Abstract
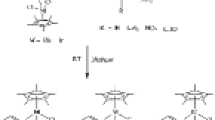
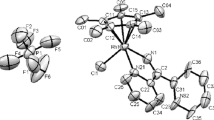
Reactions of [Cp*MCl2]2 (Cp* is η5-C5Me5, M = Ir, Rh) with carboranylamidinate ligand CabNH (1a) (CabNH is 1-R1-1,2-C2B10H11, R1 = (DippNH)C(=NDipp), Dipp is diisopropylphenyl) in the presence of the BuLi excess produced 16-electron half-sandwich iridium and rhodium complexes Cp*M(CabN-Dipp) (2a,b) (CabN-Dipp is (DippN)C(=NDipp)(closo-1,2-C2B10H10), M = Ir (a), Rh (b)). Unexpected neutral closo-metallacarboranes closo-[1-R2-3-(η5-Cp*)-3,1,2-MC2B9H10] (3a,b) (R2 = (NHDipp)C(=NDipp), M = Ir (a), Rh (b)) were prepared by the reaction of the deboronated zwitterionic ligand DcabNH (1b) (DcabNH is nido-7-[C(NHR3)2]+[7,8-C2B9H11]−, R3 = Dipp) with [Cp*MCl2]2. All compounds were fully characterized by elemental analysis, IR spectroscopy, mass spectrometry, and NMR spectroscopy. Structures of compounds 1a,b and 3a,b were confirmed by single crystal X-ray diffraction.
Similar content being viewed by others
References
V. I. Bregadze, Chem. Rev., 1992, 92, 209.
M. F. Hawthorne, Z.-P. Zheng, Acc. Chem. Res., 1997, 30, 267.
J. F. Valliant, K. J. Guenther, A. S. King, P. Morel, P. Schaffer, O. O. Sogbein, K. Stephenson, Coord. Chem. Rev., 2002, 232, 173.
G.-X. Jin, Coord. Chem. Rev., 2004, 248, 587.
A. D. Zdetsis, Nanoscale Res. Lett., 2011, 6, 362.
Z. Xie, Acc. Chem. Res., 2003, 36, 1.
A. F. Armstrong, J. F. Valliant, Dalton Trans., 2007, 4240.
A. M. Cioran, A. D. Musteti, F. Teixidor, Z. Krpetic, I. A. Prior, Q. He, C. J. Kiely, M. Brust, C. Viñas, J. Am. Chem. Soc., 2012, 134, 212.
L. Deng, H.-S. Chan, Z. Xie, J. Am. Chem. Soc., 2005, 127, 13774.
J. D. Lee, Y. J. Lee, K. C. Son, M. Chong, J. Ko, S. O. Kang, Organometallics, 2007, 26, 3374.
A. I. Stoica, C. Viñas, F. Teixidor, Chem. Commun., 2008, 6492.
R. J. Blanch, M. Williams, G. D. Fallon, M. G. Gardiner, R. Kaddour, C. L. Raston, Angew. Chem., Int. Ed., 1997, 36, 404.
B. Grüner, J. Plešek, J. Báčca, I. Císařová, J. F. Dozol, H. Rouquette, C. Viñas, P. Selucky, J. Rais, New J. Chem., 2002, 26, 1519.
D. Hablot, R. Ziessel, M. A. H. Alamiry, E. Bahraidah, A. Harriman, Chem. Sci., 2013, 4, 444.
E. Hao, M. G. H. Vicente, Chem. Commun., 2005, 1306.
J.-Q. Wang, C.-X. Ren, L.-H. Weng, G.-X. Jin, Chem. Commun., 2006, 162.
A. H. Soloway, W. Tjarks, B. A. Barnum, F. G. Rong, R. F. Barth, I. M. Codogni, J. G. Wilson, Chem. Rev., 1998, 98, 1515.
S. N. Mukhin, K. Z. Kabytaev, G. G. Zhigareva, I. V. Glukhov, Z. A. Starikova, V. I. Bregadze, I. P. Beletskaya, Organometallics, 2008, 27, 5937.
X. Wang, G.-X. Jin, Chem. — Eur. J., 2005, 11, 5758.
C. Viñas, A. Laromaine, F. Teixidor, H. Horáková, A. Langauf, R. Vespalec, I. Mata, E. Molins, Dalton Trans., 2007, 3369.
M. F. Hawthorne, Acc. Chem. Res., 1968, 1, 281.
G. B. Dunks, M. F. Hawthorne, Acc. Chem. Res., 1973, 6, 124.
M. F. Hawthorne, J. I. Zink, J. M. Skelton, M. J. Bayer, C. Liu, E. Livshits, R. Baer, D. Neuhauser, Science, 2004, 303, 1849.
P. Matejicek, P. Cigler, K. Prochazka, V. Kral, Langmuir, 2006, 22, 575.
M. G. H. Vicente, Curr. Med. Chem.: Anti-Cancer Agents, 2001, 1, 175.
R. Gahbauer, N. Gupta, T. Blue, J. Goodman, R. Barth, J. Grecula, A. H. Soloway, W. Sauerwein, A. Wambersie, Cancer Res., 1998, 58, 183.
D. J. Crowther, D. C. Swenson, R. F. Jordan, J. Am. Chem. Soc., 1995, 117, 10403.
N. S. Hosmane, J. A. Maguire, Eur. J. Inorg. Chem., 2003, 3989.
Z. Xie, Coord. Chem. Rev., 2006, 250, 259.
N. S. Hosmane, Z. Y. Huai, J. A. Maguire, W. Kaim, M. Takagaki, J. Organomet. Chem., 2009, 694, 1690.
P. Drose, C. G. Hrib, F. T. Edelmann, J. Am. Chem. Soc., 2010, 132, 15540.
Z.-J. Yao, G. Su, G.-X. Jin, Chem. — Eur. J., 2011, 17, 13298.
Z.-J. Yao, G.-X. Jin, Organometallics, 2012, 31, 1767.
Z.-J. Yao, B. Xu, G. Su, G.-X. Jin, J. Organomet. Chem., 2012, 721, 31.
A. Williams, I. T. Ibrahim, Chem. Rev., 1981, 81, 589.
A. L. Chen, K. L. Wei, R.-J. Jeng, J.-J. Lin, S. A. Dai, Macromolecules, 2011, 44, 46.
Z.-J. Yao, G.-X. Jin, Coord. Chem. Rev., 2013, 257, 2522.
Z.-J. Yao, Y.-J. Lin, G.-X. Jin, Chem. Eur. J., 2013, 19, 2611.
F. A. Gomez, S. E. Johnson, C. B. Knobler, M. F. Hawthorne, Inorg. Chem., 1992, 3, 3558.
I. T. Chizhevsky, P. V. Petrovskii, P. V. Sorokin, V. I. Bregadze, F. M. Dolgushin, A. I. Yanovsky, Y. T. Struchkov, A. Demonceau, A. F. Noels, Organometallics, 1996, 15, 2619.
I. V. Pisareva, I. T. Chizhevsky, P. V. Petrovskii, V. I. Bregadze, F. M. Dolgushin, A. I. Yanovsky, Organometallics, 1997, 16, 5598.
M. Green, J. L. Spencer, F. G. A. Stone, J. Chem. Soc., Dalton Trans., 1975, 179.
J. Plešek, S. Heřmánek, Inorg. Synth., 1984, 22, 231.
J. Yoo, J.-W. Hwang, Y. Do, Inorg. Chem., 2001, 40, 3445.
R. L. Thomas, A. J. Welch, J. Chem. Soc., Dalton Trans., 1997, 631.
U. Grädler, A. S. Weller, A. J. Welch, D. Reed, J. Chem. Soc., Dalton Trans., 1996, 335.
A. S. Weller, A. J. Welch, Inorg. Chem., 1996, 35, 4538.
Z. G. Lewis, A. J. Welch, J. Organomet. Chem., 1992, 430, 45.
J. Cowie, B. D. Reid, J. M. S. Watmough, A. J. Welch, J. Organomet. Chem., 1994, 481, 283.
R. M. Garrioch, G. M. Rosair, A. J. Welch, J. Organomet. Chem., 2000, 614, 153.
J. H. Kim, M. Lamrani, J. W. Hwang, Y. Do, Chem. Commun., 1997, 1761.
J. Llop, C. Viñas, F. Teixidor, L. Victori, R. Kivekäs, R. Sillanpää, Orgnometallics, 2001, 20, 4024.
J. M. Oliva, N. L. Allan, P. R. Schleyer, C. Viñas, F. Teixidor, J. Am. Chem. Soc., 2005, 127, 13538.
C. Viñas, J. Pedrajas, J. Bertran, F. Teixidor, R. Kivekäs, R. Sillanpää, Inorg. Chem., 1997, 36, 2482.
L. S. Alekseev, A. V. Safronov, F. M. Dolgushin, A. A. Korlyukov, I. A. Godovikov, I. T. Chizhevsky, J. Organomet. Chem., 2009, 694, 1727.
G. M. Sheldrick, SHELXL-97, Program for the Refinement of Crystal Structures, Universität Göttingen, Göttingen (Germany), 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
According to the materials of the International Conference “Organometallic and Coordination Chemistry: Fundamentals and Applied Aspects” (Nizhniy Novgorod, September 1–7, 2013).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 4, pp. 0963–0969, April, 2014.
Rights and permissions
About this article
Cite this article
Xu, B., Yao, Z.J. & Jin, G.X. Reactivity of half-sandwich metal complexes with sterically encumbered N,N′-bis(2,6-diisopropylphenyl) group-substituted carboranylamidinate ligands. Russ Chem Bull 63, 963–969 (2014). https://doi.org/10.1007/s11172-014-0534-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-014-0534-6