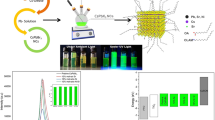
We are the first to establish that the introduction of a small amount of passivating amine (Am) during the preparation of CH3NH3PbBr3 (MAPbBr3) (MA/Am = 0.7) leads to the formation of pseudo-2D nanoparticles of this hybrid perovskite, the number of layers in which decreases going from oleylamine to hexylamine. The use of a solution of polystyrene in toluene yields more homogeneous in size stabilized MAPbBr3 nanoparticles and alters the number of layers in them. The resultant MAPbBr3 dispersions and films have strong photoluminescence (PL), whose maximum is shifted toward higher energies in comparison with a powder of this hybrid perovskite due to a quantum size effect. Dispersions and films of MAPbBr3 prepared using oleylamine have the highest PL quantum yield (about 90%) due to the formation of 2D nanoparticles with ≥ 4 layers. An advantage of the MAPbBr3 samples prepared using hexylamine is increased color purity of the PL.




Similar content being viewed by others
References
H. Wei, Y. Fang, P. Mulligan, et al., Nat. Photonics, 10, 333-339 (2016).
S. A. Veldhuis, P. P. Boix, N. Yantara, et al., Adv. Mater., 28, No. 32, 6804-6834 (2016).
Y. Zhao and K. Zhu, Chem. Soc. Rev., 45, No. 3, 655-689 (2016).
H. Sun, Z. Yang, M. Wei, et al., Adv. Mater., 28, No. 34, 1701153 (1-9) (2017).
Y. Hassan, Y. Song, R. D. Pensack, et al., Adv. Mater., 28, No. 3, 566-573 (2016).
E. Mosconi, J. M. Azpiroz, and F. De Angelis, Chem. Mater., 27, No. 13, 4885-4892 (2015).
F. Zhang, H. Zhong, C. Chen, et al., ACS Nano, 9, No. 4, 4533-4542 (2015).
B. Luo, Y.-C. Pu, S. A. Lindley, et al., Angew. Chem. Int. Ed., 55, No. 31, 8864-8868 (2016).
L. C. Schmidt, A. Pertegas, S. Gonzalez-Carrero, et al., J. Am. Chem. Soc., 136, No. 3, 850-853 (2014).
B. Luo, Y.-C. Pu, Y. Yang, et al., J. Phys. Chem. C, 119, No. 47, 26672-26682 (2015).
H. Huang, A. S. Susha, S. V. Kershaw, et al., Adv. Sci., 2, No. 9, 1500194 (2015).
L. Protesescu, S. Yakunin, M. I. Bondarchuk, et al., Nano Lett., 15, No. 6, 3692-3696 (2015).
X. Ji, X. Peng, Q. Wang, et al., Org. Electron., 52, 350-355 (2018).
V. M. Sorokin, N. V. Konoshchuk, D. M. Khmil, et al., Teor. Éksp. Khim., 55, No. 4, 201-209 (2019). [Theor. Exp. Chem., 55, No. 4, 223-231 (2019) (English translation).]
A. J. Gordon and R. A. Ford, The Chemist’s Companion, Wiley, New York (1972).
Y. Wei, X. Deng, Z. Xie, et al., Adv. Funct. Mater., 27, No. 39, 1703535 (2017).
O. Yu. Posudievsky, N. V. Konoshchuk, A. G. Shkavro, et al., ACS Appl. Nano Mater., 1, No. 8, 4145-4155 (2018).
A. M. Brouwer, Pure Appl. Chem., 83, No. 12, 2213-2228 (2011).
R. E. Kellogg and R. G. Bennett, J. Chem. Phys., 41, No. 10, 3042-3045 (1964).
A. Jana, M. Mittal, A. Singla, and S. Sapra, Chem. Commun., 53, 3046-3049 (2017).
K.-H. Wang, L.-C. Li, M. Shellaiah, and K. W. Sun, Sci. Rep., 7, 13643 (2017).
P. Tyagi, S. M. Arveson, W. A. Tisdale, et al., J. Phys. Chem. Lett., 6, No. 10, 1911-1916 (2015).
X. Kong, K. Zong, and S. S. Lee, Chem. Mater., 31, No. 14, 4953-4970 (2019).
Y. Rakita, N. Kedem, S. Gupta, et al., Cryst. Growth Des., 16, No. 10, 5717-5725 (2016).
G. C. Papavassiliou, G. A. Mousdis, and G. C. Anyfantis, Z. Naturforsch., 65, No. 4, 516-520 (2010).
J. A. Sichert, Y. Tong, N. Mutz, et al., Nano Lett., 15, No. 10, 6521-6527 (2015).
J. Cho, Y.-H. Choi, T. E. O’Loughlin, et al., Chem. Mater., 28, No. 19, 6909-6916 (2016).
V. A. Hintermayr, A. F. Richter, F. Ehrat, et al., Adv. Mater., 28, No. 43, 9478-9485 (2016).
I. Levchuk, P. Herre, M. Brandl, et al., Chem. Commun., 53, 244-247 (2007).
D. Sapori, M. Kepenekian, L. Pedesseau, et al., Nanoscale, 8, No. 12, 6369-6378 (2016).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Teoreticheskaya i Éksperimental’naya Khimiya, Vol. 55, No. 5, pp. 288-295, September-October, 2019.
Rights and permissions
About this article
Cite this article
Konoshchuk, N.V., Posudievsky, O.Y., Koshechko, V.G. et al. Spectral Characteristics of Mechanochemically Prepared Perovskite CH3NH3PbBr3 Nanoparticles Passivated by Amines with Different Alkyl Chain Length. Theor Exp Chem 55, 316–323 (2019). https://doi.org/10.1007/s11237-019-09623-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11237-019-09623-4