Abstract
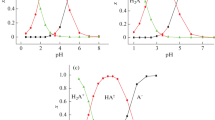
The static and complex dielectric permittivities and dc electrical conductivities of solid oxalic, malonic, and succinic acids were measured. The relaxation times of single crystal blocks (3–5 days) and mean relaxation frequencies of molecules and low-molecular-weight associates of oxalic acid dihydrate (5 MHz) and malonic acid (160 kHz) at 19°C were estimated. Aqueous oxalic acid can absorb electromagnetic energy owing to the presence of solvated electrons and holes. The electrical conductivity of this acid is protonic, and that of the anhydrous acids, electronic; the electron-hole pairs are mainly generated in these acids by lattice vibrations. The applied alternating electric field caused desorption of active gases from the acids. In some experiments with malonic acid, fast isothermal rearrangements of hydrogen bond networks and avalanche ionization of molecules were observed at a slight increase in temperature. The diagrams of charge distribution in the free acid molecules were constructed on the basis of the previous dipole moment measurements and IR data.
Similar content being viewed by others
REFERENCES
Ponomarenko, S.P., Regulyatory rosta rastenii na osnove N-oksidov proizvodnykh piridina (Plant Growth Regulators Based on N-Oxides of Pyridine Derivatives), Kiev: Tekhnika, 1999.
Borovikov, Yu.Ya. and Makovetskii, V.P., Ukr. Khim. Zh., 1991, vol. 71, no.5, p. 842.
Glasstone, S., Laidler, K.J., and Eyring, H., The Theory of Rate Processes. The Kinetics of Chemical Reactions, Viscosity, Diffusion, and Electrochemical Phenomena, New York: McGraw-Hill, 1941.
Denisov, G.S., Kul’bida, A.I., and Shraiber, V.M., in Molekulyarnaya spektroskopiya (Molecular Spectroscopy), Leningrad: Leningr. Gos. Univ., 1983, issue 5, p. 124.
Sivachek, T.E., Martynyuk, V.S., Borovikov, Yu.Ya., Makovetskii, V.P., and Reznikov, M.A., Zh. Fiz. Khim., 1994, vol. 68, no.2, p. 331.
Borovikov, Yu.Ya., Sivachek, T.E., Makovetskii, V.P., Chernilevskaya, G.I., and Novikov, V.P., Zh. Obshch. Khim., 1997, vol. 67, no.2, p. 319.
Belov, A.A. and Shakhparonov, M.I., Zh. Fiz. Khim., 1987, vol. 61, no.10, p. 2848.
Poplavko, Yu.M., Fizika dielektrikov (Physics of Dielectrics), Kiev: Vishcha Shkola, 1980.
Ponomarenko, S.P., Dul’nev, P.G., Borovikov, Yu.Ya., Sivachek, T.E., and Makovetskii, V.P., Zh. Obshch. Khim., 2001, vol. 71, no.11, p. 1788.
Gutmann, F. and Lyons, L.E., Organic Semiconductors, New York: Wiley, 1967.
Boguslavskii, L.I. and Vannikov, A.V., Organicheskie poluprovodniki i biopolimery (Organic Semiconductors and Biopolymers), Moscow: Nauka, 1968.
Gur’yanova, E.N., Gol’dshtein, I.P., and Romm, I.P., Donorno-aktseptornaya svyaz’ (Donor-Acceptor Bond), Moscow: Khimiya, 1973.
Ponomarenko, S.P., Borovikov, Yu.Ya., and Kukhar’, V.P., Zh. Obshch. Khim., 2000, vol. 70, no.11, p. 1915.
Borovikov, Yu.Ya., Dul’nev, P.G., Ponomarenko, S.P., and Sivachek, T.E., Zh. Obshch. Khim., 2001, vol. 71, no.5, p. 842.
Nicolis, G. and Prigogine, I. Self-Organization in Nonequilibrium Systems: from Dissipative Structures to Order through Fluctuations, New York: Wiley, 1977.
Borovikov, Yu.Ya., Sivachek, T.E., Makovetskaya, V.V., and Pivovarova, N.S., Zh. Obshch. Khim., 1996, vol. 66, no.1, p. 116.
Spravochnik khimika (Chemist’s Handbook), Nikol’skii, B.P., Ed., Leningrad: Khimiya, 1967.
Borovikov, Yu.Ya., Dielektrometriya v organicheskoi khimii (Dielectrometry in Organic Chemistry), Kiev: Naukova Dumka, 1987.
Raduly, S., Mraiec, M., Ciubotarik, D., Muste, A., and Simon, Z., Rev. Roum. Chim., 1982, vol. 27, no.1, p. 121.
Vilaseca, E., Perez, E., and Mata, F., Chem. Phys. Lett., 1986, vol. 132, no.3, p. 305.
Author information
Authors and Affiliations
Additional information
Translated from Zhurnal Obshchei Khimii, Vol. 74, No. 11, 2004, pp. 1828–1834.
Original Russian Text Copyright © 2004 by Ponomarenko, Borovikov, Sivachek.
Rights and permissions
About this article
Cite this article
Ponomarenko, S.P., Borovikov, Y.Y. & Sivachek, T.E. Specific features of physical chemistry and electronic structure of lower dicarboxylic acids. Russ J Gen Chem 74, 1702–1707 (2004). https://doi.org/10.1007/s11176-005-0086-4
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0086-4