Abstract
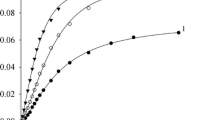
The kinetics of 3-methyl-3-chloro-1-butene solvolysis at 25°C in MeOH, EtOH, BuOH, i-BuOH, PentOH, 2-PrOH, 2-BuOH, HexOH, OctOH, t-BuOH, t-PentOH, cyclohexanol, and allyl alcohol was studied by the verdazyl method; v = k[C5H9Cl], SN1 + E1 mechanism. The reaction rate shows a satisfactory correlation with the parameter of the solvent ionizing power E T and is independent of the solvent nucleophilicity.
Similar content being viewed by others
RERERENCES
Ponomarev, N.E., Stambirskii, M.V., and Dvorko, G.F., Zh. Obshch. Khim., 2005, vol. 75, no.6, p. 937.
Dvorko, G.F., Ponomareva, E.A., and Kulik, N.I., Usp. Khim., 1984, vol. 53, no.6, p. 948.
Bentley, T.W. and Llewellyn, G., Prog. Phys. Org. Chem., 1990, vol. 17, p. 121.
Bentley, T.W., Carter, G.E., and Roberts, K., J. Org. Chem., 1984, vol. 49, no.26, p. 5183.
Dvorko, G.F., Ponomarev, N.E., and Ponomareva, E.A., Zh. Obshch. Khim., 1999, vol. 69, no.11, p. 1835.
Reichardt, Ch., Solvents and Solvent Effects in Organic Chemistry, Weinheim: VCH, 1988.
Takeuchi, K., Takasuka, M., Shiba, E., Kinoshita, T., Okazaki, T., Abboud, J.-L., Notario, R., and Castano, O., J. Am. Chem. Soc., 2000, vol. 122, no.30, p. 7351.
Kevill, D.N. and D'Souza, M.J., J. Phys. Org. Chem., 1992, vol. 5, no.3, p. 287.
Dvorko, G.F. and Ponomareva, E.A., Usp. Khim., 1991, vol. 60, no.10, p. 2089.
Dvorko, G.F., Pervishko, T.L., Golovko, N.N., Vasil'kevich, A.I., and Ponomareva, E.A., Zh. Org. Khim., 1993, vol. 29, no.9, p. 1805.
Dvorko, G.F. and Cherevach, T.V., Zh. Obshch. Khim., 1988, vol. 58, no.6, p. 1371.
Ponomarev, N.E., Stambirskii, M.V., Dvorko, G.F., and Bazil'chuk, A.V., Zh. Org. Khim., 2004, vol. 40, no.4, p. 520.
Kaatre, U., Pottel, R., and Schumacher, A., J. Phys. Chem., 1992, vol. 96, no.20, p. 6017.
Sneen, R.A., Carter, J.X., and Kay, P.S., J. Am. Chem. Soc., 1966, vol. 88, no.11, p. 2594.
Sneen, R.A. and Kay, P.S., J. Am. Chem. Soc., 1972, vol. 94, no.20, p. 6983.
Vernon, C.A., J. Chem. Soc., 1954, part 1, p. 423.
Kovtunenko, V.O., Zagal'na stereokhimiya (Principles of Stereochemistry), Kiev: Nevtes, 2001.
Dvorko, G.F., Zaliznyi, V.V., and Ponomarev, N.E., Zh. Obshch. Khim., 2002, vol. 72, no.10, p. 1644.
Dvorko, G.F., Kulik, N.I., and Ponomarev, N.E., Zh. Obshch. Khim., 1995, vol. 65, no.6, p. 1003.
Isaacs, N.S., Physical Organic Chemistry, New York: Wiley, 1992.
Palm, V.A., Osnovy kolichestvennoi teorii organicheskikh reaktsii (Principles of the Quantitative Theory of Organic Reactions), Leningrad: Khimiya, 1977.
Makitra, R.G. and Pirig, Ya.N., Zh. Obshch. Khim., 1986, vol. 56, no.3, p. 657.
Abraham, M.H., Taft, R.W., and Kamlet, M.J., J. Org. Chem., 1981, vol. 46, no.15, p. 3053.
Marcus, Y., Chem. Soc. Rev., 1993, vol. 22, no.3, p. 409.
Abboud, J.-L.M. and Notario, R., Pure Appl. Chem., 1999, vol. 71, no.4, p. 645.
Dvorko, G.F., Zaliznyi, V.V., and Ponomarev, N.E., Zh. Obshch. Khim., 2002, vol. 72, no.9, p. 1501.
Amelichev, V.A., Trunov, Yu.P., and Saidov, G.V., Vestn. Leningr. Gos. Univ., Ser. 2: Khim., 1979, no. 10, p. 83.
Vasil'kevich, A.I., Ponomareva, E.A., and Dvorko, G.F., Zh. Org. Khim., 1990, vol. 26, no.11, p. 2267.
Kuhn, R. and Trischman, H., Monatsh. Chem., 1964, vol. 95, no.2, p. 457.
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Obshchei Khimii, Vol. 75, No. 9, 2005, pp. 1503–1509.
Original Russian Text Copyright © 2005 by Ponomarev, Zaliznyi, Dvorko.
Rights and permissions
About this article
Cite this article
Ponomarev, N.E., Zaliznyi, V.V. & Dvorko, G.F. Kinetics and Mechanism of Monomolecular Heterolysis of Commercial Organohalogen Compounds: XLI. Solvent Effect on the Rate of 3-Methyl-3-chloro-1-butene Solvolysis. Correlation Analysis of Solvation Effects and Role of Solvent Nucleophilicity. Russ J Gen Chem 75, 1430–1436 (2005). https://doi.org/10.1007/s11176-005-0441-5
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0441-5