Abstract
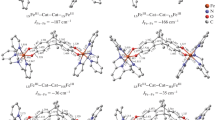
The exchange interaction parameters were calculated and the spin density distribution over the organic skeleton of the 1,3,5,7-tetramethyl-2,6-diazaadamantane N,N’-dioxyl biradical was studied based on the results of quantum chemical modeling of the biradical structure by the DFT method using various hybrid functionals (UB3LYP, LC-wPBE, UCAM-B3LYP, UHSEH1PBE) with the 6-311++G(2d,2p) basis set and by the UHF method with the same basis set. The characteristics of the direct orbital overlap between the N atoms of the two nitroxide groups were determined. The values of the J constant, obtained using different calculation methods, were found to be similar to each other. It was established that there is ferromagnetic exchange interaction between the radical sites in the system in question, which occurs predominantly according to the spin polarization mechanism in the 2,6-diazaadamantane core, and the various spin density transfer pathways through the C atoms of the organic skeleton were found to be nonequivalent. The direct overlap of the upper singly-occupied МОs with localization on the nitroxide groups led to a noticeable additional contribution of the antiferromagnetic exchange interaction. Despite the latter factor, the total contribution of these two mechanisms (spin polarization and direct through-space exchange) resulted in the triplet ground state in the biradical studied.
Similar content being viewed by others
References
C. Train, L. Norel, M. Baumgarten, Coord. Chem. Rev., 2009, 253, 2342.
I. Ratera, J. Veciana, Chem. Soc. Rev., 2012, 41, 303.
M. Tamura, Y. Nakazawa, D. Shiomi, K. Nozawa, Y. Hoso-koshi, M. Ishikawa, M. Takahashi, M. Kinoshita, Chem. Phys. Lett., 1991, 186, 401.
R. Chiarelli, M. A. Novak, A. Rassat, J. L. Tholence, Nature, 1993, 363, 147.
A. J. Banister, N. Bricklebank, I. Lavender, J. M. Rawson, C. I. Gregory, B. K. Tanner, W. Clegg, M. R. J. Elsegood, F. Palacio, Angew. Chem., Int. Ed. Engl., 1996, 35, 2533.
D. Bhattacharya, S. Shil, T. Goswami, A. Misra, A. Panda, D. J. Klein, Comput. Theor. Chem., 2013, 1024, 15.
D. Bhattacharya, S. Shil, T. Goswami, A. Misra, D. J. Klein, Comp. Theor. Chem., 2014, 1039, 11.
S. Lloyd, Science, 1993, 261, 1569.
A. K. Ekert, R. Jozsa, Rev. Mod. Phys., 1996, 68, 733.
L. K. Grover, Phys. Rev. Lett., 1997, 79, 4709.
M. Mas-Torrent, N. Crivillers, V. Mugnaini, I. Ratera, C. Rovira, J. Veciana, J. Mater. Chem., 2009, 19, 1691.
S. Nakazawa, S. Nishida, T. Ise, T. Yoshino, N. Mori, R. D. Rahimi, K. Sato, Y. Morita, K. Toyota, D. Shiomi, M. Kitagawa, H. Hara, P. Carl, P. Höfer, T. Takui, Angew. Chem., Int. Ed., 2012, 51, 9860.
D. Cho, K. C. Ko, J. Y. Lee, Int. J. Quant. Chem., 2016, 116, 578.
N. R. Khafizov, T. I. Madzhidov, O. N. Kadkin, R. Tamu-ra, I. S. Antipin, Int. J. Quant. Chem., 2016, 116, 1064.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fuku-da, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Mar-tin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salva-dor, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Far-kas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, D. J. Fox, Gaussian 09, Version B.01, 2010.
L. Noodleman, J. Chem. Phys., 1981, 74, 5737.
K. Yamaguchi, J. Mol. Struct. Theochem., 1983, 103, 101.
K. Yamaguchi, H. Fukui, T. Fueno, Chem. Lett., 1986, 15, 625.
P. Fluekiger, J. Weber, R. Chiarelli, A. Rassat, Y. Ellinger, Int. J. Quantum Chem., 1993, 45, 649.
V. N. Parmon, A. I. Kokorin, G. M. Zhidomirov, Stabiínye Biradikaly [Stable Biradicals], Nauka, Moscow, 1980, 240 pp. (in Russian).
V. A. Tran, A. I. Kokorin, G. Grampp, Appl. Magn. Reson., 2009, 35, 389.
A. I. Kokorin, V. N. Khrustalev, E. N. Golubeva, Appl. Magn. Reson., 2014, 45, 397.
A. Calder, A. R. Forrester, P. G. James, G. R. Luckhurst, J. Am. Chem. Soc., 1969, 91, 3724.
M. Dvolaitzky, R. Chiarelli, A. Rassat, Angew. Chem., Int. Ed. Engl., 1992, 31, 180.
F. Kanno, K. Inoue, N. Koga, H. Iwamura, J. Am. Chem. Soc., 1993, 115, 847.
J. Fujita, M. Tanaka, H. Suemune, N. Koga, R. Matsuna-ga, H. Iwamura, J. Am. Chem. Soc., 1996, 118, 9347.
A. Rajca, K. Lu, S. Rajca, C. R. Ross II, Chem. Commun., 1999, 1249.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2028—2034, November, 2017.
Rights and permissions
About this article
Cite this article
Khafizov, N.R., Madzhidov, T.I., Kadkin, O.N. et al. Exchange interaction mechanisms in 1,3,5,7-tetramethyl-2,6-diazaadamantane N,N’-dioxyl biradical. Russ Chem Bull 66, 2028–2034 (2017). https://doi.org/10.1007/s11172-017-1977-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-017-1977-3