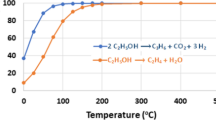
Reaction pathways of catalytic conversion of ethanol into propene are considered. It is shown that the highest propene yield is achieved over catalysts that direct the process in the following reaction pathway: ethanol → acetaldehyde → acetone → propene. A new promising propene production method that involves a two-stage process is discussed; ethanol is converted into acetone in the first stage while acetone is converted into propene in the second stage.








Similar content being viewed by others
References
X. Li, A. Kant, Y. He, et al., Catal. Today, 276, 62-77 (2016).
V. Zacharopoulou and A. A. Lemonidou, Catalysts, 8, 2-19 (2018).
R. Huang, V. Fung, Z. Wu, and D. Jiang, Catal. Today, 350, 19-24 (2020).
J. Becerra, M. Figueredo, and M. Cobo, J. Environ. Chem. Eng., 5, 1554-1564 (2017).
H. T. Abdulrazzaq and T. J. Schwartz, in: Ethanol: Science and Engineering, A. Basile, A. Iulianelli, F. Dalena, and T. Nejat Veziroglu (eds.), Elsevier (2018), pp. 1-24.
F. Dalena, A. Senatore, A. Iulianelli, et al., in: Ethanol: Science and Engineering, A. Basile, A. Iulianelli, F. Dalena, and T. Nejat Veziroglu (eds.), Elsevier (2018), pp. 25-59.
A. Kuut, K. Ritslaid, K. Kuut, et al., in: Ethanol: Science and Engineering, A. Basile, A. Iulianelli, F. Dalena, and T. Nejat Veziroglu (eds.), Elsevier (2018), pp. 61-101.
M. Kohler, in: Ethanol: Science and Engineering, A. Basile, A. Iulianelli, F. Dalena, and T. Nejat Veziroglu (eds.), Elsevier (2018), pp. 505-521.
J. Pang, M. Zheng, and T. Zhang, Adv. Catal., 64, 89-191 (2019).
J. Sun and Y. Wang, ACS Catal., 4, 1078-1090 (2014).
V. F. Tret’yakov, Y. I. Makarfi, K. V. Tret’yakov, et al., Catal. Ind., 2, 402-420 (2010).
Z. X. Song, A. Takahashi, N. Mimura, and T. Fujitani, Catal. Lett., 131, 364-369 (2009).
Y. Furumoto, Y. Harada, N. Tsunoji, et al., Appl. Catal., 399, 262-267 (2011).
D. Goto, Y. Harada, Y. Furumoto, et al., Appl. Catal. A, 383, 89-95 (2010).
A. G. Gayubo, A. M. Tarrio, A. T. Aguayo, et al., Ind. Eng. Chem. Res., 40, 3467-3474 (2001).
Y. Xue, Y. Niu, H. Zheng, et al., J. Fuel Chem. Technol., 49, 1111-1121 (2021).
T. Meng, D. S. Mao, Q. S. Guo, and G. Z. Lu, Catal. Commun., 21, 52-57 (2012).
Y. Takamitsu, K. Yamamoto, S. Yoshida, et al., J. Porous Mater., 21, 433-440 (2014).
F. F. Madeira, N. S. Gnep, P. Magnoux, et al., Appl. Catal. A, 367, 39-46 (2009).
Z. X. Song, A. Takahashi, I. Nakamura, and T. Fujitani, Appl. Catal. A., 384, 201-205 (2010).
J. Lu, Y. Liu, and N. Li, J. Nat. Gas Chem., 20, 423-427 (2011).
M. Inaba, K. Murata, I. Takahara, and K. Inoue, J. Chem. Technol. Biotechnol., 86, 95-104 (2011).
K. Inoue, K. Okabe, M. Inaba, et al., React. Kinet. Mech. Catal., 101, 477-489 (2010).
K. Ramesh, L. M. Hui, Y.-F. Han, and A. Borgna, Catal.Commun., 10, 567-571 (2009).
K. Ramesh, C. Jie, Y.-F. Han, and A. Borgna, Ind. Eng. Chem. Res., 49, 4080-4090 (2010).
A. Takahashi, W. Xia, I. Nakamura, et al., Appl. Catal. A, 423-424, 162-167 (2012).
Z. Song, W. Liu, C. Chen, et al., React. Kinet. Mech. Catal., 109, 221-231 (2013).
A. G. Gayubo, A. Alonso, B. Valle, et al., Fuel, 89, 3365-3372 (2010).
A. G. Gayubo, A. Alonso, B. Valle, et al., Chem. Eng. J., 167, 262-277 (2011).
C. Duan, X. Zhang, R. Zhou, et al., Catal. Lett., 141, 1821-1827 (2011).
C. Duan, X. Zhang, R. Zhou, et al., Fuel Process. Technol., 108, 31-40 (2013).
N. Tsunoji, Y. Furumoto, Y. Ide, et al., J. Japan Pet. Inst., 56, 22-31 (2013).
K. Inoue, K. Okabe, M. Inaba, et al., React. Kinet. Mech. Catal., 101, 227-235 (2010).
M. Iwamoto, Catal. Today, 242, 243-248 (2015).
T. Haishi, K. Kasai, and M. Iwamoto, Chem. Lett., 40, 614-616 (2011).
M. Iwamoto and Y. Kosugi, J. Phys. Chem. Lett. C, 111, 13-15 (2007).
M. Iwamoto, Catal. Surv. Asia, 12, 28-37 (2008).
A. T. Aguayo, A. G. Gayubo, A. Atutxa, et al., Ind. Eng. Chem. Res., 41, 4216-4224 (2002).
M. Inaba, K. Murata, M. Saito, and I. Takahara, Green Chem., 9, 638-646 (2007).
M. Hartmann, A. Poppl, and L. J. Kevan, Phys. Chem., 100, 9906-9910 (1996).
Y. Tanaka, N. Sawamura, and M. Iwamoto, Tetrahedron Lett., 39, 9457-9460 (1998).
M. Iwamoto, Molecules, 16, 7844-7863 (2011).
M. Iwamoto, K. Kasai, and T. Haishi, Chem. Sus. Chem., 4, 1055-1058 (2011).
K. Ikeda, Y. Kawamura, T. Yamamoto, and M. Iwamoto, Catal. Commun., 9, 106-110 (2008).
Y. Kato, Y. Nishioka, T. Wada, et al., Int. J. Modem Phys. B., 24, 3253-3256 (2010).
Y. Kato, Y. Nishioka, T. Wada, et al., 20th Symp. Chem. Eng., Korea, Abstract (Dec. 2007).
S. Sugiyama, Y. Kato, T. Wada, et al., Top. Catal., 53, 550-554 (2010).
K. Kasai, T. Haishi, and M. Iwamoto, Shokubai, 49, 126-128 (2007).
R. D. Stull, E. F. Westrum, and G. C. Sinke, The chemical thermodynamics of organic compounds, John Wiley, New York (1969).
Y. I. Pyatnytsky and P. E. Strizhak, Calculating Equilibrium and Simulating Kinetics of Heterogeneous Catalytic Reactions, https://www.free-ebooks.net/ebook/Calculating-Equilibrium-and-Simulating-Kinetics-of-Heterogeneous-Catalytic-Reactions (2018).
K. Larmier, C. Chizallet, N. Cadran, et al., ACS Catal., 5, 4423-4437 (2015).
L. M. Senchilo, L. Yu. Dolgykh, Y. I. Pyatnitsky, and P. E. Strizhak, Theor. Exp. Chem., 52, 175-183 (2016).
T. Lehmann and A. Seidel-Morgenstern, Chem. Eng. J., 19, 7214-7220 (2013).
F. Xue, C. Miao, Y. Yue, et al., Green Chem., 19, 5582-5590 (2017).
M. Iwamoto, S. Mizuno, and M. Tanaka, Chem. Eur. J., 19, 7214-7220 (2013).
M. Iwamoto, M. Tanaka, S. Hirakawa, et al., ACS Catal., 4, 3463-3469 (2014).
S. Mizuno, M. Kurosawa, M. Tanaka, and M. Iwamoto, Chem. Lett., 41, 892-894 (2012).
F. Hayashi and M. Iwamoto, ACS Catal., 3, 14-17 (2013).
F. Hayashi, M. Tanaka, D. Lin, and M. Iwamoto, J. Catal., 316, 112-120 (2014).
W. Xia, F. Wang, X. Mu, et al., Catal. Commun., 90, 10-13 (2017).
F. Wang, W. Xia, X. Mu, et al., Appl. Surf. Sci., 439, 405-412 (2018).
C. R. V. Matheus, L. H. Chagas, G. Gonzalez, et al., ACS Catal., 8, 7667-7678 (2018).
C. R. V. Matheus and E. F. S. Aguiar, Catal. Commun., 145, 106096 (2020).
A. F. F. Lima, P. C. Zonetti, C. P. Rodrigues, and L. G. Appel, Catal. Today, 279, 252-259 (2017).
Y. I. Pyatnytsky, L. Y. Dolgykh, L. M. Senchilo, et al., Theor. Exp. Chem., 55, 50-55 (2019).
Y. Pyatnytsky, L. Dolgykh, and L. Senchilo, Chem. Papers, 75, 5773-5779 (2021).
R. S. Murthy, P. Patnaik, P. Sidheswaran, and M. Jayamani, J. Catal., 109, 298-302 (1988).
T. Nakajima, T. Yamaguchi, and K. Tanabe, J. Chem. Soc. Chem. Commun., 394-395 (1987).
T. Nakajima, K. Tanabe, T. Yamaguchi, et al., Appl. Catal., 52, 237-248 (1989).
T. Nakajima, H. Nameta, S. Mishima, et al., J. Mater. Chem., 4, 853-858 (1994).
T. Nishiguchi, T. Matsumoto, H. Kanai, et al., Appl. Catal. A., 279, 273-277 (2005).
L. Y. Dolgykh, I. L. Stolyarchuk, L.A. Staraya, and Y. I. Pyatnytsky, Theor. Exp. Chem., 54, 349-357 (2018).
L. Yu. Dolgikh, Y. I. Pyatnytsky, and P. E. Strizhak, in: Bioethanol and Beyond: Advances in Production Process and Future Directions, M. Brienzo (ed.), New York, Nova Sci. (2018), Ch. 14, pp. 381-427.
J. Bussi, S. Parodi, B. Irigaray, and R. Kieffer, Appl. Catal. A., 172, 117-129 (1998).
T. M. Yurieva, Catal. Today, 51, 457-467 (1999).
J. Cunningham, G. H. Al-Sayyed, J. Cronin, et al., J. Catal., 102, 160-171 (1986).
J. Cunningham, G. H. Al-Sayyed, J. Cronin, et al., Appl. Catal., 25, 129-138 (1986).
T. Ohkubo, K. Fujiwara, T. Fujita, and M. Ishibashi, Olefin Production Process, Patent US 8552239, Publ. 2013.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Teoretychna ta Eksperymentalna Khimiya, Vol. 58, No. 1, pp. 14-24, January-February, 2022.
Rights and permissions
About this article
Cite this article
Pyatnitsky, Y.I., Senchylo, L.M., Dolgikh, L.Y. et al. Processes of Propene Production from Ethanol: Catalysts, Reaction Pathways and Thermodynamic Aspects: A Review. Theor Exp Chem 58, 15–28 (2022). https://doi.org/10.1007/s11237-022-09717-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11237-022-09717-6