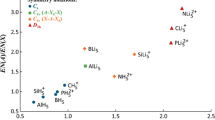
Abstract
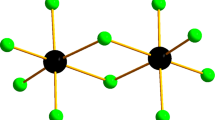
Molecular structure of crystalline complex SbCl5 • Py was determined for the first time and the structure of the complex SbCl5 • AN was refined by means of single crystal X-ray diffraction analysis. It is shown that donor-acceptor Sb–N bond lengths for these complexes are equal within the experimental error. Quantum chemical calculations of the complexes in the gas phase reveal that the energies of the donor-acceptor bond Sb–N are 88 and 180 kJ mol−1 for SbCl5 • AN and SbCl5 • Py, respectively. It is established that the length of the donor-acceptor bond Sb–N in the crystal cannot serve as a measure of its strength.
Similar content being viewed by others
References
E. N. Gur’yanova, I. P. Gol’dshtein, I. P. Romm, Donor-Acceptor Bond, New York, Wiley, 1975, 259.
I. P. Romm, Yu. G. Noskov, A. A. Malkov, Russ. Chem. Bull., 2007, 56, 1935.
K. R. Leopold, M. Canagaratna, J. A. Phillips, Acc. Chem. Res., 1997, 30, 57.
A. Y. Timoshkin, M. Bodensteiner, T. N. Sevastianova, A. S. Lisovenko, E. I. Davydova, M. Scheer, C. Graßl, A. V. Butlak, Inorg. Chem., 2012, 51, 11602.
V. Gutmann, A. Steininger, E. Wychera, Monatsh. Chem., 1966, 97, 460.
H. Binas, Z. Anorg. Allg. Chem., 1967, 352, 271.
T. M. Klapötke, Y. Nöth, T. Schütt, M. Suter, M. Warchhold, Z. Anorg. Allg. Chem., 2001, 627, 1582.
J. C. Jochims, R. Abu-El-Halawa, L. Zsolnai, G. Huttner, Chem. Ber., 1984, 117, 1161.
H.-D. Hausen, W. Schwarz, G. Rajca, J. Weidlein, Z. Naturforsch., 1986, 41, 1223.
D. Bellamy, N. C. Brown, N. G. Connelly, A. G. Orpen, J. Chem. Soc., Dalton Trans., 1999, 3191.
S. I. Troyanov, Zh. Neorg. Khim., 2005, 50, 1845 [Russ. J. Inorg. Chem. (Engl. Transl.), 2005, 50] (in Russian).
O. Kh. Poleshchuk, E. L. Shevchenko, V. Branchadell, M. Lein, G. Frenking, Int. J. Quant. Chem., 2005, 101, 869.
CrysAlisPro Program., Rigaku Oxford Diffraction, 2018.
G. M. Sheldrick, Acta Crystallogr., Ser. C, 2015, 71, 3.
C. R. Groom, I. J. Bruno, M. P. Lightfoot, S. C. Ward, Acta Crystallogr., Ser. B, 2016, 72, 171.
D. A. Doinikov, I. V. Kazakov, I. S. Krasnova, A. Yu. Timoshkin, Russ. J. Phys. Chem. (Engl. Transl.), 2017, 91, 1603.
E. I. Davydova, D. A. Doinikov, I. V. Kazakov, I. S. Krasnova, T. N. Sevast’yanova, A. V. Suvorov, A. Yu. Timoshkin, Russ. J. Gen. Chem. (Engl. Transl.), 2019, 89, 1069.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, D. J. Fox, Gaussian 16, Revision A.03; Gaussian, Wallingford (CT), 2016.
Y. Zhao, D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215.
D. Feller, J. Comp. Chem., 1996, 17, 1571.
K. L. Schuchardt, B. T. Didier, T. Elsethagen, L. Sun, V. Gurumoorthi, J. Chase, J. Li, T. L. Windus, J. Chem. Inf. Model., 2007, 47, 1045.
A. Yu. Timoshkin, T. N. Sevast’yanova, E. I. Davydova, A. V. Suvorov, H. F. Schaefer, Russ. J. Gen. Chem. (Engl. Transl.), 2002, 72, 1911.
A. Y. Timoshkin, E. I. Davydova, T. N. Sevastianova, A. V. Suvorov, H. F. Schaefer, Int. J. Quant. Chem., 2002, 88, 436.
S. F. Boys, F. Bernardi, Mol. Phys., 1970, 19, 553.
F. B. van Duijneveldt, J. G. C. M. van Duijneveldt-van de Rijdt, J. H. van Lenthe, Chem. Rev., 1994, 94, 1873.
E. D. Glendening, A. E. Reed, J. E. Carpenter, F. Weinhold, NBO, Version 3.1.
J. P. Foster, F. Weinhold, J. Am. Chem. Soc., 1980, 102, 7211.
A. E. Reed, R. B. Weinstock, F. Weinhold, J. Chem. Phys., 1985, 83, 735.
A. E. Reed, L. A. Curtiss, F. Weinhold, Chem. Rev., 1988, 88, 899.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the 5th EUChemS Inorganic Chemistry Conference (EICC-5) (June 24–28, 2019, Moscow, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 00843–0090, January, 2020.
Rights and permissions
About this article
Cite this article
Davydova, E.I., Virovets, A.V., Peresypkina, E.V. et al. Structures of molecular complexes of SbCl5 with pyridine and acetonitrile: equal bond lengths, different stability. Russ Chem Bull 69, 84–90 (2020). https://doi.org/10.1007/s11172-020-2726-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2726-6