Abstract
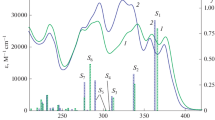
Photoinduced transformations of 2-styrylquinazolinones in solutions were studied using absorption and NMR spectroscopy methods. A possibility of control of the photochemical isomerization rate of quinazolinone 2-(hydroxyaryl)ethenyl derivatives by changing the pH of the medium was demonstrated. The bases and the solvent nature also affect the luminescence intensity of solutions of these compounds in the wavelength range of 550—650 nm. The differences in the steric organization of the ortho-hydroxystyryldiazinone system in crystals and in solutions related to the turn of the aryl group were found. Their influence on the competing processes of luminescence and photochemical transformation of the ethylene fragment were shown. The fact of reversible photo/thermal E-Z-isomerization was established for (E)-2-(2-hydroxystyryl)-3-phenylquinazolin-4(3H)-one.
Similar content being viewed by others
References
B. N. Krasovitskii, B. M. Bolotin, Organicheskiye Lyuminofory [Organic Luminophores], Khimiya, Moscow, 1984, 336 pp. (in Russian).
M. Irie, Chem. Rev., 2000, 100, 1685.
S. P. Gromov, M. V. Alfimov, Russ. Chem. Bull. (Engl. Transl.), 1997, 46, 638 [Izv. Akad. Nauk, Ser. Khim., 1997, 641].
G. N. Lipunova, E. V. Nosova, T. V. Trashakhova, V. N. Charushin, Russ. Chem. Rev., 2011, 80, 1115.
S. K. Lower, M. A. El-Sayed, Chem. Rev., 1966, 66, 199.
M. Dabiri, M. Baghbanzadeh, A. S. Delbari, J. Comb. Chem., 2008, 10, 700.
S. M. Bakalova, A. G. Santos, I. Timcheva, J. Kaneti, I. L. Filipova, G. M. Dobrikov, V. D. Dimitrov, J. Mol. Struct., 2004, 710, 229.
S. H. Mashraqui, S. S. Ghorpade, S. Tripathi, S. Britto, Tetrahedron Lett., 2012, 53, 765.
T. V. Trashakhova, E. V. Nosova, M. S. Valova, P. A. Slepukhin, G. N. Lipunova, V. N. Charushin, Russ. J. Org. Chem. (Engl. Transl.), 2011, 47, 753 [Zh. Org. Khim., 2011, 47, 748].
E. V. Nosova, T. V. Stupina, G. N. Lipunova, M. S. Valova, P. A. Slepukhin, V. N. Charushin, Int. J. Org. Chem., 2012. 2, 56.
C. A. Parker, Photoluminescence of Solutions, with Applications to Photochemistry and Analytical Chemistry, Elsevier, Amsterdam, 1968, p. 208.
G. N. Walker, J. Am. Chem. Soc., 1955, 77, 6698.
E. V. Nosova, T. V. Stupina, G. N. Lipunova, V. N. Charushin, Butlerovskiye Soobshch. [Butlerov Commun.] 2013, 33, 61 (in Russian).
E. L. Luk´yanova, Izmeritel´srednei moshchnosti i energii lazernogo izlucheniya IMO-2N [IMO-2N Meter of Average Power and Laser Irradiation Energy], Volgograd, 1980 (in Russian).
CrysAlis Pro, Vers. 171.33.66, Oxford Diffraction Ltd.
G. M. Sheldrick, Acta Crystallogr., Sect. A, 2008, 64, 112.
E. V. Tulyakova, O. A. Fedorova, Yu. V. Fedorov, A. V. Anisimov, J. Photochem. Photobiol. A: Chemistry, 2008, 200, 90.
F. A. Carey, R. J. Sundberg, Adv. Org. Chem., Part A, 2007, 12, 1073.
E. N. Marvell, G. C. T. A. Gosink, G. Zimmer, J. Am. Chem. Soc., 1966, 88, 619.
J. Rodriguez-Otero, J. Org. Chem., 1999, 64, 6842.
I. V. Kurdyukova, A. A. Ishchenko, N. A. Derevyanko, D. D. Mysyk, Chem. Heterocycl. Compd., 2013, 49, 281. [Khim. Geterotsikl. Soedin., 2013, 305].
M. M. Oliveira, M. A. Salvador, S. Delbaere, J. Berthet, G. Vermeersch, J. C. Micheau, P. J. Coelho, L. M. Carvalho, J. Photochem. Photobiol. A: Chemistry, 2008, 198, 242.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences Yu. N. Bubnov on the occasion of his 80th birthday and to the 60th anniversary of A. N. Nesmeyanov Institute of Organoelement Compounds of the Russian Academy of Sciences.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2467—2477, November, 2014.
Rights and permissions
About this article
Cite this article
Ovchinnikova, I.G., Kim, G.A., Matochkina, E.G. et al. Synthesis, photochemical and luminescent properties of (E)-2-(2-hydroxyarylethylene)-3-phenylquinazolin-4(3H)-ones. Russ Chem Bull 63, 2467–2477 (2014). https://doi.org/10.1007/s11172-014-0764-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-014-0764-7