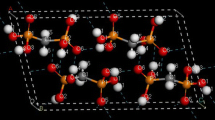
Abstract
The structures of lithium and potassium acetates were studied by the RHF/6-31G** quantum-chemical method. The metal-oxygen bonds in the dimers (CH3COOLi)2 and (CH3COOK)2 are electrostatic in nature. The bridging lithium bond is intermediate between hydrogen and ionic, and its peculiarity is determined by the deformation of molecular orbitals in the field of the lithium cation. The resulting data allow the variety of glassy smectic mesophases of lithium acetate.
Similar content being viewed by others
REFERENCES
Baranova, G.I., Bus'ko, E.A., Glebovskii, D.N., and Grigor'eva, L.I., Fiz. Khim. Stekla, 1998, vol. 24, no.5, p. 582.
Ault, B.S. and Pimentel, G.C., J. Phys. Chem., 1975, vol. 79, no.6, p. 621.
Kulkarni, G.V. and Rao, C.N.R., J. Mol. Struct., 1983, vol. 100, p. 531.
Szczesniak, M.M., Latajka, Z., Piecuch, P., Ratajczak, H., Orville-Thomas, W.J., and Rao, C.N.R., Chem. Phys., 1985, vol. 94, no.1, p. 55.
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., Elbert, S.T., Gordon, M.S., Jensen, J.J., Koseki, S., Matsunaga, N., Nguyen, K.A., Su, S., Windus, T.L., Dupius, M., and Montgomery, J.A., J. Comput. Chem., 1993, vol. 14, p. 1347.
Matsuta, S., Asada, T., and Kitaura, K., J. Electrochem. Soc., 2000, vol. 147, no.5, p. 1695.
Turi, L. and Dannenberg, J.J., J. Phys. Chem., 1993, vol. 97, no.47, p. 12197.
Huheey, J.E., Inorganic Chenistry: Principles of Structure and Reactivity, New York: Harper and Row, 1983, 3rd ed.
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Obshchei Khimii, Vol. 75, No. 9, 2005, pp. 1479–1484.
Original Russian Text Copyright © 2005 by Panteleev, Semenov, Glebovskii.
Rights and permissions
About this article
Cite this article
Panteleev, I.A., Semenov, S.G. & Glebovskii, D.N. Nature of Bridging Bonds in Lithium and Potassium Acetate Dimers. Russ J Gen Chem 75, 1406–1410 (2005). https://doi.org/10.1007/s11176-005-0438-0
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0438-0