Abstract
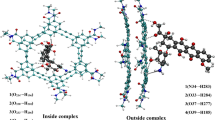
The structures of H-complexes of the anthracycline antibiotic doxorubicin with poly-N-vinylpyrrolidone segments in an aqueous medium were examined using the density functional theory and in terms of the quantum theory “Atoms in molecule” (QTAIM analysis). Quantum chemical calculations of key geometric and energy parameters of the complexes were carried out and the thermodynamic stabilities of the complexes relative to one another were compared. Based on the results obtained, possible structures of the system under study in aqueous solutions are described and the assumption is made that N-vinylpyrrolidone copolymers can be used as polymer carriers for the targeted delivery of doxorubicin.
Similar content being viewed by others
References
T. M. Fahmy, P. M. Fong, A. Goyal, W. M. Saltzman, Mater. Today, 2005, 8, 18; DOI: https://doi.org/10.1016/s1369-7021(05)71033-6.
Z. P. Aguilar, Nanomater. Med. Appt., 2013, 181; DOI: https://doi.org/10.1016/b978-0-12-385089-8.00005-4.
K. S. Yadav, D. K. Mishra, A. Deshpande, A. M. Pethe, Basic Fund. Drug Deliv., 2019, 269; DOI: https://doi.org/10.1016/b978-0-12-817909-3.00007-8.
G. M. R. Wetzels, L. H. Koole, Biomaterials, 1999, 20, 1879; DOI: https://doi.org/10.1016/S0142-9612(99)00067-8.
F. Haaf, A. Sanner, F. Straub, Polym. J., 1985, 17, 143.
C. F. Brunius, U. Edlund, A. C. Albertsson, J. Polym. Sci. A Polym. Chem., 2002, 40, 3652.
Y. Kaneda, Y. Tsutsumi, Y. Yoshioka, H. Kamada, Y. Yamamoto, H. Kodaira, S. Tsunoda, T. Okamoto, Y. Mukai, H. Shibata, S. Nakagawa, T. Mayumi, Biomaterials, 2004, 25, 3259.
R. Thirumaran, G. C. Prendergast, P. B. Gilman, Cancer Immunother., 2007, 101; DOI: https://doi.org/10.1016/b978-012372551-6/50071-7.
D. Agudelo, P. Bourassa, G. Bérubé, H. A. Tajmir-Riahi, J. Photochem. Photobiol. B: Biol., 2016, 158, 274; DOI: https://doi.org/10.1016/j.jphotobiol.2016.02.032.
S. K. Hira, A. K. Mishra, B. Ray, P. P. Manna, PLoS ONE, 2014, 9, e94309; DOI: https://doi.org/10.1371/journal.pone.0094309.
V. Kozlovskaya, F. Liu, Y. Yang, K. Ingle, S. Qian, G. V. Halade, V. S. Urban, E. Kharlampieva, Biomacromolecules, 2019, 20, 3989; DOI: https://doi.org/10.1021/acs.biomac.9b01026.
D. Habibi, S. Kaamyabi, M. M. Amini, Appl. Surf. Sci., 2014, 320, 301; DOI: https://doi.org/10.1016/j.apsusc.2014.09.092.
K. Suknuntha, V. Tantishaiyakul, V. Vao-Soongnern, Y. Espidel, T. Cosgrove, J. Polym. Sci. Part B: Polym. Phys., 2008, 46, 1258; DOI: https://doi.org/10.1002/polb.21460.
V. M. Ignat’ev, N. S. Emel’yanova, N. V. Fadeeva, S. V. Kurmaz, Russ. J. Phys. Chem. A, 2020, 94, 939; DOI: https://doi.org/10.1134/s0036024420050106.
L. Türker, J. Molec. Struct.: THEOCHEM, 2002, 583, 81; DOI: https://doi.org/10.1016/s0166-1280(01)00814-4.
S. V. Kurmaz, N. A. Obraztsova, A. A. Balakina, A. A. Terent’ev, Russ. Chem. Bull., 2016, 65, 2097; DOI: https://doi.org/10.1007/s11172-016-1558-x.
S. V. Kurmaz, N. V. Fadeeva, V. M. Ignat’ev, V. A. Kurmaz, S. A. Kurochkin, N. S. Emel’yanova, Molecules, 2020, 25, 6015; DOI: https://doi.org/10.3390/molecules25246015.
S. V. Kurmaz, N. V. Fadeeva, B. S. Fedorov, G. I. Kozub, N. S. Emel’yanova, V. A. Kurmaz, R. A. Manzhos, A. A. Balakina, A. A. Terentyev, Mendeleev Commun., 2020, 30, 22; DOI: https://doi.org/10.1016/j.mencom.2020.01.007.
S. V. Kurmaz, V. D. Sen, A. V. Kulikov, D. V. Konev, V. A. Kurmaz, A. A. Balakina, A. A. Terent’ev, Russ. Chem. Bull., 2019, 68, 1769; DOI: https://doi.org/10.1007/s11172-019-2623-z.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, D. J. Fox, Gaussian 09, Revision E.01, Gaussian, Inc., Wallingford CT, 2009.
J. M. Tao, J. P. Perdew, V. N. Staroverov, G. E. Scuseria, Phys. Rev. Lett., 2003, 91, 146401.
S. V. Kurmaz, N. V. Fadeeva, V. M. Ignat’ev, V. A. Kurmaz, S. A. Kurochkin, N. S. Emel’yanova, Molecules, 2020, 25, 6015; DOI: https://doi.org/10.3390/molecules25246015.
T. A. Keith, T. K. Gristmill, AIMAll (version 10.05.04), Software, Overland Park KS, USA, 2010; aim.tkgristmill.com.
E. Espinosa, E. Molins, C. Lecomte, Chem. Phys. Lett., 1998, 285, 170.
Chemcraft — graphical software for visualization of quantum chemistry computations;https://www.chemcraftprog.com.
Y. Maréchal, The Hydrogen Bond and the Water Molecule, Elsevier Science, 2007, p. 3.
V. P. Gupta, Principles and Applications of Quantum Chemistry, Academic Press, 2016, p. 359; DOI:https://doi.org/10.1016/b978-0-12-803478-1.00011-x.
I. S. Bushmarinov, K. A. Lyssenko, M. Y. Antipin, Russ. Chem. Rev., 2009, 78, 283.
A. Karpfen, J. Ladik, P. Russegger, P. Schuster, S. Suhai, Theor. Chim. Acta, 1974, 34, 115.
N. Kobko, J. J. Dannenberg, J. Phys. Chem. A, 2003, 107, 10389.
C.-M. Saak, Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology, Acta Universitatis Upsaliensis, Uppsala, 2019, 90 pp.
A. K. Sopper, Chem. Phys., 2000, 258, 121.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1269–1274, July, 2021.
This work was carried out as part of the State Assignment (Theme No. AAAA-A19-119071890015-6).
This paper does not contain descriptions of studies on animals or humans.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Ignat’ev, V.M., Emel’yanova, N.S., Kurmaz, S.V. et al. Hydrogen bonds formed upon encapsulation of doxorubicin into amphiphilic N-vinylpyrrolidone copolymer: a quantum chemical study. Russ Chem Bull 70, 1269–1274 (2021). https://doi.org/10.1007/s11172-021-3210-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3210-7