Abstract
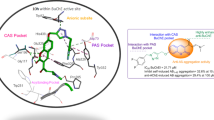
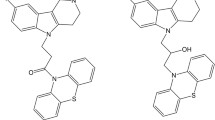
The paper considers the influence of the structure of aminoadamantane–carbazole conjugates linked by five different spacers on their ability to inhibit acetylcholinesterase, butyrylcholinesterase (BChE), and carboxylesterase, the radical scavenging and mitoprotective activity, and tubulin polymerization. The nature of the spacer was found to be determinant for the appearance of anti-BChE and radical-scavenging activity. Aminoadamantane–carbazole conjugates linked by flexible spacers proved to be most effective in these assays. The influence of the conjugates on mitochondrial functions and tubulin polymerization largely depends on the structure of the carbazole moiety. A series of lead compounds promising for further study was identified as potential drugs for the treatment of neurodegenerative diseases, in particular, Alzheimer’s disease.
Similar content being viewed by others
References
Y. Huang, L. Mucke, Cell, 2012, 148, 1204; DOI: 10.1016/j. cell.2012.02.040.
M. L. Bolognesi, A. Cavalli, ChemMedChem, 2016, 11, 1190; DOI: 10.1002/cmdc.201600161.
M. Carreiras, E. Mendes, M. Perry, A. Francisco, J. Marco-Contelles, Curr. Top. Med. Chem., 2013, 13, 1745; DOI: 10.2174/15680266113139990135.
E. F. Shevtsova, D. V. Vinogradova, M. E. Neganova, M. Avila-Rodriguez, G. M. Ashraf, G. E. Barreto, S. O. Bachurin, G. Aliev, CNS Neurol. Disord. Drug Targets, 2017, 16, 677; DOI: 10.2174/1871527316666170424114444.
N. Lermontova, N. Lukoyanov, T. Serkova, E. Lukoyanova, S. Bachurin, Mol. Chem. Neuropathol., 1998, 33, 51; DOI: 10.1007/bf02815859.
H. W. Klafki, M. Staufenbiel, J. Kornhuber, J. Wiltfang, Brain, 2006, 129, 2840; DOI: 10.1093/brain/awl280.
J. Rodda, J. Carter, BMJj, 2012, 344, e2986; DOI: 10.1136/bmj.e2986.
F. Mangialasche, A. Solomon, B. Winblad, P. Mecocci, M. Kivipelto, Lancet Neurol., 2010, 9, 702; DOI: 10.1016/S1474-4422(10)70119-8.
M. Rosini, E. Simoni, A. Minarini, C. Melchiorre, Neurochem. Res., 2014, 39, 1914; DOI: 10.1007/s11064-014-1250-1.
A. Agis-Torres, M. Solhuber, M. Fernandez, J. M. Sanchez-Montero, Curr. Neuropharmacol., 2014, 12, 2; DOI: 10.2174/1570159X113116660047.
S. O. Bachurin, E. V. Bovina, A. A. Ustyugov, Med. Res. Rev., 2017, 37, 1186; DOI: 10.1002/med.21434.
S. O. Bachurin, E. F. Shevtsova, G. F. Makhaeva, V. V. Grigoriev, N. P. Boltneva, N. V. Kovaleva, S. V. Lushchekina, P. N. Shevtsov, M. E. Neganova, O. M. Redkozubova, E. V. Bovina, A. V. Gabrelyan, V. P. Fisenko, V. B. Sokolov, A. Y. Aksinenko, V. Echeverria, G. E. Barreto, G. Aliev, Sci. Rep., 2017, 7, 45627; DOI: 10.1038/srep45627.
G. F. Makhaeva, V. B. Sokolov, E. F. Shevtsova, N. V. Kovaleva, S. V. Lushchekina, N. P. Boltneva, E. V. Rudakova, A. Y. Aksinenko, P. N. Shevtsov, M. E. Neganova, L. G. Dubova, S. O. Bachurin, Pure Appl. Chem., 2017, 89; DOI: 10.1515/pac-2017-0308.
S. O. Bachurin, Zh. Nevrol. Psikhiatr. Im. S. S. Korsakova, 2016, 116, 77; DOI: 10.17116/jnevro20161168177-87.
A. Cavalli, M. L. Bolognesi, A. Minarini, M. Rosini, V. Tumiatti, M. Recanatini, C. Melchiorre, J. Med. Chem., 2008, 51, 347; DOI: 10.1021/jm7009364.
V. B. Sokolov, A. Y. Aksinenko, T. V. Goreva, T. A. Epishina, V. V. Grigor’ev, A. V. Gabrel’yan, D. V. Vinogradova, M. E. Neganova, E. F. Shevtsova, S. O. Bachurin, Russ. Chem. Bull., 2016, 65, 1354; DOI: 10.1007/s11172-016-1461-5.
V. B. Sokolov, A. Y. Aksinenko, T. A. Epishina, T. V. Goreva, S. O. Bachurin, Russ. Chem. Bull., 2017, 66, 2110; DOI: 10.1007/s11172-017-1988-0.
S. O. Bachurin, V. B. Sokolov, A. Y. Aksinenko, T. A. Epishina, T. V. Goreva, E. F. Shevtsova, P. N. Shevtsov, Pat. RF 2608631, 2017 (in Russian).
A. Nordberg, C. Ballard, R. Bullock, T. Darreh-Shori, M. Somogyi, Prim. Care Companion. CNS Disord., 2013, 15, PCC.12r01412; DOI: 10.4088/PCC.12r01412.
G. L. Ellman, K. D. Courtney, V. Andres, M. Feather-Stone, Biochem. Pharmacol., 1961, 7, 88; DOI: 10.1016/0006-2952(61)90145-9.
S. H. Sterri, B. A. Johnsen, F. Fonnum, Biochem. Pharmacol., 1985, 34, 2779; DOI: 10.1016/0006-2952(85)90579-9.
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Rice-Evans, Free Radic. Biol. Med., 1999, 26, 1231; DOI: 10.1016/s0891-5849(98)00315-3.
E. R. Milaeva, D. B. Shpakovsky, Y. A. Gracheva, T. A. Antonenko, D. I. Osolodkin, V. A. Palyulin, P. N. Shevtsov, M. E. Neganova, D. V. Vinogradova, E. F. Shevtsova, J. Organomet. Chem., 2015, 782, 96; DOI: 10.1016/j.jorganchem.2014.12.013.
P. N. Shevtsov, E. F. Shevtsova, G. Burbaeva, Bull. Exp. Biol. Med., 2016, 161, 451; DOI: 10.1007/s10517-016-3436-9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2121–2126, November, 2018.
Rights and permissions
About this article
Cite this article
Makhaeva, G.F., Shevtsova, E.F., Kovaleva, N.V. et al. Aminoadamantane conjugates with carbazole derivatives as potential multitarget agents for the treatment of Alzheimer’s disease. Effect of the spacer structure. Russ Chem Bull 67, 2121–2126 (2018). https://doi.org/10.1007/s11172-018-2338-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2338-6