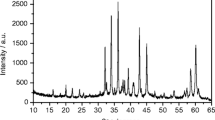
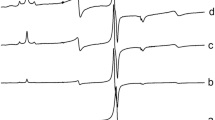
Using the EPR method, an identification of paramagnetic centers formed in TlNO3 under γ-irradiation at 77 and 300 K is performed. Their spectral characteristics, corresponding to the NO3 and NO2 radicals, are determined. The paramagnetic center NO3 is attributed to the NO3 (В) radical, which is thermally stable at 77 K. The NO3 (В) radicals exist in two forms – I and II, which differ by their spatial orientations. The accumulation of paramagnetic centers NO3 and NO2 is investigated and so are the optical reflection spectra of polycrystalline TlNO3 specimens. The final products of radiolysis of thallium nitrate are identified to be the thallium nitrite and peroxonitrite ions.
Similar content being viewed by others
References
L. G. Harrison and H. N. Ng, J. Chem. Soc., Faraday Trans., 69, No. 1, 1432–1439 (1973).
V. Anan’ev and V. Pak, Nucl. Instrum. Methods Phys. Res. Part B, 326, No. 1, 65–67 (2014).
V. A. Anan’ev, V. Pak, S. Yu. Lyrschikov, and D. V. Balykov, Izvestiya VUZov. Fiz., 56, No. 1/2, 191–193 (2013).
V. A. Nevostruev, High Energy Chem., 20, No. 5. p. 329–333 (1986).
M. C.R. Symons, D. X. West, and J. G. Wilkinson, J. Chem. Soc., Dalton Trans., No. 7, 553–556 (1975).
G. H. Fuller, J. Phys. Chem. Ref. Data, 5, No. 4, 835–1092 (1976).
A. Reuveni and Z. Luz, J. Magnetic Resonance, 23, No. 2, 271–274 (1976).
S. I. Bannov and V. A. Nevostruev, Radiat. Phys. Chem., 68, No. 5, 917–924 (2003).
R. Kissner, Adv. Inorganic Chem., 67, 335–354 (2015).
E. Frears, N. Nazhat, D. Blake, and M. E. Symons, Free Rad. Res., 27, 31–35 (1997).
F. H. Jarke and N. A. Ashford, J. Chem. Phys., 62, No. 7, 2923–2924 (1975).
D. V. Korabelnikov and Yu. N. Zhuravlev, Russ. Phys. J., 59, No. 12, 2122–2130 (2017).
D. W. Ovenall and D. H. Whiffen, Mol. Phys., 4, No. 2, 135–144 (1961).
Иванов V. I. Ivanov, A Course Book on Dosimetry [in Russian], Atomizdat, Moscow (1970).
J. Wertz and J. Bolton, Electron Paramagnetic Resonance. Elementary Theory and Practical Applications, John Weily&Sons, Inc., NY (1994).
J. Gabbay, Y. Almog, M. Davidson and A. E. Donagi, Analyst, 102, 371–376 (1977).
L. D. Kriger, M. B. Miklin, E. P. Dyagileva, and V. A. Anan’ev, Zh. Fiz. Khim., 87, No. 2, 326–330 (2013).
J. Samuel and S. Culas, Oriental J. Chem., 29, No. 4, 1535–1542 (2013).
P. W. Atkins, M. C. R. Symons, The Structure of Inorganic Radicals, Elsevier, Amsterdam (1967).
W. G. Mosley and W. G. Moulton, J. Chem. Phys., 43, No. 4, 1207–1214 (1965).
M. V. Korovkin and V. N. Salnikov, Russ. Phys. J., 59, No. 9, 1440–1445 (2017).
V. Kh. Pak, A. V. Zernyukov, M. B. Miklin, et al., Proc. 12-th Intern. Conf. “Radiation Physics of Solids”, Sebastopol, NII PMT MGIEM, Moscow (2002).
B. A. Khisamov, V. Kh. Pak, and V. A. Nevostruev, Khim. Vysok. Energ., 23, No. 2, 151–155 (1989).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 5, pp. 22–28, May, 2019.
Rights and permissions
About this article
Cite this article
Pak, V.K., Ghyngazov, S.A., Miklin, M.B. et al. Hole Localization in Thallium Nitrate (I). Russ Phys J 62, 756–762 (2019). https://doi.org/10.1007/s11182-019-01775-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11182-019-01775-z