 78:642–646
Author information
Authors and Affiliations
Corresponding author
Additional information
Dr. Hursting has received consultancy fees from GlaxoSmithKline. Dr. Jang has received research grant and honorarium from Mitsubishi Pharma Corporation, Japan.
Rights and permissions
About this article
Cite this article
Hursting, M.J., Jang, IK. Effect of body mass index on Argatroban therapy during percutaneous coronary intervention. J Thromb Thrombolysis 25, 273–279 (2008). https://doi.org/10.1007/s11239-007-0051-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-007-0051-7