Abstract
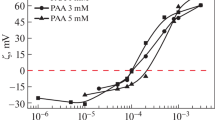
The aggregation characteristics of binary systems based on a novel pyrrolidinium surfactant with the hexadecyl hydrocarbon tail and hydroxyethyl fragment at the nitrogen atom were studied in the presence of polyacrylic acid by a complex of physicochemical methods (tensiometry, conductometry, dynamic light scattering, fluorescence spectroscopy, and spectrophotometry). The formation of surfactant–polyelectrolyte complexes leads to a decrease in the concentration thresholds of aggregation by an order of magnitude to form aggregates ~70 nm in size was established by tensiometry and fluorimetry. The solubilization capacity of the formed mixed aggregates was tested using the Orange OT hydrophobic dye.
Similar content being viewed by others
References
S. Svenson, Curr. Opin. Colloid Interface Sci., 2004, 9, 201; DOI: 10.1016/j.cocis.2004.06.008.
M. C Jennings, K. P. C. Minbiole, W. M. Wuest, ACS Infect. Dis., 2016, 1, No. 7, 288; DOI: 10.1021/acsinfecdis.5b00047.
N. J. Turro, M. Gratzel, A. M. Braun, Angew Chem. Int. Ed. Engl., 1980, 19, 675; DOI: org/10.1002/anie.198006751.
L. Ya. Zakharova, E. P. Zhiltsova, A. B. Mirgorodskaya, A. I. Konovalov, From Molecules to Functional Architecture. Supramolecular Interactions, Donetsk, East Publisher House, 2012, 415 pp.
K. S. Egorova, V. P. Ananikov, J. Mol. Liq., 2018, 272, 271; DOI: 10.1016/j.molliq.2018.09.025.
M. W. Zhao, L. Q. Zheng, Chem. Phys., 2011, 13, 1332; DOI: 10.1039/C0CP00342E.
G. A. Baker, S. Pandey, S. Pandey, Analyst, 2004, 129, 890; DOI: 10.1039/B410301G.
V. P. Schnee, G. A. Baker, E. Rauk, C. P. Palmer, Electrophoresis, 2006, 27, 4141.
K. K. Karukstis, J. R. McDonough, Langmuir, 2005, 21, 5716.
N. V. Sastry, N. M. Vaghela, V. K. Aswal, Fluid Phase Equilib., 2012, 327, 22; DOI: 10.1016/j.fluid.2012.04.013.
L. Shi, M. Zhao, L. Zheng, Colloids Surf. A, 2011, 392, 305; DOI: 10.1016/j.colsurfa.2011.09.064.
L. Zhou, T. Tian, J. Xiao, T. Wang, L. Yu, J. Mol. Liq., 2017, 225, 50; DOI: 10.1016/j.molliq.2016.10.142.
Y. Tian, R. Wei, B. Cai, J. Dong, B. Deng, Y. Xiao, J. Chromatogr. A, 2016, 1475, 95; DOI: 10.1016/j.chroma.2016.11.001.
A. B. Mirgorodskaya, E. I. Yackevich, S. S. Lukashenko, L. Ya. Zakharova, A. I. Konovalov, J. Mol. Liq., 2012, 169, 106; DOI: 10.1016/j.molliq.2012.02.012.
L. Ya. Zakharova, A. B. Mirgorodskaya, E. I. Yackevich, V. V. Syakaev, Sh. K. Latypov, A. I. Konovalov, J. Chem. Eng. Data, 2012, 57, 3153; DOI: 10.1021/je300753d.
E. A. Vasilieva, S. S. Lukashenko, A. D. Voloshina, A. S. Strobykina, L. A. Vasileva, L. Ya. Zakharova, Russ. Chem. Bull., 2018, 67, 1280.
D. A. Samarkina, D. R. Gabdrakhmanov, S. S. Lukashenko, A. R. Khamatgalimov, V. I. Kovalenko, L. Ya. Zakharova, Colloids Surf., A, 2017, 529, 990; DOI: 10.1016/j.colsurfa. 2017.07.018.
A. B. Mirgorodskaya, E. I. Yacke vich, D. R. Gabdrakhmanov, S. S. Lukashenko, Yu. F. Zuev, L. Ya. Zakharova, J. Mol. Liq., 2016, 220, 992; DOI: 10.1016/j.molliq.2016.05.010.
M. Zhao, J. Yuan, L. Zheng, Colloids and Surfaces A: Physicochem. Eng. Aspects, 2012, 407, 116.
P. Singla, O. Singh, Sh. Chabba, R. K. Mahajan, J. Mol. Liq., 2018, 249, 294.
K. Holmberg, B. Jönsson, B. Kronberg, B. Lindman, Surfactants and Polymers in Aqueous Solution, John Wiley&Sons Ltd., Chichester, 2003, 547 pp.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences A. I. Konovalov on the occasion of his 85th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 2, pp. 0341–0346, February, 2019.
Rights and permissions
About this article
Cite this article
Vasilieva, E.A., Lukashenko, S.S., Vasileva, L.A. et al. Aggregation behavior of the surfactant bearing pyrrolidinium head group in the presence of polyacrylic acid. Russ Chem Bull 68, 341–346 (2019). https://doi.org/10.1007/s11172-019-2390-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-019-2390-x