Abstract
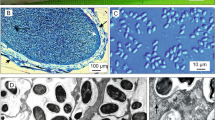
A new microsporidian species is described from farmed red sea bream Pagrus major (Temminck & Schlegel) (Teleostei: Sparidae). Large numbers of spherical whitish xenomas were observed throughout the visceral organs of the host. Histological examination showed that the microsporidia caused several xenomas that were embedded in the intestinal muscularis externa or submucosa. Light and transmission electron microscopy examination of the spores also revealed morphological features typical of species of Glugea Thélohan, 1891. This microsporidian parasite has two different types of mature spores: microspores and macrospores. The spores are elongate-ovoid, with a large posterior vacuole. The polaroplast is bi-partite, with anterior and posterior parts comprising densely packed lamellae and loose membranes, respectively, and occupies approximately the anterior half of the spore. The polar filament is anisofilar, with 12–13 coils in a single layer almost touching the posterior spore wall. Comparison of the small subunit rDNA sequences revealed 92.7–98.1% identity with the sequences available from other Glugea spp. from piscine hosts. Phylogenetic analysis demonstrated that the microsporidian species studied clustered within the Glugea clade with strong support. Based on the differences in the morphological characteristics and molecular data, the microsporidian infecting P. major is considered to represent a species new to science, Glugea pagri n. sp.




Similar content being viewed by others
References
Abdel-Ghaffar, F., Bashtar, A. R., Morsy, K., Mehlhorn, H., Al Quraishy, S., Al-Rasheid, K., & Abdel-Gaber, R. (2012). Morphological and molecular biological characterization of Pleistophora aegyptiaca sp. nov. infecting the Red Sea fish Saurida tumbil. Parasitology Research, 110, 741–752.
Brown, A. M. V., Kent, M. L., & Adamson, M. L. (2010). Description of five new Loma (Microsporidia) species in Pacific fishes with redesignation of the type species Loma morhua Morrison & Sprague, 1981, based on morphological and molecular species-boundaries tests. Journal of Eukaryotic Microbiology, 57, 529–553.
Dezfuli, B. S., Giari, L., Simoni, E., Shinn, A. P., & Bosi, G. (2004). Immunohistochemistry, histopathology and ultrastructure of Gasterosteus aculeatus tissues infected with Glugea anomala. Diseases of Aquatic Organisms, 58, 193–202.
Hall, T. A. (1999). BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series, 41, 95–98.
Hauck, A. K. (1984). A mortality and associated tissue reactions of chinook salmon, Oncorhynchus tshawytscha (Walbaum), caused by the microsporidan Loma sp. Journal of Fish Diseases, 7, 217–229.
Karpov, S. A., Mamkaeva, M. A., Aleoshin, V. V., Nassonova, E., Lilje, O., & Gleason, F. H. (2014). Morphology, phylogeny, and ecology of the aphelids (Aphelidea, Opisthokonta) and proposal for the new superphylum Opisthosporidia. Frontiers in Microbiology, 5, 112.
Kent, M. L., & Speare, D. J. (2005). Review of the sequential development of Loma salmonae (Microsporidia) based on experimental infections of rainbow trout (Oncorhynchus mykiss) and Chinook salmon (O. tshawytscha). Folia Parasitologica, 52, 63–68.
Keohane, E. M., Takvorian, P. M., Cali, A., Tanowitz, H. B., Wittner, M., & Weiss, L. M. (1996). Identification of a microsporidian polar tube protein reactive monoclonal antibody. Journal of Eukaryotic Microbiology, 43, 26–31.
Khan, R. A. (2004). Effect, distribution, and prevalence of Glugea stephani (Microspora) in winter flounder (Pleuronectes americanus) living near two pulp and paper mills in Newfoundland. Journal of Parasitology, 90, 229–233.
Larsson, J. R., & Koie, M. (2005). Ultrastructual study and description of Paramyxoides nephtys gen. n., sp n. a parasite of Nephtys caeca (Fabricius, 1780) (Polychaeta: Nephtyidae). Acta Protozoologica, 44, 175–187.
Lom, J., & Dyková, I. (2005). Microsporidian xenomas in fish seen in wider perspective. Folia Parasitologica, 52, 69–81.
Lom, J., Noga, E. J., & Dyková, I. (1995). Occurrence of a microsporean with characteristics of Glugea anomala in ornamental fish of the family Cyprinodontidae. Diseases of Aquatic Organisms, 21, 239.
Lovy, J., Kostka, M., Dykova, I., Arsenault, G., Peckova, H., Wright, G. M., & Speare, D. J. (2009). Phylogeny and morphology of Glugea hertwigi from rainbow smelt Osmerus mordax found in Prince Edward Island, Canada. Diseases of Aquatic Organisms, 86, 235–243.
Matos, E., Corral, L., & Azevedo, C. (2003). Ultrastructural details of the xenoma of Loma myrophis (phylum Microsporidia) and extrusion of the polar tube during autoinfection. Diseases of Aquatic Organisms, 54, 203–207.
Morsy, K., Abdel-Ghaffar, F., Mehlhorn, H., Bashtar, A. R., & Abdel-Gaber, R. (2012). Ultrastructure and molecular phylogenetics of a new isolate of Pleistophora pagri sp. nov. (Microsporidia, Pleistophoridae) from Pagrus pagrus in Egypt. Parasitology Research, 111, 1587–1597.
Nath, B. S., Gupta, S. K., & Bajpai, A. K. (2012). Molecular characterization and phylogenetic relationships among microsporidian isolates infecting silkworm, Bombyx mori using small subunit rRNA (SSU-rRNA) gene sequence analysis. Acta Parasitologica, 57, 342–353.
Pekcan-Hekim, Z., Rahkonen, R., & Horppila, J. (2005). Occurrence of the parasite Glugea hertwigi in young-of-the-year smelt in Lake Tuusulanjärvi. Journal of Fish Biology, 66, 583–588.
Ramsay, J. M., Watral, V., Schreck, C. B., & Kent, M. L. (2009). Pseudoloma neurophilia infections in zebrafish Danio rerio: effects of stress on survival, growth, and reproduction. Diseases of Aquatic Organisms, 88, 69–84.
Reite, O. B. (1998). Mast cells/eosinophilic granule cells of teleostean fish: A review focusing on staining properties and functional responses. Fish & Shellfish Immunology, 8, 489–513.
Rodriguez-Tovar, L. E., Speare, D. J., Markham, R. J. F., & Daley, J. (2004). Predictive modelling of post-onset xenoma growth during microsporidial gill disease (Loma salmonae) of salmonids. Journal of Comparative Pathology, 131, 330–333.
Sanders, J. L., Watral, V., Clarkson, K., & Kent, M. L. (2013). Verification of intraovum transmission of a microsporidium of vertebrates: Pseudoloma neurophilia infecting the zebrafish, Danio rerio. PLOS One, 8, e76064.
Shaw, R. W., Kent, M. L., & Adamson, M. L. (1998). Modes of transmission of Loma salmonae (Microsporidia). Diseases of Aquatic Organisms, 33, 151–156.
Sprague, V., & Hussey, K. L. (1980). Observations on Ichthyosporidium giganteum (Microsporida) with particular reference to the host-parasite relations during merogony. Journal of Eukaryotic Microbiology, 27, 169–175.
Stentiford, G. D., Feist, S. W., Stone, D. M., Bateman, K. S., & Dunn, A. M. (2013). Microsporidia: Diverse, dynamic, and emergent pathogens in aquatic systems. Trends in Parasitology, 29, 567–578.
Takvorian, P. M., & Cali, A. (1996). Polar tube formation and nucleoside diphosphatase activity in the microsporidian, Glugea stephani. Journal of Eukaryotic Microbiology, 43, 102S–103S.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, 30, 2725–2729.
Vagelli, A., Paramá, A., Sanmartín, M. L., & Leiro, J. (2005). Glugea vincentiae n. sp. (Microsporidia: Glugeidae) infecting the Australian marine fish Vincentia conspersa (Teleostei: Apogonidae). Journal of Parasitology, 91, 152–157.
Acknowledgements
This study was supported financially by Grants from the Special Scientific Research Funds for Central Non-profit Institutes, South China Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences (codes 2012TS08, 2012A0503 and 2013A0604). We are grateful to all the laboratory members for their technical advice and helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Su, Y., Feng, J., Sun, X. et al. A new species of Glugea Thélohan, 1891 in the red sea bream Pagrus major (Temminck & Schlegel) (Teleostei: Sparidae) from China. Syst Parasitol 89, 175–183 (2014). https://doi.org/10.1007/s11230-014-9519-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11230-014-9519-y