Abstract
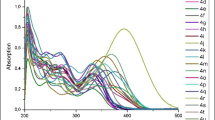
A new procedure for the synthesis of azo dyes based on 8-methoxy-2,2,4-trimethyl-1,2-dihydroquinoline and 1- or 2-substituted tetrazoles results in a crucial increase in the yields of the target compounds. The structures of the synthesized dyes containing Me, But, and Ad substituents at the 1 and 2 positions of the tetrazole moiety were established by NMR spectroscopy and X-ray diffraction analysis. The most stable conformations of the dyes depending on the substituent and the solvent were determined. The electronic absorption spectra in different solvents were characterized experimentally and theoretically.
Similar content being viewed by others
References
E. Merino, Chem. Soc. Rev., 2011, 40, 3835; DOI: https://doi.org/10.1039/C0CS00183J.
S. Crespi, N. A. Simeth, B. König, Nat. Rev. Chem., 2019, 3, 133; DOI: https://doi.org/10.1038/s41570-019-0074-6.
Z. F. Liu, K. Hashimoto, A. Fujishima, Nature, 1990, 347, 658; DOI: https://doi.org/10.1038/347658a0.
T. Ikeda, O. Tsutsumi, Science, 1995, 268, 1873; DOI: https://doi.org/10.1126/science.268.5219.1873.
J. Broichhagen, J. A. Frank, D. Trauner, Acc. Chem. Res., 2015, 48, 1947; DOI: https://doi.org/10.1021/acs._ccounts.5b00129.
M. Zhu, H. Zhou, Org. Biomol. Chem., 2018, 16, 8434; DOI: https://doi.org/10.1039/C8OB02157K.
R. J. Mart, R. K. Allemann, Chem. Commun., 2016, 52, 12262; DOI: https://doi.org/10.1039/C6CC04004G.
I. W. Willner, S. Rubin, Angew. Chem., Int. Ed., Engl., 1996, 35, 367; DOI: https://doi.org/10.1002/anie.199603671.
A. Nojiri, N. Kumagai, M. Shibasaki, Chem. Commun., 2013, 49, 4628; DOI: https://doi.org/10.1039/C3CC00008G.
G. A Selivanova, Russ. Chem. Bull., 2021, 70, 213; DOI: https://doi.org/10.1007/s11172-021-3080-z.
Y. Xu, C. Gao, J. Andréasson, M. Grøtli, Org. Lett., 2018, 20, 4875; DOI: https://doi.org/10.1021/acs.orglett.8b02014.
D. Cameron, S. Eisler, J. Phys. Org. Chem., 2018, 31, e3858; DOI: https://doi.org/10.1002/poc.3858.
A. Saylam, Z. Seferoğlu, N. Ertan, Dyes Pigments, 2008, 76, 470; DOI: https://doi.org/10.1016/j.dyepig.2006.10.005.
S. Samanta, T. M. McCormick, S. K. Schmidt, D. S. Seferos, G. A. Woolley, Chem. Commun., 2013, 49, 10314; DOI: https://doi.org/10.1039/C3CC46045B.
J. Calbo, C. E. Weston, A. J. P. White, H. S. Rzepa, J. Contreras-Garcia, M. J. Fuchter, J. Am. Chem. Soc., 2017, 139, 1261; DOI: https://doi.org/10.1021/jacs.6b11626.
N. A. Simeth, S. Crespi, M. Fagnoni, B. König, J. Am. Chem. Soc., 2018, 140, 2940; DOI: https://doi.org/10.1021/jacs.7b12871.
O. N. Lygo, E. N. Khodot, V. A. Ogurtsov, I. V. Shelaev, F. E. Gostev, T. D. Nekipelova, High Energy Chem., 2014, 48, 325; DOI: https://doi.org/10.1134/S0018143914050099.
T. D. Nekipelova, E. N. Khodot, O. N. Klimovich (Lygo), L. N. Kurkovskaya, I. I. Levina, V. A. Kuzmin, Photochem. Photobiol. Sci., 2016, 15, 1558; DOI: https://doi.org/10.1039/C6PP00251J.
I. L. Shegal, K. V. Stanovkina, N. G. Kovalenko, L. M. Shegal, Chem. Heterocycl. Compd., 1974, 10, 369; DOI: https://doi.org/10.1007/BF00472433.
H. H. Davey, R. D. Lee, T. J. Marks, J. Org. Chem., 1999, 64, 4976; DOI: https://doi.org/10.1021/jo990235x.
R. A. Henry, J. Heterocycl. Chem., 1976, 13, 391; DOI: https://doi.org/10.1002/jhet.5570130240.
A. O. Koren, P. N. Gaponik, Chem. Heterocycl. Compd., 1990, 26, 1366; DOI: https://doi.org/10.1007/BF00473965.
T. M. Klapötke, C. Miró Sabaté, A. Penger, M. Rusan, J. M. Welch, Eur. J. Inorg. Chem., 2009, 880; DOI: https://doi.org/10.1002/ejic.200800995.
A. V. Logvinov, I. N. Polyakova, E. L. Golod, Russ. J. Gen. Chem., 2009, 79, 2230; DOI: https://doi.org/10.1134/S1070363209100247.
A. V. Logvinov, I. N. Polyakova, E. L. Golod, Russ. J. Gen. Chem., 2010, 80, 2366; DOI: https://doi.org/10.1134/S1070363210110228.
P. B. Gordeev, G. A. Smirnov, Russ. Chem. Bull., 2021, 70, 1613; DOI: https://doi.org/10.1007/s11172-021-3258-4.
F. Neese, F. Wennmohs, U. Becker, C. J. Riplinger, Chem. Phys., 2020, 152, 224108; DOI: https://doi.org/10.1063/5.0004608.
A. V. Marenich, C. J. Cramer, D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378; DOI: https://doi.org/10.1021/jp810292n.
C. Adamo, V. Barone, J. Chem. Phys., 1999, 110, 6158; DOI: https://doi.org/10.1063/1.478522.
A. Schafer, C. Huber, R. Ahlrichs, J. Chem. Phys., 1994, 100, 5829; DOI: https://doi.org/10.1063/1.467146.
S. Grimme, J. Antony, S. Ehrlich, H. Krieg, J. Chem. Phys., 2010, 132, 154104; DOI: https://doi.org/10.1063/1.3382344.
F. Neese, G. Olbrich, Chem. Phys. Lett., 2002, 362, 170; DOI: https://doi.org/10.1016/S0009-2614(02)01053-9.
CrysAlisPro, Version 1.171.41.106a, Rigaku Oxford Diffraction, 2021.
G. M. Sheldrick, Acta Cryst., 2015, A71, 3; DOI: https://doi.org/10.1107/S2053273314026370.
G. M. Sheldrick, Acta Cryst., 2015, C71, 3; DOI: https://doi.org/10.1107/S2053229614024218.
A. L. Spek, J. Appl. Crystallogr., 2003, 36, 7; DOI: https://doi.org/10.1107/S0021889802022112.
C. F. Macrae, P. R. Edgington, P. McCabe, E. Pidcock, G. P. Shields, R. Taylor, Towler M., J. van de Streek, J. Appl. Crystallogr., 2006, 39, 453; DOI: https://doi.org/10.1107/S002188980600731X.
Funding
This work was performed within the framework of the state assignment of the N. M. Emanuel Institute of Biochemical Physics of the Russian Academy of Sciences (Theme No. 1201253303).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 2207–2217, October, 2022.
No human or animal subjects were used in this research.
The authors declare no competing interests. References
Rights and permissions
About this article
Cite this article
Khodot, E.N., Golovina, G.V., Timokhina, E.N. et al. New azo dyes based on 8-methoxy-2,2,4-trimethyl-1,2-dihydroquinoline and N-substituted tetrazoles. Russ Chem Bull 71, 2207–2217 (2022). https://doi.org/10.1007/s11172-022-3647-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3647-3