Abstract
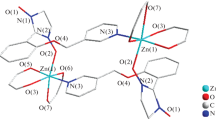
New nitronyl nitroxide radicals, 2-(4′(5′)-methylimidazol-2′-yl)- and 2-(4′(5′)-trifluoromethylimidazol-2′-yl)-4,4,5,5-tetramethyl-4,5-dihydro-1H-imidazole-1-oxyl 3-oxide, were synthesized. The molecular and crystal structures of the paramagnetic trifluoromethyl derivative were determined. A previously unknown type of hydrogen bonding of imidazol-2-yl-substituted nitronyl nitroxides to form ribbons was established. Within the ribbons, the molecules face in the same direction, with all oxygen atoms of the paramagnetic moieties being involved in H-bonding. The spectral and electrochemical properties of the radicals were compared. The first quantum chemical calculations of the electronic spectra of nitronyl nitroxide radicals taking account of the vibrational structure were performed.
Similar content being viewed by others
References
E. V. Tretyakov, V. I. Ovcharenko, Russ. Chem. Rev., 2009, 78, 971.
E. V. Tretyakov, V. I. Ovcharenko, A. O. Terentyev, I. B. Kylov, T. V. Magdesieva, D. B. Mazhukin, N. P. Gritsan, Russ. Chem. Rev., 2022, 91, RCR5025.
E. V. Tretyakov, P. A. Fedyushin, Russ. Chem. Bull., 2021, 70, 2298.
A. Caneschi, D. Gatteschi, R. Sessoli, Acc. Chem. Res., 1989, 22, 392.
H. Iwamura, K. Inoue, N. Koga, New J. Chem., 1998, 22, 201.
V. I. Ovcharenko, R. Z. Sagdeev, Russ. Chem. Rev., 1999, 68, 345.
M. T. Lemaire, Pure Appl. Chem., 2011, 83, 141.
D. Luneau, Eur. J. Inorg. Chem., 2020, 597.
A. Lannes, Y. Suffren, J. B. Tommasino, R. Chiriac, F. Toche, L. Khrouz, F. Molton, C. Duboc, I. Kieffer, J. L. Hazemann, C. Reber, A. Hauser, D. Luneau, J. Am. Chem. Soc., 2016, 138, 16493.
C. Lecourt, Y. Izumi, L. Khrouz, F. Toche, R. Chiriac, N. Bélanger-Desmarais, C. Reber, O. Fabelo, K. Inoue, C. Desroches, D. Luneau, Dalton Trans., 2020, 49, 15646.
V. Romanov, I. Bagryanskaya, N. Gritsan, D. Gorbunov, Yu. Vlasenko, M. Yusubov, E. Zaytseva, D. Luneau, E. Tretyakov, Crystals, 2019, 9, 219.
D. G. B. Boocock, E. F. Ullman, J. Am. Chem. Soc., 1968, 90, 6873.
E. F. Ullman, J. H. Osiecki, D. G. B. Boocock, R. Darcy, J. Am. Chem. Soc., 1972, 94, 7049.
G. V. Romanenko, O. V. Kuznetsova, E. V. Tretyakov, V. I. Ovcharenko, Russ. Chem. Bull., 2021, 70, 864.
C. Stroh, P. Turek, R. Ziessel, Chem. Commun., 1998, 2337.
N. Daro, J.-P. Sutter, M. Pink, O. Kahn, J. Chem. Soc., Perkin Trans. 2, 2000, 1087.
R. Ziessel, C. Stroh, H. Heise, F. H. Köhler, P. Turek, N. Claiser, M. Souhassou, C. Lecomte, J. Am. Chem. Soc., 2004, 126, 12604.
E. V. Tretyakov, S. E. Tolstikov, G. V. Romanenko, Yu. G. Shvedenkov, R. Z. Sagdeev, V. I. Ovcharenko, Russ. Chem. Bull., 2005, 54, 2169.
CN Pat. 103980256.
Y. Naoki, I. Munetoshi, M. Yuichiro, K. Takanari, I. Hidenari, O. Shigeru, Chem. Lett., 1997, 26, 251.
A. J. Bard, L. R. Faulkner, Electrochemical Methods, 2nd Ed., John Wiley and Sons, New York, 2001.
R. S. Nicholson, Anal. Chem., 1965, 37, 1351.
EP Pat. 1564213.
V. I. Ovcharenko, S. V. Fokin, G. V. Romanenko, I. V. Korobkov, P. Rey, Russ. Chem. Bull., 1999, 48, 1519.
EP Pat. 2226322.
F. Neese, Wiley Interdisciplinary Reviews: Computational Molecular Science, 2012, 2, 73.
F. Neese, Wiley Interdisciplinary Reviews: Computational Molecular Science, 2017, 8, e1327.
F. Neese, F. Wennmohs, U. Becker, C. Riplinger, J. Chem. Phys., 2020, 152, 224108.
F. Weigend, R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297.
E. Caldeweyher, C. Bannwarth, S. Grimme, J. Chem. Phys., 2017, 147, 034112.
F. Weigend, Phys. Chem. Chem. Phys., 2006, 8, 1057.
B. de Souza, G. Farias, F. Neese, R. Izsak, J. Chem. Theory Comput., 2019, 15, 1896.
B. de Souza, F. Neese, R. Izsak, J. Chem. Phys., 2018, 148, 034104.
E. van Lenthe, E. J. Baerends, J. G. Snijders, J. Chem. Phys., 1993, 99, 4597.
C. van Wullen, J. Chem. Phys., 1998, 109, 392.
C. Adamo, V. Barone, J. Chem. Phys., 1999, 110, 6158.
G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 4, pp. 722–734, April, 2022.
This study was financially supported by the Russian Science Foundation (Project No. 21-73-20079; development of an efficient method for the preparation of fluorine-containing paramagnet; production and studying of nitroxides by ESR, CV, and UV and mass spectroscopy; and quantum chemical calculations) and by the Russian Foundation for Basic Research (Project No. 19-29-08005; X-ray diffraction study).
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Fedyushin, P.A., Zayakin, I.A., Tolstikov, S.E. et al. Synthesis and redox properties of imidazol-2-yl-substituted nitronyl nitroxides. Russ Chem Bull 71, 722–734 (2022). https://doi.org/10.1007/s11172-022-3472-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3472-8