Abstract
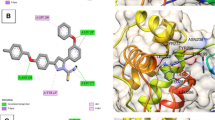
Based on the data obtained by molecular modeling of the non-covalent interaction of non-symmetric N-benzylbispidin-9-ol amides with the active site of the main protease 3CLpro of the SARS-CoV-2 virus, a series of compounds was synthesized, and their inhibitory activity against 3CLpro was studied and compared with that of the known inhibitor ML188 (IC50 = 1.56±0.55 µmol L−1). It was found that only compound 1g containing the 1,4-dihydroindeno[1,2-c]pyrazole fragment showed moderate activity (IC50 = 100±5.7µmol L−1) and was characterized by the highest calculated binding energy among the studied bispidine derivatives according to molecular docking data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
I. Tomassoli, D. Gündisch, Curr. Top. Med. Chem., 2016, 16, 1314–1342; DOI: https://doi.org/10.2174/1568026615666150915111434.
A. V. Medved’ko, A. I. Dalinger, V. N. Nuriev, V. S. Semashko, A. V. Filatov, A. A. Ezhov, A. V. Churakov, J. A. K. Howard, A. A. Shiryaev, A. E. Baranchikov, V. K. Ivanov, S. Z. Vatsadze, Nanomaterials, 2019, 9, 89–106; DOI: https://doi.org/10.3390/nano9010089.
A. I. Dalinger, A. V. Medved’ko, M. A. Kalinin, V. A. Sereda, A. V. Churakov, S. Z. Vatsadze, Russ. Chem. Bull., 2021, 70, 1002–1005; DOI: https://doi.org/10.1007/s11172-021-3180-9.
M. A. Kalinin, S. M. Antropov, A. V. Medvedko, A. O. Gudovannyy, K. A. Lyssenko, S. Z. Vatsadze, Russ. Chem. Bull., 2021, 70, 2247–2251; DOI: https://doi.org/10.1007/s11172-021-3341-x.
N. S. Zefirov, V. A. Palyulin, in Topics In Stereochemestry, vol. 20, Eds E. L. Eliel, S. H. Wilen, John Wiley & Sons, Inc., 1991, p. 171.
P. Comba, M. Kerscher, W. Schiek, Progr. Inorg. Chem., 2007, 55, 613–704; DOI: https://doi.org/10.1002/9780470144428.ch9.
A. V. Medved’ko, B. V. Egorova, A. A. Komarova, R. D. Rakhimov, D. P. Krut’ko, S. N. Kalmykov, S. Z. Vatsadze, ACS Omega, 2016, 1, 854–867; DOI: https://doi.org/10.1021/acsomega.6b00237.
K. V. Kudryavtsev, D. A. Shulga, V. I. Chupakhin, E. I. Sinauridze, F. I. Ataullakhanov, S. Z. Vatsadze, Tetrahedron, 2014, 70, 7854–7864; DOI: https://doi.org/10.1016/j.tet.2014.09.009.
S. Z. Vatsadze, D. A. Shulga, Y. D. Loginova, I. A. Vatsadze, L. Yu, H. Wang, K. V. Kudryavtsev, Mendeleev Commun., 2016, 26, 212–213; DOI: https://doi.org/10.1016/j.mencom.2016.04.011.
V. Haridas, K. S. Rajgokul, S. Sadanandan, T. Agrawal, V. Sharvani, M. V. S. Gopalakrishna, M. B. Bijesh, K. L. Kumawat, A. Basu, G. R. Medigeshi, PLoS Negl. Trop. Dis., 2013, 7, e2005; DOI: https://doi.org/10.1371/journal.pntd.0002005.
D. Shcherbakov, D. Baev, M. Kalinin, A. Dalinger, V. Chirkova, S. Belenkaya, A. Khvostov, D. Krut’ko, A. Medved’ko, E. Volosnikova, E. Sharlaeva, D. Shanshin, T. Tolstikova, O. Yarovaya, R. Maksyutov, N. Salakhutdinov, S. Vatsadze, ACS Med. Chem. Lett, 2022, 13, 140–147; DOI: https://doi.org/10.1021/acsmedchemlett.1c00299.
S. Z. Vatsadze, V. S. Tyurin, A. I. Zatsman, M. A. Manaenkova, V. S. Semashko, D. P. Krut’ko, N. V. Zyk, A. V. Churakov, L. G. Kuz’mina, Russ. J. Org. Chem., 2006, 42, 1225–1231; DOI: https://doi.org/10.1134/S1070428006080215.
S. Günther, P. Y. A. Reinke, Y. Fernández-García, J. Lieske, T. J. Lane, H. M. Ginn, F. H. M. Koua, C. Ehrt, W. Ewert, D. Oberthuer, O. Yefanov, S. Meier, K. Lorenzen, B. Krichel, J.-D. Kopicki, L. Gelisio, W. Brehm, I. Dunkel, B. Seychell, H. Giesler, B. Norton-Baker, B. Escudero-Pérez, M. Domaracky, S. Saouane, A. Tolstikova, T. A. White, A. Hänle, M. Groessler, H. Fleckenstein, F. Trost, M. Galchenkova, Y. Gevorkov, C. Li, S. Awel, A. Peck, M. Barthelmess, F. Schluenzen, P. Lourdu Xavier, N. Werner, H. Andaleeb, N. Ullah, S. Falke, V. Srinivasan, B. A. França, M. Schwinzer, H. Brognaro, C. Rogers, D. Melo, J. J. Zaitseva-Doyle, J. Knoska, G. E. Peña-Murillo, A.R. Mashhour, V. Hennicke, P. Fischer, J. Hakanpää, J. Meyer, P. Gribbon, B. Ellinger, M. Kuzikov, M. Wolf, A. R. Beccari, G. Bourenkov, D. von Stetten, G. Pompidor, I. Bento, S. Panneerselvam, I. Karpics, T. R. Schneider, M. M. Garcia-Alai, S. Niebling, C. Günther, C. Schmidt, R. Schubert, H. Han, J. Boger, D.C.F. Monteiro, L. Zhang, X. Sun, J. Pletzer-Zelgert, J. Wollenhaupt, C. G. Feiler, M. S. Weiss, E.-C. Schulz, P. Mehrabi, K. Karničar, A. Usenik, J. Loboda, H. Tidow, A. Chari, R. Hilgenfeld, C. Uetrecht, R. Cox, A. Zaliani, T. Beck, M. Rarey, S. Günther, D. Turk, W. Hinrichs, H. N. Chapman, A. R. Pearson, C. Betzel, A. Meents, Science, 2021, 372, 642–646; DOI: https://doi.org/10.1126/science.abf7945.
N. Drayman, J. K. DeMarco, K. A. Jones, S. A. Azizi, H. M. Froggatt, K. Tan, N. I. Maltseva, S. Chen, V. Nicolaescu, S. Dvorkin, K. Furlong, R. S. Kathayat, M. R. Firpo, V. Mastrodomenico, E. A. Bruce, M. M. Schmidt, R. Jedrzejczak, M. Á. Muñoz-Alía, B. Schuster, V. Nair, K. Han, A. O’Brien, A. Tomatsidou, B. Meyer, M. Vignuzzi, D. Missiakas, J. W. Botten, C. B. Brooke, H. Lee, S. C. Baker, B. C. Mounce, N. S. Heaton, W. E. Severson, K. E. Palmer, B. C. Dickinson, A. Joachimiak, G. Randall, S. Tay, Science, 2021, 373, 931–936; DOI: https://doi.org/10.1126/science.abg5827.
A. Clyde, S. Galanie, D. W. Kneller, H. Ma, Y. Babuji, B. Blaiszik, A. Brace, T. Brettin, K. Chard, R. Chard, L. Coates, I. Foster, D. Hauner, V. Kertesz, N. Kumar, H. Lee, Z. Li, A. Merzky, J. G. Schmidt, L. Tan, M. Titov, A. Trifan, M. Turilli, H. Van Dam, S. C. Chennubhotla, S. Jha, A. Kovalevsky, A. Ramanathan, M. S. Head, R. Stevens, J. Chem. Inf. Model., 2022, 62, 116–128; DOI: https://doi.org/10.1021/acs.jcim.1c00851.
G. J. Lockbaum, A. C. Reyes, J. M. Lee, R. Tilvawala, E. A. Nalivaika, A. Ali, N. Kurt Yilmaz, P. R. Thompson, C. A. Schiffer, Viruses, 2021, 13, 174; DOI: https://doi.org/10.3390/v13020174.
C. H. Zhang, E. A. Stone, M. Deshmukh, J. A. Ippolito, M. M. Ghahremanpour, J. Tirado-Rives, K. A. Spasov, S. Zhang, Y. Takeo, S. N. Kudalkar, Z. Liang, F. Isaacs, B. Lindenbach, S. J. Miller, K. S. Anderson, W. L. Jorgensen, ACS Cent. Sci., 2021, 7, 467–475; DOI: https://doi.org/10.1021/acscentsci.1c00039.
C. Ma, Y. Hu, J. A. Townsend, P. I. Lagarias, M. T. Marty, A. Kolocouris, J. Wang, ACS Pharmacol. Transl. Sci., 2020, 3, 1265–1277; DOI: https://doi.org/10.1021/acsptsci.0c00130.
E. López-López, E. Fernández-de Gortari, J. L. Medina-Franco, Drug Discov. Today, 2022, 27, 2353–2362; DOI: https://doi.org/10.1016/j.drudis.2022.05.005.
J. J. Irwin, J. Comput. Aided. Mol. Des., 2008, 22, 193–199; DOI: https://doi.org/10.1007/s10822-008-9189-4.
O. Bagheri, H. Dehghani, M. Afrooz, RSC Adv., 2015, 5, 86191–86198; DOI: https://doi.org/10.1039/C5RA15894J.
C. Yang, A. R. Teixeira, Y. Shi, S. C. Born, H. Lin, Y. Li Song, B. Martin, B. Schenkel, M. Peer Lachegurabi, K. F. Jensen, Green Chem., 2018, 20, 886–893; DOI: https://doi.org/10.1039/C7GC03469E.
E. Harder, W. Damm, J. Maple, C. Wu, M. Reboul, J. Y. Xiang, L. Wang, D. Lupyan, M. K. Dahlgren, J. L. Knight, J. W. Kaus, D. S. Cerutti, G. Krilov, W. L. Jorgensen, R. Abel, R. A. Friesner, J. Chem. Theory Comput., 2016, 12, 281–296; DOI: https://doi.org/10.1021/acs.jctc.5b00864.
R. A. Friesner, R. B. Murphy, M. P. Repasky, L. L. Frye, J. R. Greenwood, T. A. Halgren, P. C. Sanschagrin, D. T. Mainz, J. Med. Chem., 2006, 49, 6177–6196; DOI: https://doi.org/10.1021/jm051256o.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was performed under financial support of the Russian Foundation for Basic Research (Project No. 20-04-60215). Biological testing was carried out under financial support of Ministry of Science and Higher Education of the Russian Federation (Agreement No. 075-15-2021-1355 dated 12.10.2021) as part of the implementation of certain activities of the Federal Scientific and Technical Program for the Development of Synchrotron and Neutron Research and Research Infrastructure for 2019–2027.
The authors express their gratitude to the Information and Computing Center of Novosibirsk State University for the provided computing resources.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Vatsadze Sergey Zurabovich, born in 1967, Doctor of Chemical Sciences, Professor of the Russian Academy of Sciences, head of the laboratory of the N. D. Zelinsky Institute of Organic Chemistry of the Russian Academy of Sciences, candidate for corresponding member of the Russian Academy of Sciences in the elections in 2022, specialist in organic, supramolecular, and coordination chemistry, co-author of 162 scientific papers, 1 monograph and 6 patents. In his works the application of supramolecular approaches in organic chemistry, medicinal chemistry, and organic materials science have been developed, the design, synthesis, and properties of new molecules as components of functional supramolecular ensembles and materials based on them have been considered. He developed methods for the synthesis of polydentate polyheterocyclic compounds, synthesized luminescent biotags and materials for OLEDs based on substituted pyrazolecarboxylic acids, studied in detail the interactions of original compounds with the main SARS-CoV-2 viral protease using experimental and theoretical methods, revealed new promising representatives of supramolecular and coordination polymers, and developed new catalysts based on conjugates of bispidines and natural chiral terpenes. S. Z. Vatsadze teaches at the Higher Chemical College of the Russian Academy of Sciences, since 2012 he has taught courses on modern organic chemistry created by him for postgraduate students of the Division of Organic Chemistry of Moscow State University. Under his leadership, 6 candidate dissertations were prepared. S. Z. Vatsadze is executive secretary of the editorial board of the Russian Chemical Bulletin, member of dissertation councils of Peoples’ Friendship University of Russia, Lomonosov Moscow State University, D. Mendeleev University of Chemical Technology of Russia.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, Vol. 72, No. 1, pp. 239–247, January, 2023.
Rights and permissions
About this article
Cite this article
Dalinger, A.I., Baev, D.S., Yarovaya, O.I. et al. Synthesis of non-symmetric N-benzylbispidinol amides and study of their inhibitory activity against the main protease of the SARS-CoV-2 virus. Russ Chem Bull 72, 239–247 (2023). https://doi.org/10.1007/s11172-023-3729-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-023-3729-x