Abstract
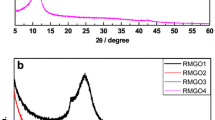
Graphene oxide was obtained from expanded graphite according to the modified Hummers method. The reaction time was significantly reduced (from 24 to 8 h) by applying the expanded graphite in the oxidation process. The interlayer distance in obtained samples was equal to 0.70–0.74 nm. Structural features of the material were studied by the powder X-ray diffraction, thermogravimetric analysis, scanning electron microscopy, transmission electron microscopy, XPS spectroscopy, and NMR spectroscopy.
Similar content being viewed by others
References
G. Wang, X. Sun, C. Li, J. Lian, Appl. Phys. Lett., 2011, 99: 053114:1–053114:3.
K. Krishnamoorthy, R. Mohan, S.-J. Kim, Appl. Phys. Lett., 2011, 98: 244101:1–244101:3.
U. Khan, P. May, A. O. Neill, J. N. Coleman, Carbon, 2010, 48: 4035–4041.
Q. Cheng, J. Tang, J. Ma, H. Zhang, N. Shinya, L.-C. Qin, Carbon, 2011, 49: 2917–2925.
Ping-Ping Zuo, Hua-Feng Feng, Zhi-Zhen Xu, Ling-Fan Zhang, Yu-Long Zhang, Wei Xia, Wen-Qing Zhang, Chem. Central J., 2013, 7: 49.
D. A. Dikin, S. Stankovich, E. J. Zimney, R. D. Piner, G. H. Dommett, G. Evmenenko, Nature, 2007, 448: 457–460.
D. Long, W. Li, L. Ling, J. Miyawaki, I. Mochida, S.-H. Yoon, Langmuir, 2010, 26: 16096–16102.
D. R. Dreyer, S. Park, C. W. Bielawski, R. S. Ruoff, Chem. Soc. Rev., 2010, 39: 228–240.
J. Kim, L. J. Cote, F. Kim, W. Yuan, K. R. Shull, J. J. Huang, J. Kim, Am. Chem. Soc., 2010, 132: 8180–8186.
B. C. Brodie, Philos Trans. R. Soc. London, 1859, 149: 249–259.
L. Staudenmaier, Ber. Dtsch. Chem. Ges., 1898, 31: 1481–1487.
W. S. Hummers, R. E. Offeman, J. Am. Chem. Soc., 1958, 80: 1339.
C. M. Daniela, V. K. Dmitry, M. B. Jacob, S. Alexander, S. Zhengzong, S. Alexander, B. A. Lawrence, L. Wei, ACS Nano, 2010, 4: 4806–4814.
N. A. Kotov, I. Dekany, J. H. Fendler, Adv. Mater., 1996, 8: 637.
K. Krishnamoorthy, M. Veerapandian, K. Yun, S.-J. Kim, Carbon, 2013, 53: 38–49.
S. V. Tkachev, E. Yu. Buslaeva, A. V. Naumkin, S. L. Kotova, I. V. Laure, S. P. Gubin, Neorgan. Mater., 2012, 48: 796–802 [Inorg. Mater. (Engl. Transl.), 2012, 48].
V. M. Samoilov, A. V. Nikolaeva, E. A. Danilov, G. A. Erpuleva, N. N. Trofimova, S. S. Abramchuk, K. V. Ponkratov, Neorgan. Mater., 2015, 51: 98–105 [Inorg. Mater. (Engl. Transl.), 2015, 51].
G. Furdin, Fuel, 1998, 77: 479–485.
A. S. Fialkov, Uglerod. Mezhsloevye soedineniya i kompozity na ikh osnove [Carbon. Interlayer Compounds and Their Composites], Aspekt-Press, Moscow, 1997, 718 pp (in Russian).
N. V. Chesnokov, B. N. Kuznetsov, N. M. Mikova, Zh. Sib. Fed. Univ., Khimiya [J. Siberian Federal State Univ., Chemistry], Krasnoyarsk, 2013, 6, No. 1, 11–22 (in Russian).
N. E. Sorokina, V. V. Avdeev, A. S. Tikhomirov, M. A. Lutfullin, M. I. Sandaminov, in Kompozitsionnye nanomaterialy na osnove interkalirovannogo grafita. Uchebnoe posobie dlya studentov po spetsialnosti “Kompozitsionnye nanomaterialy” [Composite Materials Based on Intercalated Graphite. Handbook for Students in the Major “Composite Materials”], Moscow State University, Moscow, 2010, p. 50 (in Russian).
G. P. Khokhlova, Ch. N. Barnakov, A. N. Popova, L. M. Khitsova, Keks i Khimiya [Coke and Chemistry], 2015, 58: 268–274 (in Russian).
C. Gómez-Navarro, R. Thomas Weitz, A. M. Bittner, M. Scolari, A. Mews, M. Burghard, K. Kern, Nano Lett., 2007, 7: 3499–3503.
A. V. Eletskii, I. M. Iskandarova, A. A. Knizhnik, D. N. Krasikov, Usp. Fiz. Nauk [Physics-Uspekhi], 2011, 54: 227–259 (in Russian).
S. Stankovich, D. A. Dikin, R. D. Piner, K. A. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S-B. T. Nguyen, R. S. Ruoff, Carbon, 2007, 45: 1558–1565.
Y. Huafeng, S. Changsheng, L. Fenghua, H. Dongxue, Z. Qixian, L. Niu, Chem. Commun., 2009, 26: 3880–3882.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 0986–0990, June, 2018.
Rights and permissions
About this article
Cite this article
Ilkevich, L.V., Tkachenko, T.B., Samarov, A.V. et al. Fabrication and physicochemical characterization of graphene oxide derived from thermally expanded graphite. Russ Chem Bull 67, 986–990 (2018). https://doi.org/10.1007/s11172-018-2168-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2168-6