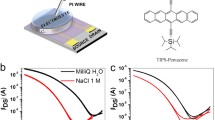
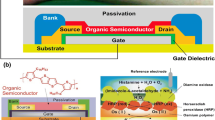
Abstract
Electrolyte-gated organic field-effect transistors (EGOFETs) provide a versatile platform for ultrasensitive, fast, and reliable detection of biological molecules in liquid media using low-cost bioelectronic sensors. The key functional layers of the EGOFETs include the semiconductor and biorecognition layers based on conjugated organic molecules, which must meet high requirements for the operational stability in various electrolytes when detecting analytes. In this work, EGOFETs based on 2,6-dioctyltetrathienoacene as the semiconductor material were fabricated by the doctor blade method compatible with printing technologies. We also report on EGOFETs with the biorecognition layer based on a biotin-containing derivative of [1]benzothieno[3,2-b]benzothiophene, which was applied by the Langmuir—Schaeffer method. The possibility of stable operation of the fabricated EGOFETs in various electrolytes and their sensor responses to the electrolyte pH value and streptavidin are demonstrated.
Similar content being viewed by others
References
L. Torsi, M. Magliulo, K. Manoli, G. Palazzo, Chem. Soc. Rev., 2013, 42, 8612; DOI: https://doi.org/10.1039/C3CS60127G.
S. Casalini, F. Leonardi, T. Cramer, F. Biscarini, Org. Electron., 2013, 14, 156; DOI: https://doi.org/10.1016/j.orgel.2012.10.027.
M. Berto, C. Diacci, R. D’Agata, M. Pinti, E. Bianchini, M. Di Lauro, S. Casalini, A. Cossarizza, M. Berggren, D. Simon, G. Spoto, F. Biscarini, C. A. Bortolotti, Adv. Biosyst., 2018, 2, 1; DOI: https://doi.org/10.1002/adbi.201700072.
M. Berto, E. Vecchi, L. Baiamonte, C. Condò, M. Sensi, M. Di Lauro, M. Sola, A. De Stradis, F. Biscarini, A. Minafra, C. A. Bortolotti, Sens. Actuators, B Chem., 2019, 281, 150; DOI: https://doi.org/10.1016/j.snb.2018.10.080.
P. Seshadri, K. Manoli, N. Schneiderhan-Marra, U. Anthes, P. Wierzchowiec, K. Bonrad, C. Di Franco, L. Torsi, Biosens. Bioelectron., 2018, 104, 113; DOI: https://doi.org/10.1016/j.bios.2017.12.041.
K. Hong, S. H. Kim, A. Mahajan, C. D. Frisbie, ACS Appl. Mater. Interfaces, 2014, 6, 18704; DOI: https://doi.org/10.1021/am504171u.
R. A. Picca, K. Manoli, E. Macchia, L. Sarcina, C. Di Franco, N. Cioffi, D. Blasi, R. Österbacka, F. Torricelli, G. Scamarcio, L. Torsi, Adv. Funct. Mater., 2019, 1904513, 1; DOI: https://doi.org/10.1002/adfm.201904513.
R. A. Picca, K. Manoli, E. Macchia, A. Tricase, C. Di Franco, G. Scamarcio, N. Cioffi, L. Torsi, Front. Chem., 2019, 7, 1; DOI: https://doi.org/10.3389/fchem.2019.00667.
C. Diacci, M. Berto, M. Di Lauro, E. Bianchini, M. Pinti, D. T. Simon, F. Biscarini, C. A. Bortolotti, Biointerphases, 2017, 12, 05F401; DOI: https://doi.org/10.1116/1.4997760.
P. A. Shaposhnik, S. A. Zapunidi, M. V. Shestakov, E. V. Agina, S. A. Ponomarenko, Russ. Chem. Rev., 2020, 89, 1483; DOI: https://doi.org/10.1070/RCR4973.
T. K. Nguyen, H. V. Tran, T. T. Vu, S. Reisberg, V. Noël, G. Mattana, M. C. Pham, B. Piro, Biosens. Bioelectron., 2019, 127, 118; DOI: https://doi.org/10.1016/j.bios.2018.12.005.
G. Palazzo, D. De Tullio, M. Magliulo, A. Mallardi, F. Intranuovo, M. Y. Mulla, P. Favia, I. Vikholm-Lundin, L. Torsi, Adv. Mater., 2015, 27, 911; DOI: https://doi.org/10.1002/adma.201403541.
O. Knopfmacher, M. L. Hammock, A. L. Appleton, G. Schwartz, J. Mei, T. Lei, J. Pei, Z. Bao, Nat. Commun., 2014, 5, 2954; DOI: https://doi.org/10.1038/ncomms3954.
T. P. Nguy, R. Hayakawa, V. Kilinc, M. Petit, J. M. Raimundo, A. Charrier, Y. Wakayama, Jpn. J. Appl. Phys., 2019, 58, SDDH02; DOI: https://doi.org/10.7567/1347-4065/ab09d2.
K. Schmoltner, J. Kofler, A. Klug, E. J. W. List-Kratochvil, Adv. Mater., 2013, 25, 6895; DOI: https://doi.org/10.1002/adma.201303281.
D. Wang, V. Noël, B. Piro, Electronics, 2016, 5, 9; DOI: https://doi.org/10.3390/electronics5010009.
H. Minemawari, T. Yamada, H. Matsui, J. Tsutsumi, S. Haas, R. Chiba, R. Kumai, T. Hasegawa, Nature, 2011, 475, 7356, 364; DOI: https://doi.org/10.1038/nature10313.
P. A. Shaposhnik, D. A. Anisimov, A. A. Trul, E. V. Agina, S. A. Ponomarenko, Dokl. Phys. Chem., 2021, 496, 20; DOI: https://doi.org/10.1134/S0012501621020019.
M. S. Skorotetcky, O. V. Borshchev, M. S. Polinskaya, E. A. Zaborin, V. P. Chekusova, E. Yu. Poimanova, D. S. Anisimov, A. A. Trul, A. V. Bakirov, E. V. Agina, S. A. Ponomarenko, J. Mater. Chem. C, 2021, 9, 10216; DOI: https://doi.org/10.1039/D1TC01469B.
Y. Yuan, J. Huang, Adv. Opt. Mater., 2016, 4, 264; DOI: https://doi.org/10.1002/adom.201500560.
Q. Zhang, F. Leonardi, S. Casalini, I. Temiño, M. Mas-Torrent, Sci Rep., 2016, 6, 39623; DOI: https://doi.org/10.1038/srep39623.
E. Yu. Poimanova, P. A. Shaposhnik, D. S. Anisimov, E. G. Zavyalova, A. A. Trul, M. S. Skorotetcky, O. V. Borshchev, D. Z. Vinnitskiy, M. S. Polinskaya, V. B. Krylov, N. E. Nifantiev, E. V. Agina, S. A. Ponomarenko, ACS Appl. Mater. Interfaces, 2022, 14, 16462; DOI: https://doi.org/10.1021/acsami.1c24109.
I. K. Yakushchenko, N. N. Pozdeeva, A. A. Terentiev, S. Ya. Gadomsky, Russ. Chem. Bull., 2022, 71, 595; DOI: https://doi.org/10.1007/s11172-022-3456-8.
A. N. Vereshchagin, K. A. Karpenko, M. P. Egorov, Russ. Chem. Bull., 2020, 69, 620; DOI: https://doi.org/10.1007/s11172-020-2808-5.
V. A. Kokorekin, V. M. Khodonov, S. V. Neverov, N. E. Grammatikova, V. A. Petrosyan, Russ. Chem. Bull., 2021, 70, 600; DOI: https://doi.org/10.1007/s11172-021-3131-5.
Funding
This work in the part of fabrication of the pH sensor was financially supported by Ministry of Higher Education and Science of Russian Federation within the framework of the State Assignment No. FFSM-2022-0001, while in the part of fabrication and investigation of the biorecognition layer of the EGOFETs it was financially supported by the Russian Science Foundation (Project No. 19-73-30028).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 2116–2122, October, 2022.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Poimanova, E.Y., Shaposhnik, P.A., Karaman, P.N. et al. Electrolyte-gated organic field-effect transistors based on 2,6-dioctyltetrathienoacene as a convenient platform for fabrication of liquid biosensors. Russ Chem Bull 71, 2116–2122 (2022). https://doi.org/10.1007/s11172-022-3635-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3635-7