Abstract
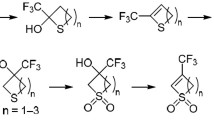
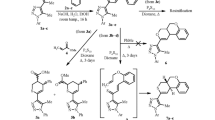
A three-component one-pot procedure to synthesize 2,4-disubstituted b-fused 3,4-dihydro-2H-thiopyran derivatives by the Diels—Alder reaction of the in situ generated α,β-enethiones with maleic acid derivatives was developed. The reactivity of different unsaturated thioketones as dienes and maleic acid derivatives as dienophiles under the developed reaction conditions was studied.
Similar content being viewed by others
References
M. Rajabi, M. A. Khalilzadeh, J. Mehrzad, DNA Cell Biol., 2012, 31, 128.
M. J. Brown, P. S. Carter, A. E. Fenwick, A. P. Fosberry, D. W. Hamprecht, M. J. Hibbs, R L. Jarvest, L. Mensah, P. H. Milner, P. J. O’Hanlon, A. J. Pope, C. M. Richardson, A. West, D. R. Witty, Bioorg. Med. Chem. Lett., 2002, 12, 3171.
R. G. Wilde, J. T. Billheimer, S. J. Germain, E. A. Hausner, P. C. Meunier, D. A. Munzer, J. K. Stoltenborg, P. J. Gillies, Bioorg. Med. Chem., 1996, 4, 1493.
R. S. Glass, Q. Guo, Y. Liu, Tetrahedron, 1997, 53, 12273.
J. K. Gallos, C. C. Dellios, J. Heterocycl. Chem., 2001, 38, 579.
E. Block, R. Iyer, S. Grisoni, C. Saha, S. Belman, F. P. Lossing, J. Am. Chem. Soc., 1988, 110, 7813.
J. R. Mahajan, G. A. L. Ferreira, H. C. Araújo, B. J. Nunes, Synthesis, 1976, 113.
K. Gollnick, K. Knutzen-Mies, J. Org. Chem., 1991, 56, 4027–4031.
V. G. Kharchenko, S. N. Chalaya, Tiopirany, soli thiopiriliya irodstvennye soedineniya [Thiopyrans, Thiopyrylium Salts, and the Related Compounds], Izd-vo Saratov Univ., Saratov, 1987, 60 pp. (in Russian).
M. Keizo, O. Masanobu, H. Yoko, U. Sachiko, T. Yuki, A. Takahiko, T. Kuniyoshi, Chem. Express, 1993, 8, 293.
M. S. J. Briggs, M. Helliwell, D. Moorcroft, E. J. Thomas, J. Chem. Soc., Perkin Trans. 1, 1992, 2223.
S. K. Klimenko, T. I. Tyrina, N. N. Sorokin, Chem. Heterocycl. Compd., 1987, 23, 506.
N. Al-Masoudi, N. A. Hussien, J. Iraqi Chem. Soc., 1986, 12, 149.
Q. Zhao, L. Li, Y. Fang, J. Org. Chem., 2009, 74, 459.
S. Wang, Y. Zhang, G. Dong, S. Wu, S. Zhu, Z. Miao, J. Yao, H. Li, J. Li, W. Zhang, C. Sheng, W. Wang, Org. Lett., 2013, 15, 5570.
S. Ohsugi, K. Nishide, M. Node, Tetrahedron, 2003, 59, 1859.
A. C. Gaumont, L. Wazneh, J.-M. Denis, Tetrahedron, 1991, 47, 4927.
K. Shunichi, O. Haruo, T. Hideyuki, M. Shinichi, Chem. Lett., 1982, 11, 793.
T. Karakasa, S. Motoki, J. Org. Chem., 1978, 43, 4147.
S. Moriyama, T. Karakasa, S. Motoki, Bull. Chem. Soc. Jpn., 1990, 63, 2540.
S. Motoki, T. Saito, T. Karakasa, T. Matsushita, E. Furuno, J. Chem. Soc., Perkin Trans. 2, 1992, 21, 2943.
T. Karakasa, S. Motoki, J. Org. Chem., 1979, 44, 4151.
T. Saito, H. Fujii, S. Hayashibe, T. Matsushita, H. Kato, K. Kobayashi, J. Chem. Soc., Perkin Trans. 1, 1996, 1897.
S. Scheibye, R. Shabana, S. Lawesson, Tetrahedron, 1982, 38, 993.
M. J. Milewska, T. Bytner, T. Połoński, Synthesis, 1996, 12, 1485.
M. V. Lakshmikantham, W. Chen, M. P. Cava, J. Org. Chem., 1989, 54, 4746.
T. Ozturk, E. Ertas, O. Mert, Chem. Rev., 2007, 107, 5210.
K. Hartke, O. Kunze, Liebigs Ann. Chem., 1989, 321.
M. P. Cava, M. I. Levinson, Tetrahedron, 1985, 41, 5061.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the V All-Russian Organic Chemistry Conference (ROCC-V) (September 10–14, 2018, Vladikavkaz, Russia).
Rights and permissions
About this article
Cite this article
Merkulova, E.A., Kolobov, A.V. & Ovchinnikov, K.L. A convenient synthesise of 3,4-dihydro-2H-thiopyran-2,3-dicarboxylic acid derivatives. Russ Chem Bull 68, 606–609 (2019). https://doi.org/10.1007/s11172-019-2462-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-019-2462-y