Abstract
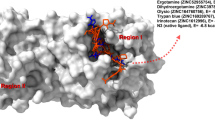
Molecular modeling tools were applied to design a potential covalent inhibitor of the main protease (Mpro) of the SARS-CoV-2 virus and to investigate its interaction with the enzyme. The compound includes a benzoisothiazolone (BZT) moiety of antimalarial drugs and a 5-fluoro-6-nitropyrimidine-2,4(1.H,3H)-dione (FNP) moiety mimicking motifs of inhibitors of other cysteine proteases. The BZT moiety provides a fair binding of the ligand on the protein surface, whereas the warhead FNP is responsible for efficient nucleophilic aromatic substitution reaction with the catalytic cysteine residue in the Mpro active site, leading to a stable covalent adduct. According to supercomputer calculations of the reaction energy profile using the quantum mechanics/molecular mechanics method, the energy of the covalent adduct is 21 kcal mol−1 below the energy of the reactants, while the highest barrier along the reaction pathway is 9 kcal mol−1. These estimates indicate that the reaction can proceed efficiently and can block the Mpro enzyme. The computed structures along the reaction path illustrate the nucleophilic aromatic substitution (SNAr) mechanism in enzymes. The results of this study are important for the choice of potential drugs blocking the development of coronavirus infection.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Ullrich, C. Nitsche, Bioorg. Med. Chem. Lett., 2020, 30, 127377.
L. Zhang, D. Lin, X. Sun, U. Curth, C. Drosten, L. Sauerhering, S. Becker, K. Rox, R. Hilgenfeld, Science, 2020, 368, 409.
D. Mondal, A. Warshel, Biochemistry, 2020, 59, 4601.
Z. Jin, Y. Zhao, Y. Sun, B. Zhang, H. Wang, Y. Wu, Y. Zhu, C. Zhu, T. Hu, X. Du, Y. Duan, J. Yu, X. Yang, X. Yang, K. Yang, X. Liu, L. W. Guddat, G. Xiao, L. Zhang, H. Yang, Z. Rao, Nat. Struct. Mol. Biol., 2020, 27, 529.
J. C. Powers, J. L Asgian, Ö. D. Ekici, K. E. James, Chem. Rev., 2002, 102, 4639.
P. Klein, P. Johe, A. Wagner, S. Jung, J. Kühlborn, F. Barthels, S. Tenzer, U. Distler, W. Waigel, B. Engels, U. A. Hellmich, T. Opatz, T. Schirmeister, Molecules, 2020, 25, 1451.
K. E. Price, C. M. Armstrong, L. S. Imlay, D. M. Hodge, C. Pidathala, N. J. Roberts, J. Park, M. Mikati, R. Sharma, A. S. Lawrenson, N. H. Tolia, N. G. Berry, P. M. O’Neill, A. R. Odom John, Sci. Rep., 2016, 6, 36777.
S. D. Varfolomeev, S. V. Lushchekina, A. V. Nemukhin, Herald Russ. Acad. Sci., 2016, 86, 185.
S. D. Varfolomeev, S. V. Lushchekina, A. V. Nemukhin, A. M. Kulakova, E. D. Kots, G. F. Makhaeva, E. Delacour, O. Lockridge, P. Masson, Russ. Chem. Bull., 2016, 65, 1592.
A. G. Taranto, P. Carvalho, M. A. Avery, J. Mol. Graph. Model., 2008, 27, 275.
K. Arafet, S. Ferrer, V. Moliner, Biochemistry, 2015, 54, 3381.
K. Arafet, S. Ferrer, V. Moliner, ACS Catal., 2017, 7, 1207.
K. Arafet, K. Świderek, V. Moliner, ACS Omega, 2018, 3, 18613.
J. R. A. Silva, L. Cianni, D. Araujo, P. H. J. Batista, D. de Vita, F. Rosini, A. Leitão, J. Lameira, C. A. Montanari, J. Chem. Inf. Model., 2020, 60, 1666.
K. Świderek, V. Moliner, Chem. Sci., 2020, 11, 10626.
A. V. Nemukhin, B. L. Grigorenko, I. V. Polyakov, S. V. Lushchekina, Supercomput. Front. Innov., 2020, 7, 25.
N. A. Senger, B. Bo, Q. Cheng, J. R. Keeffe, S. Gronert, W. Wu, J. Org. Chem., 2012, 77, 9535.
A. J. J. Lennox, Angew. Chem., Int. Ed. Engl., 2018, 57, 14686.
H. M. Berman, J. Westbrook, J. Z. Feng, G. Gilliland, T. N. Bhat, H. Weissig, I. N. Shindyalov, P. E. Bourne, Nucleic Acids Res., 2000, 28, 235.
W. Humphrey, A. Dalke, K. Schulten, J. Mol. Graph. Model., 1996, 14, 33.
J. C. Phillips, R. Braun, W. Wang, J. Gumbart, E. Tajkhorshid, E. Villa, C. Chipot, R. D. Skeel, L. Kalé, K. Schulten, J. Comput. Chem., 2005, 26, 1781.
G. M. Morris, R. Huey, W. Lindstrom, M. F. Sanner, R. K. Belew, D. S. Goodsell, A. J. Olson, Comput. Chem., 2009, 16, 2785.
M. Valiev, E. Bylaska, N. Govind, K. Kowalski, T. Straatsma, H. Van Dam, D. Wang, J. Nieplocha, E. Apra, T. Windus, W. de Jong, Comput. Phys. Commun., 2010, 181, 1477.
C. Adamo, V. Barone, J. Chem. Phys., 1999, 110, 6158.
M. T. Carroll, J. R. Cheeseman, R. Osman, H. Weinstein, J. Phys. Chem., 1989, 93, 5120.
X. Zhai, T. D. Meek, Biochemistry, 2018, 57, 3176.
F. N. Novikov, V. S. Stroylov, I. V. Svitanko, V. E. Nebolsin, Russ. Chem. Rev., 2020, 89, 858.
V. S. Stroilov, I. V. Svitanko, Mendeleev Commun., 2020, 30, 419.
Funding
The authors are grateful to M. G. Khrenova and I. V. Polyakov for help in the study and some valuable remarks. The calculations were carried out using the equipment of the shared research facilities of HPC computing resources at Lomonosov Moscow State University. The authors also acknowledge the use of supercomputer resources of the Joint Supercomputer Center of the Russian Academy of Sciences.
This study was financially supported by the Russian Science Foundation (Project No. 19-73-20032).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences O. M. Nefedov on the occasion of his 90th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2084–2089, November, 2021.
This paper does not contain descriptions of studies on animals or humans.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Nemukhin, A.V., Grigorenko, B.L., Lushchekina, S.V. et al. Supercomputer simulation of the covalent inhibition of the main protease of SARS-CoV-2. Russ Chem Bull 70, 2084–2089 (2021). https://doi.org/10.1007/s11172-021-3319-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3319-8