Abstract
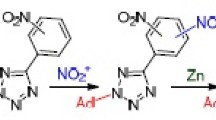
Thermolysis of N-imidoyltetrazoles generated under conditions of phase-transfer catalysis from 5-aryltetrazoles and N-(2-naphthyl)benzimidoyl chloride yields 3H-naphtho[2,1-e][1,2,4]triazepines, and acid hydrolysis of the latter leads to formation of 3-arylbenz[e]indazoles. Acid hydrolysis of 3H-naphtho[1,2-e]-[1,2,4]triazepine gives the corresponding amino ketone.
Similar content being viewed by others

REFERENCES
Artamonova, T.V., Zatsepina, M.V., and Koldobskii, G.I., Russ. J. Org. Chem., 2004, vol. 40, p. 1318.
Koldobskii, G., Ivanova, S., Nikonova, I., and Zhivich, A., Acta Chem. Scand., 1994, vol. 48, p. 596.
Koldobskii, G.I. and Ivanova, S.E., Russ. J. Org. Chem., 1995, vol. 31, p. 1435.
Morgenstern, O., Pharmazie, 2000, vol. 55, p. 871.
Artamonova, T.V., Alam, L.V., and Koldobskii, G.I., Russ. J. Org. Chem., 2000, vol. 36, p. 1700.
Nikulin, V.V., Artamonova, T.V., and Koldobskii, G.I., Russ. J. Org. Chem., 2003, vol. 39, p. 611.
Nikulin, V.V., Artamonova, T.V., and Koldobskii, G.I., Russ. J. Org. Chem., 2003, vol. 39, p. 1525.
Zakharov, M.Yu., Kochubei, V.S., and Rodin, O.G., Izv. Timiryaz. Sel’skokhoz. Akad., 2002, no. 4, p. 117; Chem. Abstr., 2004, vol. 140, no. 146 105 u.
Nikulin, V.V., Artamonova, T.V., and Koldobskii, G.I., Russ. J. Org. Chem., 2004, vol. 40, p. 1707.
Adger, B.M., Bradbury, S., Keating, M., Rees, C.W., Storr, R.C., and Williams, M.T., J. Chem. Soc., Perkin Trans. 1, 1975, p. 31.
Author information
Authors and Affiliations
Additional information
Dedicated to Full Member of the Russian Academy of Sciences V.I. Minkin on his 70th Anniversary
__________
Translated from Zhurnal Organicheskoi Khimii, Vol. 41, No. 3, 2005, pp. 451–456.
Original Russian Text Copyright © 2005 by Nikulin, Artamonova, Koldobskii.
For communication XLVII, see [1].
Rights and permissions
About this article
Cite this article
Nikulin, V.V., Artamonova, T.V. & Koldobskii, G.I. Tetrazoles: XLVIII. 3H-Naphtho[2,1-e]-and 3H-Naphtho[1,2-e][1,2,4]triazepines from 5-Aryltetrazoles. Physical and Chemical Properties. Russ J Org Chem 41, 444–449 (2005). https://doi.org/10.1007/s11178-005-0185-z
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11178-005-0185-z