Abstract
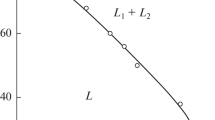
According to the study of phase equilibria in a neonol AF 9–10—neonol AF 9–12—water system made by the visual polythermal method, the immiscibility area in the system exists at temperatures above 68 °C and its formation is associated with a limited solubility of the surfactant in water at the temperatures studied. The conditions for the quantitative extraction of thallium(iii) in the form of bromide and thiocanate complexes and iron(iii) in the form of thiocyanate complexes in the neonol AF 9–10—water system at 85 °C were established. The use of a mixture of neonols in the absence of a salting agent gives no special advantages over the use of the neonol AF 9–10—water system.
Similar content being viewed by others
References
I. V. Pletnev, S. V. Smirnova, A. V. Sharov, Y. A. Zolotov, Russ. Chem. Rev., 2021, 90, 1109; DOI: https://doi.org/10.1070/RCR5007.
B. I. Petrov, A. E. Lesnov, S. A. Denisova, J. Anal. Chem., 2015, 70, 563; DOI: https://doi.org/10.7868/S0044450215040179.
A. Hamta, M. R. Dehghani, J. Mol. Liq., 2017, 231, 20; DOI: https://doi.org/10.1016/j.molliq.2017.01.084.
W. I. Mortada, Microchem. J., 2020, 157, 105055; DOI: https://doi.org/10.1016/j.microc.2020.105055.
S. S. Arya, A. M. Kaimal, M. Chib, S. K. Sonawane, P. L. Show, J. Food Sci. Technol., 2019, 56, 524; DOI: https://doi.org/10.1007/s13197-018-3546-7.
K. B. Gavazov, I. Hagarová, R. Halko, V. Andruch, J. Mol. Liq., 2019, 281, 93; DOI: https://doi.org/10.1016/j.molliq.2019.02.071.
C. B. Ojeda, F. S. Rojas, Microchim. Acta, 2012, 177, 1; DOI: https://doi.org/10.1007/s00604-011-0717-x.
V. S. Starova, S. A. Kulichenko, Izv. Vuzov. Khimiya Khim. Tekhnologiya [Bulletin of Higher Educational Institutions, Chem. Chem. Technol.], 2009, 52, No. 11, 74 (in Russian).
Yu. I. Isaeva, A. M. Elokhov, S. A. Denisova, O. S. Kudryashova, A. E. Lesnov, Russ. J. Phys. Chem., 2019, 93, 255; DOI: https://doi.org/10.1134/S004445371902016X.
S. Tagashira, T. Ichimaru, K. Nozaki, Y. Murakami, Solvent Extr. Res. Dev., Jpn, 2013, 20, 39; DOI: https://doi.org/10.15261/serdj.20.39.
H. Liang, Q. Chen, C. Xu, X. Shen, Sep. Purif. Technol., 2019, 210, 835; DOI: https://doi.org/10.1016/j.seppur.2018.08.071.
N. K. Temel, R. Gurkan, J. Anal. Chem., 2019, 74, 1174; DOI: https://doi.org/10.1134/S1061934819120128.
Y. G. Abou El-Reash, H. A. Tantawy, E. Abdel-Latif, W. I. Mortada, Microchem. J., 2020, 158, 105280; DOI: https://doi.org/10.1016/j.microc.2020.105280.
C. Dai, Y. Liu, Sh. Wang, M. Du, D. Peng, K. Wang, Zh. Yang, M. Zhao, Colloids Surf., A., 2015, 468, 322; DOI: https://doi.org/10.1016/j.colsurfa.2014.12.061.
J. Zhang, Y. Wang, Q. Peng, Korean J. Chem. Eng., 2013, 30, 1284; DOI: https://doi.org/10.1007/s11814-013-0040-9.
J. Xu, X. Li, C. Li, J. Chen, Y. Xiao, Food Chem., 2018, 242, 122; DOI: https://doi.org/10.1016/j.foodchem.2017.09.030.
Y. Sürme, A. T. Bişgin, M. Uçan, İ. Narin, J. Anal. Chem., 2018, 73, 140; DOI: https://doi.org/10.1134/S1061934818020120.
Y. Sürme, Iran J. Sci. Technol. Trans. Sci., 2022, 46, 121; DOI: https://doi.org/10.1007/s40995-021-01221-1.
V. P. Arkhipov, Z. Sh. Idiyatullin, Vestn. Tekhnol. Un-ta [Bull. Technol. Univ.], 2016, 19, No. 5, 8–11 (in Russian).
A. V. Stankova, A. M. Elokhov, S. A. Denisova, A. E. Lesnov, Russ. Chem. Bull., 2018, 67, 1608; DOI: https://doi.org/10.1007/s11172-018-2265-6.
A. V. Stankova, A. M. Elokhov, A. E. Lesnov, Russ. Chem. Bull., 2020, 69, 671; DOI: https://doi.org/10.1007/s11172-020-2816-5.
D. O. Shilykovskaya, A. M. Elokhov, Vestn. Perm. Unta. Ser. Khimiya [Bull. Perm Univ. Ser. Chem.], 2021, 11, 223; DOI: https://doi.org/10.17072/2223-1838-2021-4-223-233 (in Russian).
V. Ya. Anosov, M. I. Ozerova, Yu. Ya. Fialkov, Osnovy fi ziko-khimicheskogo analiza [Foundations of Physico-chemical Analysis], Nauka, Moscow, 1976, 504 pp. (in Russian).
G. Schwarzenbach, H. Flaschka, Die Komplexometrische Titration, Ferdinand Enke, Stuttgart, 1965, 399 pp.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no competing interests.
Additional information
No human or animal subjects were used in this research.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, Vol. 72, No. 9, pp. 2036–2040, September, 2023.
Rights and permissions
About this article
Cite this article
Shilykovskaya, D.O., Elokhov, A.M., Denisova, S.A. et al. Phase equilibria and extraction of metal ions in systems based on mixtures of oxyethylated nonylphenols. Russ Chem Bull 72, 2036–2040 (2023). https://doi.org/10.1007/s11172-023-3997-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-023-3997-5