Abstract
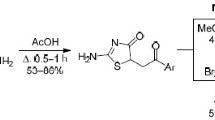
Regiospecific S-monoaminoalkylation of 5-substituted derivatives of 6-hydroxy-2-thiouracil with free N,N-dialkyl-N-(2-chloroethyl)amines in anhydrous PriOH was described for the first time. In compliance with the rules and regulations of green chemistry, this approach was used to synthesize a number of structural analogs of isothiobarbamine in high yield and purity, which are potential synthetic actoprotectors of immediate action.
Similar content being viewed by others
References
I. A. Novakov, D. S. Sheikin, V. V. Chapurkin, M. B. Nawrozkij, A. S. Babushkin, E. A. Ruchko, A. Yu. Maryshev, D. Schols, Russ. J. Gen. Chem., 2020, 90, 352; DOI: https://doi.org/10.1134/S1070363220030056.
I. A. Novakov, D. S. Sheikin, V. V. Chapurkin, M. B. Nawrozkij, A. S. Babushkin, Ya. P. Kuznetsov, E. A. Ruchko, V. V. Kachala, A. Yu. Maryshev, D. Schols, Chem. Heterocycl. Compd., 2020, 56, 67; DOI: https://doi.org/10.1007/s10593-020-02624-5.
I. A. Novakov, D. S. Sheikin, V. V. Chapurkin, M. B. Nawrozkii, I. A. Kirillov, E. A. Ruchko, Russ. Chem. Bull., 2020, 69, 2363.
I. A. Novakov, L. L. Brunilina, V. V. Chapurkin, M. B. Nawrozkij, D. S. Sheikin, Ya. P. Kuznetsov, E.A. Ruchko, D. Schols, AIP Conf. Proc., 2020, 2280, 030011–1; DOI: https://doi.org/10.1063/5.0018050.
Pat. RF 552789 (in Russian).
S. G. Soboleva, I. F. Gerasimenko, L. G. Kravchuk, L. I. Elfimova, A. A. Dvorkin, Yu. A. Simonov, S. A. Andronati, Dokl. Akad. Nauk [Dokl. Chem.], 1992, 327, 349 (in Russian).
Crown Compounds. Their Characteristics and Applications, Ed. M. Hiraoka, Tokyo, 1982, 275 pp.
R. K. Henderson, C. Jimenez-Gonzalez, D. J. C. Constable, S. R. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binksa, A. D. Curzons, Green Chem., 2011, 13, 854; DOI:https://doi.org/10.1039/c0gc00918k.
C. Welch, T. Nowak, L. A. Joyce, E. L. Regalado, ACS Sustain. Chem. Eng., 2015, 3, 1000; DOI: https://doi.org/10.1021/acssuschemeng.5b00133.
H. Yang, F. C. Thyrion, Bull. Soc. Chim. Belg., 1996, 105, 23; DOI: https://doi.org/10.1002/bscb.19961050106.
M. D. Mashkovsky, Lekarstvennyye sredstva [Medicines], V. 1, Meditsina, Moscow, 1994, 544 pp. (in Russian).
M. D. Mashkovsky, Lekarstvennyye sredstva [Medicines], Meditsina, Moscow, 1964, 240 pp. (in Russian).
J. Giovanelli, S. H. Mudd, A. H. Datko, in The Biochemistry of Plants, Ed. B. J. Miflin, Acad. Press., London, 1980, Vol. 5, p. 453; DOI: https://doi.org/10.1016/b978-0-12-675405-6.50018-8.
K. Abe, K. Matsui, J. Pharm. Soc. Jpn., 1955, 75, 896; DOI: https://doi.org/10.1248/yakushi1947.75.7_896.
L. F. Tietze, T. Eicher, Reaktionen und Synthesen im organisch-chemischen Praktikum und Forschungslaboratorium, Stuttgart, 1991.
R. S. Tipson, J. Org. Chem., 1944, 9, 235; DOI: https://doi.org/10.1021/jo01185a005.
M. Brown, J. Am. Chem. Soc., 1955, 77, 3607; DOI: https://doi.org/10.1021/ja01618a054.
W. R. Kirner, J. Am. Chem. Soc., 1928, 50, 2452.
T. Koizumi, T. Fuchigami, Z. E. Kandeel, N. Sato, T. Nonaka, Bull. Chem. Soc. Jpn., 1986, 59, 757; DOI: https://doi.org/10.1246/bcsj.59.757.
I. Ishida, Nippon Kagaku Kaishi, 1943, 64, 165.
X. Zhang, D. Li, X. Xue, Y. Zhang, J. Zhang, C. Huang, Z. Guo, N. Tadesse, Nat. Prod. Res., 2018, 32, 128; DOI: https://doi.org/10.1080/14786419.2017.1340283.
Pat. US 1739662.
W. J. Bailey, J. J. Daly, J. Org. Chem., 1964, 29, 1249; DOI:https://doi.org/10.1021/jo01028a503.
M. Windus, J. Am. Chem. Soc., 1930, 52, 2577.
J. Lee, J. Am. Chem. Soc., 1938, 60, 993; DOI:https://doi.org/10.1021/ja01272a003.
K. Abe, T. Iskisaka, Y. Tsukamoto, J. Pharm. Soc. Jpn., 1955, 75, 891; DOI:https://doi.org/10.1248/yakushi1947.75.7_891.
Pat. RF 2744470; Byul. Izobret. [Invention Bull.], 2021, No. 7 (in Russian).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences V. N. Charushin on the occasion of his 70th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 5, pp. 943–948, May, 2021.
This work was financially supported by the Russian Science Foundation (Project No. 19-13-00123), using the park of analytical instruments of the Center for Collective Use (CCU) of the N. D. Zelinsky Institute of Organic Chemistry of the Russian Academy of Sciences and N. N. Vorozhtsov Novosibirsk Institute of Organic Chemistry of the Siberian Branch of the RAS.
This work does not involve human participants and animal subjects.
The authors declare no competing interest.
Rights and permissions
About this article
Cite this article
Novakov, I.A., Sheikin, D.S., Chapurkin, V.V. et al. Regiospecific S-aminoalkylation of 5-substituted 6-hydroxy-2-thiouracil derivatives in the synthesis of structural analogs of isothiobarbamine. Russ Chem Bull 70, 943–948 (2021). https://doi.org/10.1007/s11172-021-3171-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3171-x