Abstract
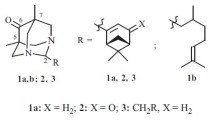
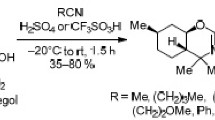
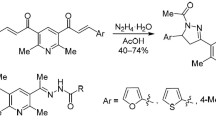
Conjugates of terpenoids with 1,3,5-trisubstituted pyrazoles were synthesized by the cross-coupling of methyl 16-(2-chloro-2-oxoacetyl)labdatrienoate with terminal arylacetylenes via the Castro—Stephens reaction and heterocyclization of arylalkyne-1,2-diones with arylhydr-azines. The structure of one reaction product was established by X-ray diffraction. The conditions for the formation of furanolabdanoid arylalkyne-1,2-diones were found. The newly synthesized pyrazoles exhibit analgesic activity in a model of chemical irritation.
Similar content being viewed by others
References
E. E. Shul´ts, M. E. Mironov, Yu. V. Kharitonov, Chem. Nat. Compd., 2014, 50, 2.
T. G. Tolstikova, I. V. Sorokina, T. V. Voevoda, E. E. Shul´ts, G. A. Tolstikov, Dokl. AN, 2001, 376, 271.
H.-S. Chae, Y.-W. Chin, Immunopharm. & Immunotoxicol., 2012, 34, 250.
M.-S. Lee, S.-O. Lee, S.-H. Kim, E.-O. Lee, H.-J. Lee, Int. J. Mol. Sci., 2016, 17, 1066.
J. H. Lee, H.-J. Lee, D. Y. Sim, J. H. Jung, K. R. Kim, S.-H. Kim, Phytother. Res., 2018, 32, 1755.
D. S. Ahn, H. J. Lee, J. Hwang, H. Han, B. Kim, B. Shim, S.-H. Kim, Int. J. Mol. Sci., 2018, 19, 1476.
E. E. Shults, J. Velder, H.-G. Schmalz, S. V. Chernov, T. V. Rubalova, Y. V. Gatilov, G. Henze, G. A. Tolstikov, A. Prokop, Bioorg. Med. Chem. Lett., 2006, 16, 4228.
M. E. Mironov, Y. V. Kharitonov, E. E. Shul´ts, M. M. Shakirov, I. Yu. Bagryanskaya, G. A. Tolstikov, Chem. Nat. Compd., 2010, 46, 233.
E. A. Morozova, T. G. Tolstikova, E. E. Shults, S. V. Chernov, Y. V. Kharitonov, M. E. Mironov, G. A. Tolstikov, Chem. Sustainable Develop., 2010, 18, 409.
M. E. Mironov, M. A. Pokrovsky, Y. V. Kharitonov, M. M. Shakirov, A. G. Pokrovsky, E. E. Shults, ChemistrySelect, 2016, 1, 417.
O. I. Kremenko, Yu. V. Kharitonov, E. E. Shul´ts, Russ. J. Org. Chem., 2017, 53, 35.
J. V. Faria, P. F. Vegi, A. C. C. Miguita, M. C. dos Santos, N. Boechat, A. M. R. Bernardino, Bioorg. Med. Chem., 2017, 25, 5891.
H. H. Wasserman, C. A. Blum, Tetrahedron Lett., 1994, 35, 9787.
E. Merkul, J. Dohe, C. Gers, F. Rominger, T. J. J. Müller, Angew. Chem., Int. Ed., 2011, 50, 2966.
C. Boersch, E. Merkul, T. J. J. Müller, Angew. Chem., Int. Ed., 2011, 50, 10448.
C. F. Gers, J. Nordmann, C. Kumru, W. Frank, T. J. J. Müller, J. Org. Chem., 2014, 79, 3296.
Z. Zhang, X. Jiang, Org. Lett., 2014, 16, 4400.
A. K. Mukherjee, P. Margaretha, W. C. Agosta, J. Org. Chem., 1996, 61, 3388.
Y. Liu, M. Liu, S. Guo, H. Tu, Y. Zhou, H. Gao, Org. Lett., 2006, 8, 3445.
Z. Zhang, Z. Dai, X. Jiang, Asian J. Org. Chem., 2016, 5, 52.
X. Kong, G. Zhang, S. Yang, X. Liu, X. Fang, Adv. Synth. Catal., 2017, 359, 2729.
I. A. Ibarra, A. Islas-Jácome, E. González-Zamora, Org. Biomol. Chem., 2018, 16, 1402.
C. G. Neochoritis, T. Zhao, A. Domling, Chem. Rev., 2019, 119, 1970.
F. S. Al-Hajjar, S. S. Sabri, J. Heterocycl. Chem., 1986, 23, 727.
B. S. Holla, K. V. Udupa, K. P. Sridhar, Bull. Chem. Soc. Jpn, 1989, 62, 3409.
J. P. Waldo, S. Mehta, R. C. Larock, J. Org. Chem., 2008, 73, 6666.
M. Zora, A. Kivrak, C. Yazici, J. Org. Chem., 2011, 76, 6726.
V. A. Savel´ev, A. A. Kotova, T. V. Rybalova, E. E. Shults, Chem. Heterocycl. Compd., 2019, 55, 943.
F. H. Allen, O. Kenard, D. G. Watson, L. Bramer, A. G. Orpen, R. Taylor, J. Chem. Soc., Perkin Trans. 2, 1987, 1.
Cambridge Structural Database, Version 5.39, University of Cambridge, UK.
F. H. Allen, Acta Crystallogr., Sect. B, 2002, 58, 380.
R. S. Rowland, R. Taylor, J. Phys. Chem., 1996, 100, 7384.
G. M. Sheldrick, SADABS, Program for Area Detector Adsorption Correction, Institute for Inorganic Chemistry, University of Goettingen (Germany), 1996.
G. M. Sheldrick, SHELX-97, Programs for Crystal Structure Analysis, Release 97-2, University of Göttingen, Göttingen (Germany).
R. Koster, M. Anderson, E. J. De Beer, Fed. Proc. 1959, 18, 412.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences M. S. Yunusov on the occasion of his 80th birthday.
We would like to acknowledge the staff of the Multi-Access Chemical Service Center of the Siberian Branch of the Russian Academy of Sciences for spectral and analytical measurements.
This study was financially supported by the Russian Science Foundation (Project Nos 17-73-10274 and 18-13-00361).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 3, pp. 0537–0546, March, 2020.
Rights and permissions
About this article
Cite this article
Mironov, M.E., Poltanovich, A.I., Rybalova, T.V. et al. Synthesis and analgesic activity of 1,3,5-trisubstituted pyrazoles containing a diterpenoid moiety. Russ Chem Bull 69, 537–546 (2020). https://doi.org/10.1007/s11172-020-2795-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2795-6