Abstract
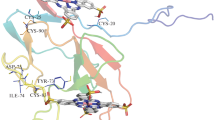
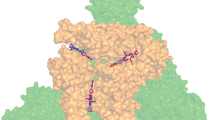
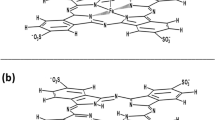
The multistage purposeful synthesis of 5,15-bis(4′-l-N-tyrosinylamidophenyl)-10,20-bis(N-methylpyridin-3′-yl)porphine diiodide was carried out, and the optimum synthesis conditions were determined. 5,15-Bis(4′-nitrophenyl)-10,20-bis(pyridin-3′-yl)porphine served as the starting porphyrin. The structure, individual character, and purity of the target compound were proved by electron spectroscopy, 1H NMR spectroscopy, mass spectrometry (MALDI TOF), and TLC. Specific features of the interaction of the synthesized porphyrin with S-protein of SARS-CoV-2 were studied using spectral and thermochemical methods, including conditions of photoirradiation. The photoirradiation of the synthesized porphyrin in a complex with the SARS-CoV-2 S-protein can result in the partial oxidation of amino acid residues of the protein and distort its primary and secondary structures. The photoirradiation of the S-protein complex with the porphyrin decreases its thermal resistance to melting by 15 °C compared to the free S-protein and causes porphyrin release.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. V. Nemukhin, B. L. Grigorenko, S. V. Lushchekina, S. D. Varfolomeev, Russ. Chem. Bull., 2021, 70, 2084; DOI: https://doi.org/10.1007/s11172-021-3319-8.
M. Takeda, Microbiol. Immunol., 2022, 66, No. 1, 15; DOI: https://doi.org/10.1111/1348-0421.12945.
N. Lebedeva, Y. Gubarev, M. Koifman, O. Koifman, Molecules, 2020, 25, 4368; DOI: https://doi.org/10.3390/molecules25194368.
N. S. Lebedeva, O. I. Koifman, Russ. J. Bioorg. Chem., 2022, 48, 1; DOI: https://doi.org/10.1134/S1068162022010071.
O. I. Nikolaeva, T. A. Ageeva, O. I. Koifman, Russ. Chem. Bull., 2021, 70, 1822; DOI: https://doi.org/10.1007/s11172-021-3288-y.
S. S. Shatokhin, V. A. Tuskaev, S. C. Gagieva, É. T. Oganesyan, Russ. Chem. Bull., 2021, 70, 1011; DOI: https://doi.org/10.1007/s11172-021-3183-6.
O. I. Koifman, T. A. Ageeva, I. P. Beletskaya, A. D. Averin, A. A. Yakushev, L. G. Tomilova, T. V. Dubinina, A. Yu. Tsivadze, Yu. G. Gorbunova, A. G. Martynov, D. V. Konarev, S. S. Khasanov, R. N. Lyubovskaya, T. N. Lomova, V. V. Korolev, E. I. Zenkevich, T. Blaudeck, Ch. von Borczyskowski, D. R. T. Zahn, A. F. Mironov, N. A. Bragina, A. V. Ezhov, K. A. Zhdanova, P. A. Stuzhin, G. L. Pakhomov, N. V. Rusakova, N. N. Semenishyn, S. S. Smola, V. I. Parfenyuk, A. S. Vashurin, S. V. Makarov, I. A. Dereven’kov, N. Zh. Mamardashvili, T. S. Kurtikyan, G. G. Martirosyan, V. A. Burmistrov, V. V. Aleksandriiskii, I. V. Novikov, D. A. Pritmov, M. A. Grin, N. V. Suvorov, A. A. Tsigankov, A. Yu. Fedorov, N. S. Kuzmina, A. V. Nyuchev, V. F. Otvagin, A. V. Kustov, D. V. Belykh, D. B. Berezin, A. B. Solovieva, P. S. Timashev, E. R. Milaeva, Yu. A. Gracheva, M. A. Dodokhova, A. V. Safronenko, D. B. Shpakovsky, S. A. Syrbu, Yu. A. Gubarev, A. N. Kiselev, M. O. Koifman, N. Sh. Lebedeva, E. S. Yurina, Macroheterocycles, 2020, 13, 4; DOI: https://doi.org/10.6060/mhc200814k.
A. Semeikin, in Uspekhi khimii porfirinov [Advances in the Chemistry of Porphyrin], Ed. O. A. Golubchikov, NII Khimii SPbGU, St. Petersburg, 1997, p. 52 (in Russian).
A. S. Semeykin, S. A. Syrbu, O. I. Koifman, in Chemical Processes with Participation of Biological and Related Compounds, Eds T. N. Lomova, G. E. Zaikov, Brill, The Netherlands, 2008, p. 45.
E. Dolušić, H. T. Ngo, W. Maes, W. Dehaen, Arkivoc, 2007, 10, 307; DOI: https://doi.org/10.3998/ark.5550190.0008.a20.
M. A. Salnikova, T. V. Lubimova, A. V. Glazunov, S. A. Syrbu, A. S. Semeikin, Macroheterocycles, 2014, 7, 249; DOI: https://doi.org/10.6060/mhc140510s.
P. Laszlo, J. Luchetti, Chem. Lett., 1993, 22, 449; DOI: https://doi.org/10.1246/cl.1993.449.
K. C. Bauer, S. Suhm, A. K. Wöll, J. Hubbuch, Int. J. Pharm., 2017, 516, 82; DOI: https://doi.org/10.1016/j.ijpharm.2016.11.009.
D. M. Byler, H. Susi, Biopolymers: Original Research on Biomolecules, 1986, 25, 469; DOI: https://doi.org/10.1002/bip.360250307.
A. Dong, B. Kendrick, L. Kreilgård, J. Matsuura, M. C. Manning, J. F. Carpenter, Arch. Biochem. Biophys., 1997, 347, 213; DOI: https://doi.org/10.1006/abbi.1997.0349.
N. S. Lebedeva, Y. A. Gubarev, E. S. Yurina, S. A. Syrbu, J. Mol. Liq., 2018, 265, 664; DOI: https://doi.org/10.1016/j.molliq.2018.06.030.
A. Ogunsipe, T. Nyokong, J. Mol. Struct., 2004, 689, 89; DOI: https://doi.org/10.1016/j.molstruc.2003.10.024.
A. B. Ormond, H. S. Freeman, Dyes Pigm., 2013, 96, 440; DOI: https://doi.org/10.1016/j.dyepig.2012.09.011.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Russian Foundation for Basic Research (Project No. 20-04-60067). Some research works was carried out using resources of the Upper Volga Regional Center of Physicochemical Research at the G. A. Krestov Institute of Solution Chemistry of the Russian Academy of Sciences and the Center for Collective Use at the Ivanovo State University of Chemistry and Technology.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Based on the materials of the V Russian Conference on Medicinal Chemistry with international participation “MedChem-Russia 2021” (May 16–19, 2022, Volgograd, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2495–2504, November, 2022.
Rights and permissions
About this article
Cite this article
Syrbu, S.A., Semeikin, A.S., Lebedeva, N.S. et al. Synthesis of water-soluble porphyrin with tyrosine fragments and study of its interaction with S-protein of SARS-CoV-2. Russ Chem Bull 71, 2495–2504 (2022). https://doi.org/10.1007/s11172-022-3679-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3679-8