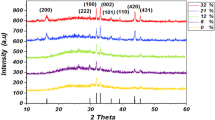
Size characteristics, structure, and spectral and luminescent properties of nanoparticles fabricated by laser ablation of zinc metal targets in water and ethanol are experimentally investigated upon excitation by Nd:YAG-laser radiation (1064 nm, 7 ns, and 15 Hz). It is demonstrated that zinc oxide nanoparticles with average sizes of 10 nm (in water) and 16 nm (in ethanol) are formed in the initial stage as a result of ablation. The kinetics of the absorption and luminescence spectra, transmission electron microscopy, and x-ray structural analysis demonstrate that during long storage of water dispersions and their drying, nanoparticles efficiently interact with carbon dioxide gas of air that leads to the formation of water-soluble Zn(CO3)2(OH)6. In ethanol, Zn oxidation leads to the formation of stable dispersions of ZnO nanoparticles with 99% of the wurtzite phase; in this case, the fluorescence spectra of ZnO nanoparticles change with time, shifting toward longer wavelength region from 550 to 620 nm, which is caused by the changed nature of defects.
Similar content being viewed by others
References
A. I. Gusev, Nanomaterials, Nanostructures, and Nanotechnologies [in Russian], Fizmatlit, Moscow (2005).
V. E. Borisenko and N. K. Tolochko, eds., Nanomaterials and Nanotechnologies [in Russian], Publishing Center of Belarussian State University, Minsk (2008).
A. V. Simakin, V. V. Voronov, and G. A. Shafeev, Tr. A. M. Prokhorov Inst. Obshch. Fiz., 60, 83–107 (2004).
R. Mahfouz, F. J. Cadete Santos Aires, A. Brenier, et al., Appl. Surf. Sci., 254, 5181–5190 (2008).
T. X. Phuoc, B. H. Howard, D. V. Martello, et al., Opt. Las. Eng., 46, 829–834 (2008).
S. I. Dolgaev, A. V. Simakin, V. V. Voronov, et al., Appl. Surf. Sci., 186, 546–551 (2002).
H. Usui, Y. Shimizu, T. Sasaki, and N. Koshizaki, J. Phys. Chem., B109, No. 1, 120–124 (2005).
B. C. Lin, P. Shen, and S. Y. Chen, J. Phys. Chem., C115, 5003–5010 (2011).
E. Chelnokov, M. Rivoal, Y. Colignon, et al., Appl. Surf. Sci., 258, No. 23, 9408–9411 (2012).
C. Zhao, Y. Huang, and J. T. Abiade, Mater. Lett., 85, 164–167 (2012).
C. Jagadish and S. Pearton, eds., Zinc Oxide Bulk, Thin Films and Nanostructures: Processing, Properties and Applications, Elsevier (2006).
A. Moezzi, A. M. McDonagh, and M. B. Cortie, Chem. Eng. J., 185–186, 1–22 (2012).
L. Schmidt-Mende and J. L. Mac Manus-Driscoll, Mater. Today, 10, No. 5, 40–48 (2007).
A. B. Djurisic, A. M. C. Ng, and X. Y. Chen, Prog. Quant. Electron., 34, 191–259 (2010).
R. Yu, Q. Lin, S.-F. Leung, and Z. Fan, Nano Energy, 1, No. 1, 57–72 (2012).
A. Wei, L. Pan, and W. Huang, Mater. Sci. Eng., B176, 1409–1421 (2011).
K. Rajeshwar, M. E. Osugi, W. Chanmanee, et al., J. Photochem. Photobiol., C9, 171–192 (2008).
S. K. Arya, S. Saha, J. E. Ramirez-Vick, et al., Anal. Chim. Acta, 737, 1–21 (2012).
C. L. Sajti, S. Giorgio, V. Khodorkovsky, and W. Marine, Appl. Phys., A89, 315–319 (2007).
H. Ma, P. L. Williams, and S. A. Diamond, Environm. Pollution, 172, 76–85 (2013).
H.-J. Egelhaaf and D. Oelkrug, J. Crystal Growth, 161, Nos. 1–4, 190–194 (1996).
J. A. Sans, A. Segura, M. Mollar, and B. Mari, Thin Solid Films, 453–454, 251–255 (2004).
X. Liu, X. Wu, H. Cao, and R. P. H. Chang, J. Appl. Phys., 95, No. 6, 3141–3147 (2004).
I. Shalish, H. Temkin, and V. Narayanamurti, Phys. Rev., B69, No. 24, 1–4 (2004).
H. Zeng, W. Cai, J. Hu, et al., Appl. Phys. Lett., 88, Paper No. 171910, 1–3 (2006).
C. He, T. Sasaki, Y. Shimizu, and N. Koshizaki, Appl. Surf. Sci., 254, 2196–2202 (2008).
I. T. Goronovskii, Yu. P. Nazarenko, and E. F. Nekryach, Brief Chemist’s Guide [in Russian], A.T. Pilipenko, ed., Naukova Dumka, Kiev (1987).
Z. Yan, R. Bao, and D. B. Chrisey, Chem. Phys. Lett., 497, 205–207 (2010).
C. He, T. Sasaki, H. Usui, et al., J. Photochem. Photobiol., A191, 66–73 (2007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 5, pp. 86–91, May, 2013.
Rights and permissions
About this article
Cite this article
Svetlichnyi, V.A., Lapin, I.N. Structure and properties of nanoparticles fabricated by laser ablation of Zn metal targets in water and ethanol. Russ Phys J 56, 581–587 (2013). https://doi.org/10.1007/s11182-013-0071-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11182-013-0071-z