Abstract
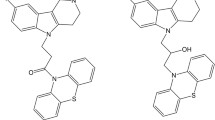
A comparative analysis of the esterase profile and antiradical activity of two groups of hybrid compounds, viz., tetrahydro-γ-carboline conjugates with carbazoles and tetrahydrocarbazoles (I) and carbazole conjugates with carbazoles and tetrahydrocarbazoles (II), was performed. The replacement of the tetrahydro-γ-carboline moiety (conjugates I) by the carbazole group (conjugates II) was shown to significantly reduce the ability of the compounds to inhibit butyrylcholinesterase (BChE) and scavenge free radicals. The tetrahydro-γ-carboline–tetrahydrocarbazole combination is optimal in terms of both high anti-BChE activity and free radical scavenging ability. According to molecular modeling calculations, the stronger binding of tetrahydro-γ-carboline conjugates (I) in the BChE active site compared to carbazole conjugates (II) is attributed to the ability of I to form ionic and π-cation interactions with amino acid residues lining the BChE gorge. Therefore, conjugates of tetrahydro-γ-carboline and tetrahydrocarbazole derivatives are the most promising compounds for the design of new multitarget drugs combining cognitive-stimulating and antioxidant properties.
Similar content being viewed by others
References
M. C. Carreiras, E. Mendes, M. J. Perry, A. P. Francisco, J. Marco-Contelles, Curr. Top. Med. Chem., 2013, 13, 1745; DOI: 10.2174/15680266113139990135.
S. O. Bachurin, E. V. Bovina, A. A. Ustyugov, Med. Res. Rev., 2017, 37, 1186; DOI: 10.1002/med.21434.
S. O. Bachurin, E. F. Shevtsova, G. F. Makhaeva, V. V. Grigoriev, N. P. Boltneva, N. V. Kovaleva, S. V. Lushchekina, P. N. Shevtsov, M. E. Neganova, O. M. Redkozubova, E. V. Bovina, A. V. Gabrelyan, V. P. Fisenko, V. B. Sokolov, A. Y. Aksinenko, V. Echeverria, G. E. Barreto, G. Aliev, Sci. Rep., 2017, 7, 45627; DOI: 10.1038/srep45627.
M. L. Bolognesi, A. Cavalli, ChemMedChem, 2016, 11, 1190; DOI: 10.1002/cmdc.201600161.
S. O. Bachurin, Neurosci. Behav. Phisicl., 2016, 116, 77; DOI: 10.17116/jnevro20161168177-87.
G. F. Makhaeva, S. V. Lushchekina, N. P. Boltneva, V. B. Sokolov, V. V. Grigoriev, O. G. Serebryakova, E. A. Vikhareva, A. Y. Aksinenko, G. E. Barreto, G. Aliev, S. O. Bachurin, Sci. Rep., 2015, 5, 13164; DOI: 10.1038/srep13164.
A. Agis-Torres, M. Solhuber, M. Fernandez, J. M. Sanchez-Montero, Curr. Neuropharmacol., 2014, 12, 2; DOI: 10.2174/1570159X113116660047.
M. L. Bolognesi, E. Simoni, M. Rosini, A. Minarini, V. Tumiatti, C. Melchiorre, Curr. Top. Med. Chem., 2011, 11, 2797; DOI: 10.2174/156802611798184373.
K. S. Dias, C. Viegas, Jr., Curr. Neuropharmacol., 2014, 12, 239; DOI: 10.2174/1570159X1203140511153200.
E. J. Mufson, S. E. Counts, S. E. Perez, S. D. Ginsberg, Expert Rev. Neurother., 2008, 8, 1703; DOI: 10.1586/14737175.8.11.1703.
N. Lermontova, N. Lukoyanov, T. Serkova, E. Lukoyanova, S. Bachurin, Mol. Chem. Neuropathol., 1998, 33, 51; DOI: 10.1007/bf02815859.
H. W. Klafki, M. Staufenbiel, J. Kornhuber, J. Wiltfang, Brain, 2006, 129, 2840; DOI: 10.1093/brain/awl280.
F. Mangialasche, A. Solomon, B. Winblad, P. Mecocci, M. Kivipelto, Lancet Neurol., 2010, 9, 702; DOI: 10.1016/S1474-4422(10)70119-8.
J. Rodda, J. Carter, BMJ, 2012, 344, e2986; DOI: 10.1136/bmj.e2986.
P. I. Moreira, S. L. Siedlak, G. Aliev, X. Zhu, A. D. Cash, M. A. Smith, G. Perry, J. Neural Transm. (Vienna), 2005, 112, 921; DOI: 10.1007/s00702-004-0242-8.
R. Floyd, K. Hensley, Neurobiol. Aging, 2002, 23, 795; DOI: 10.1016/s0197-4580(02)00019-2.
M. Rosini, E. Simoni, M. Bartolini, A. Tarozzi, R. Matera, A. Milelli, P. Hrelia, V. Andrisano, M. L. Bolognesi, C. Melchiorre, Eur. J. Med. Chem., 2011, 46, 5435; DOI: 10.1016/j.ejmech.2011.09.001.
N. Guzior, A. Wieckowska, D. Panek, B. Malawska, Curr. Med. Chem., 2015, 22, 373; DOI: 10.2174/092986732166614 1106122628.
N. Oztaskin, Y. Cetinkaya, P. Taslimi, S. Goksu, I. Gulcin, Bioorg. Chem., 2015, 60, 49; DOI: 10.1016/j.bioorg.2015.04.006.
A. Rampa, F. Belluti, S. Gobbi, A. Bisi, Curr. Top. Med. Chem., 2011, 11, 2716; DOI: 10.2174/156802611798184409.
G. F. Makhaeva, V. B. Sokolov, E. F. Shevtsova, N. V. Kovaleva, S. V. Lushchekina, N. P. Boltneva, E. V. Rudakova, A. Y. Aksinenko, P. N. Shevtsov, M. E. Neganova, L. G. Dubova, S. O. Bachurin, Pure Appl. Chem., 2017, 89, 1167; DOI: 10.1515/pac-2017-0308.
G. F. Makhaeva, V. V. Grigoriev, A. N. Proshin, N. V. Kovaleva, E. V. Rudakova, N. P. Boltneva, I. V. Serkov, S. O. Bachurin, Dokl. Biochem. Biophys., 2017, 477, 405; DOI: 10.1134/S1607672917060163.
M. Singh, M. Kaur, N. Chadha, O. Silakari, Mol. Divers., 2016, 20, 271; DOI: 10.1007/s11030-015-9628-9.
M. Rosini, Future Med. Chem., 2014, 6, 485; DOI: 10.4155/fmc.14.25.
A. Cavalli, M. L. Bolognesi, A. Minarini, M. Rosini, V. Tumiatti, M. Recanatini, C. Melchiorre, J. Med. Chem., 2008, 51, 347; DOI: 10.1021/jm7009364.
V. B. Sokolov, G. F. Makhaeva, A. Y. Aksinenko, V. V. Grigoriev, E. F. Shevtsova, S. O. Bachurin, Russ. Chem. Bull., 2017, 66, 1821; DOI: 10.1007/s11172-017-1953-y.
V. B. Sokolov, A. Y. Aksinenko, T. V. Goreva, T. A. Epishina, L. G. Dubova, E. S. Dubrovskaya, S. G. Klochkov, P. N. Shevtsov, E. F. Shevtsova, S. O. Bachurin, Russ. Chem. Bull., 2016, 65, 2306; DOI: 10.1007/s11172-016-1582-x.
V. B. Sokolov, A. Y. Aksinenko, T. V. Goreva, T. A. Epishina, V. V. Grigor’ev, A. V. Gabrel’yan, D. V. Vinogradova, L. G. Dubova, P. N. Shevtsov, E. F. Shevtsova, S. O. Bachurin, Russ. Chem. Bull., 2016, 65, 1346; DOI: 10.1007/s11172-016-1460-6.
V. B. Sokolov, A. Y. Aksinenko, T. A. Epishina, T. V. Goreva, Russ. J. Gen. Chem., 2016, 86, 1827; DOI: 10.1134/s1070363216080089.
V. B. Sokolov, A. Y. Aksinenko, T. A. Epishina, T. V. Goreva, Russ. J. Gen. Chem., 2016, 86, 2394; DOI: 10.1134/s1070363216100248.
S. Bachurin, E. Bukatina, N. Lermontova, S. Tkachenko, A. Afanasiev, V. Grigoriev, I. Grigorieva, Y. U. Ivanov, S. Sablin, N. Zefirov, Ann. N. Y. Acad. Sci., 2006, 939, 425; DOI: 10.1111/j.1749-6632.2001.tb03654.x.
R. S. Doody, S. I. Gavrilova, M. Sano, R. G. Thomas, P. S. Aisen, S. O. Bachurin, L. Seely, D. Hung, I. Dimebon, Lancet, 2008, 372, 207; DOI: 10.1016/S0140-6736(08)61074-0.
R. Otto, R. Penzis, F. Gaube, T. Winckler, D. Appenroth, C. Fleck, C. Trankle, J. Lehmann, C. Enzensperger, Eur. J. Med. Chem., 2014, 87, 63; DOI: 10.1016/j.ejmech.2014.09.048.
R. Otto, R. Penzis, F. Gaube, O. Adolph, K. J. Fohr, P. Warncke, D. Robaa, D. Appenroth, C. Fleck, C. Enzensperger, J. Lehmann, T. Winckler, J. Med. Chem., 2015, 58, 6710; DOI: 10.1021/acs.jmedchem.5b00958.
I. Okun, S. Tkachenko, A. Khvat, O. Mitkin, V. Kazey, A. Ivachtchenko, Curr. Alzheimer Res., 2010, 7, 97; DOI: 10.2174/156720510790691100.
A. Ustyugov, E. Shevtsova, S. Bachurin, Mol. Neurobiol., 2015, 52, 970; DOI: 10.1007/s12035-015-9249-4.
H. Schaffhauser, J. R. Mathiasen, A. Dicamillo, M. J. Huffman, L. D. Lu, B. A. Mckenna, J. Qian, M. J. Marino, Biochem. Pharmacol., 2009, 78, 1035; DOI: 10.1016/j. bcp.2009.06.021.
J. Vignisse, H. W. Steinbusch, A. Bolkunov, J. Nunes, A. I. Santos, C. Grandfils, S. Bachurin, T. Strekalova, Prog. Neuropsychopharmacol. Biol. Psychiatry, 2011, 35, 510; DOI: 10.1016/j.pnpbp.2010.12.007.
N. Cano-Cuenca, J. E. Solis-Garcia Del Pozo, J. Jordan, J. Alzheimers Dis., 2014, 38, 155; DOI: 10.3233/JAD-130872.
E. Malatynska, H. W. Steinbusch, O. Redkozubova, A. Bolkunov, A. Kubatiev, N. B. Yeritsyan, J. Vignisse, S. Bachurin, T. Strekalova, Exp. Gerontol., 2012, 47, 552; DOI: 10.1016/j. exger.2012.04.010.
O. M. Peters, N. Connor-Robson, V. B. Sokolov, A. Y. Aksinenko, M. S. Kukharsky, S. O. Bachurin, N. Ninkina, V. L. Buchman, J. Alzheimers Dis., 2013, 33, 1041; DOI: 10.3233/JAD-2012-121732.
P. R. Bharadwaj, K. A. Bates, T. Porter, E. Teimouri, G. Perry, J. W. Steele, S. Gandy, D. Groth, R. N. Martins, G. Verdile, Transl. Psychiatry, 2013, 3, e332; DOI: 10.1038/tp.2013.97.
J. W. Steele, S. Gandy, Autophagy, 2013, 9, 617; DOI: 10.4161/auto.23487.
A. Gluszynska, Eur. J. Med. Chem., 2015, 94, 405; DOI: 10.1016/j.ejmech.2015.02.059
K. Honarnejad, A. Daschner, A. P. Gehring, A. Szybinska, A. Giese, J. Kuznicki, F. Bracher, J. Herms, Transl. Psychiatry, 2014, 4, e489; DOI: 10.1038/tp.2014.132.
M. Bashir, A. Bano, A. S. Ijaz, B. A. Chaudhary, Molecules, 2015, 20, 13496; DOI: 10.3390/molecules200813496.
K. S. Macmillan, J. Naidoo, J. Liang, L. Melito, N. S. Williams, L. Morlock, P. J. Huntington, S. J. Estill, J. Longgood, G. L. Becker, S. L. Mcknight, A. A. Pieper, J. K. De Brabander, J. M. Ready, J. Am. Chem. Soc., 2011, 133, 1428; DOI: 10.1021/ja108211m.
H. J. Yoon, S. Y. Kong, M. H. Park, Y. Cho, S. E. Kim, J. Y. Shin, S. Jung, J. Lee, Farhanullah, H. J. Kim, J. Lee, Bioorg. Med. Chem., 2013, 21, 7165; DOI: 10.1016/j.bmc.2013.08.066.
D. Zhu, M. Chen, M. Li, B. Luo, Y. Zhao, P. Huang, F. Xue, S. Rapposelli, R. Pi, S. Wen, Eur. J. Med. Chem., 2013, 68, 81; DOI: 10.1016/j.ejmech.2013.07.029.
A. A. Pieper, S. L. Mcknight, J. M. Ready, Chem. Soc. Rev., 2014, 43, 6716; DOI: 10.1039/c3cs60448a.
E. S. Alekseev, T. V. Bogdan, Russ. J. Struct. Chem., 2016, 57, 1568; DOI: 10.1134/s0022476616080114.
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Rice-Evans, Free Radic. Biol. Med., 1999, 26, 1231; DOI: 10.1016/s0891-5849(98)00315-3.
G. L. Ellman, K. D. Courtney, V. Andres, Jr., R. M. Feather-Stone, Biochem. Pharmacol., 1961, 7, 88; DOI: 10.1016/0006-2952(61)90145-9.
S. H. Sterri, B. A. Johnsen, F. Fonnum, Biochem. Pharmacol., 1985, 34, 2779; DOI: 10.1016/0006-2952(85)90579-9.
Y. Nicolet, O. Lockridge, P. Masson, J. C. Fontecilla-Camps, F. Nachon, J. Biol. Chem., 2003, 278, 41141; DOI: 10.1074/jbc.M210241200.
S. V. Lushchekina, V. S. Polomskikh, S. D. Varfolomeev, P. Masson, Russ. Chem. Bull., 2013, 62, 2527; DOI: 10.1007/s11172-013-0366-9.
P. Masson, S. Lushchekina, L. M. Schopfer, O. Lockridge, Biochem. J., 2013, 454, 387; DOI: 10.1042/BJ20130389.
G. M. Morris, D. S. Goodsell, R. S. Halliday, R. Huey, W. E. Hart, R. K. Belew, A. J. Olson, J. Comput. Chem., 1998, 19, 1639; DOI: 10.1002/(sici)1096-987x(19981115)19:14<1639::aid-jcc10>3.0.co;2-b.
G. M. Morris, R. Huey, W. Lindstrom, M. F. Sanner, R. K. Belew, D. S. Goodsell, A. J. Olson, J. Comput. Chem., 2009, 30, 2785; DOI: 10.1002/jcc.21256.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 9, pp. 1724–1731, September, 2018.
Rights and permissions
About this article
Cite this article
Makhaeva, G.F., Boltneva, N.P., Kovaleva, N.V. et al. Influence of the γ-carboline and carbazole pharmacophore moieties on anticholinesterase and antiradical activity of multifunctional agents for the treatment of neurodegenerative diseases. Russ Chem Bull 67, 1724–1731 (2018). https://doi.org/10.1007/s11172-018-2282-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2282-5