Abstract
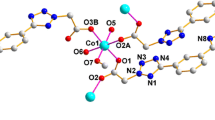
The reactions of three-mercury anticrown (o-C6F4Hg)3 (1) with acetoacetic ester (AE), malonic ester (ME), and malonodinitrile (MN) afford 1: 1 complexes {[(o-C6F4Hg)3](AE)} (3), {[(o-C6F4Hg)3](ME)} (4), and {[(o-C6F4Hg)3](MN)} (5). The structures of complexes 3–5 were determined by X-ray diffraction analysis. Complex 3 has a discrete structure in the solid state, whereas complexes 4 and 5 form in the crystal extended stacks representing polydecker sandwiches with alternating molecules of 1 and ME or MN. According to the X-ray diffraction and IR spectral data, the molecule of AE in complex 3 is in the keto form.
Similar content being viewed by others
References
V. B. Shur, I. A. Tikhonova, in Encyclopedia of Supramolecular Chemistry, Eds J. L. Atwood, J. W. Steed, Marcel Dekker, Inc., New York, 2004, p. 68.
T. W. Wedge, M. F. Hawthorne, Coord. Chem. Rev., 2003, 240, 111.
V. B. Shur, I. A. Tikhonova, Russ. Chem. Bull. (Int. Ed.), 2003, 52, 2539 [Izv. Akad. Nauk, Ser. Khim., 2003, 2401].
M. R. Haneline, R. E. Taylor, F. P. Gabbaï, Chem. Eur. J., 2003, 21, 5188.
T. J. Taylor, C. N. Burres, F. P. Gabbaï, Organometallics, 2007, 26, 5252.
J. D. Wuest, Acc. Chem. Res., 1999, 32, 81.
M. F. Hawthorne, Z. Zheng, Acc. Chem. Res., 1997, 30, 267.
I. A. Tikhonova, K. I. Tugashov, F. M. Dolgushin, A. A. Yakovenko, P. V. Petrovskii, G. G. Furin, A. P. Zaraisky, V. B. Shur, J. Organomet. Chem., 2007, 692, 953.
I. A. Tikhonova, K. I. Tugashov, F. M. Dolgushin, P. V. Petrovskii, V. B. Shur, Organometallics, 2007, 26, 5193.
V. B. Elbjeirami, C. N. Burress, F. P. Gabbaï, M. A. Omary, J. Phys. Chem. C, 2007, 111, 9522.
T. J. Taylor, O. Elbjeirami, C. N. Burress, M. Tsunoda, M. I. Bodine, M. A. Omary, F. P. Gabba-, J. Inorg. Organomet. Polym., 2008, 18, 175.
V. N. Tsupreva, O. A. Filippov, F. M. Dolgushin, K. I. Tugashov, A. I. Krylova, V. I. Bragin, I. A. Tikhonova, V. B. Shur, L. M. Epshtein, E. S. Shubina, Russ. Chem. Bull. (Int. Ed.), 2008, 57, 2540 [Izv. Akad. Nauk, Ser. Khim., 2008, 2489].
I. A. Tikhonova, K. I. Tugashov, F. M. Dolgushin, A. A. Korlyukov, P. V. Petrovskii, Z. S. Klemenkova, V. B. Shur, J. Organomet. Chem., 2009, 694, 2604.
I. A. Tikhonova, D. A. Gribanyov, K. I. Tugashov, F. M. Dolgushin, A. F. Smolýakov, A. S. Peregudov, Z. S. Klemenkova, V. B. Shur, Organometallics, 2009, 28, 6567.
A. S. Filatov, E. A. Jackson, L. T. Scott, M. A. Petrukhina, Angew. Chem., Int. Ed., 2009, 48, 8473.
I. A. Tikhonova, D. A. Gribanyov, K. I. Tugashov, F. M. Dolgushin, A. S. Peregudov, D. Yu. Antonov, V. I. Rosenberg, V. B. Shur, J. Organomet. Chem., 2010, 695, 1949.
I. A. Tikhonova, D. A. Gribanyov, K. I. Tugashov, F. M. Dolgushin, A. F. Smolýakov, A. S. Peregudov, Z. S. Klemenkova, V. B. Shur, ARKIVOC, 2011, 172.
A. S. Filatov, A. K. Greene, E. A. Jackson, L. T. Scott, M. A. Petrukhina, J. Organomet. Chem., 2011, 696, 2877.
P. Sartory, A. Golloch, Chem. Ber., 1968, 101, 2004.
M. C. Ball, D. S. Brown, A. G. Massey, D. A. Wickens, J. Organomet. Chem., 1981, 206, 265.
A. P. Zaraisky, O. I. Kachurin, L. I. Velichko, I. A. Tikhonova, G. G. Furin, V. B. Shur, J. Molec. Cat. A: Chemical, 2005, 231, 103.
I. A. Tikhonova, F. M. Dolgushin, A. A. Yakovenko, K. I. Tugashov, P. V. Petrovskii, G. G. Furin, V. B. Shur, Organometallics, 2005, 24, 3395.
Z. Yan, I. A. Tikhonova, Z. Zhou, V. B. Shur, Y. Wu, Anal. Lett., 2005, 38, 377.
I. A. Tikhonova, A. A. Yakovenko, K. I. Tugashov, F. M. Dolgushin, V. V. Novikov, M. Yu. Antipin, V. B. Shur, Organometallics, 2006, 25, 6155.
O. A. Reutov, A. L. Kurz, K. P. Butin, Organicheskaya khimiya [Organic Chemistry], Part 3, Binom, Moscow, 2004, 93.
A. J. Canty, G. B. Deacon, Inorg. Chim. Acta, 1980, 45, L 255.
P. Pyykkö, M. Straka, Phys. Chem. Chem. Phys., 2000, 2, 2489.
S. S. Batsanov, Zh. Neorg. Khim., 1991, 36, 3015 [Russ. J. Inorg. Chem. (Engl. Transl.), 1991, 36].
S. C. Nyborg, C. H. Faerman, Acta Crystallogr., Sect. B, 1985, 41, 274.
F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen, R. Taylor, J. Chem. Soc., Perkin Trans. 2, 1987, S1.
I. A. Tikhonova, F. M. Dolgushin, K. I. Tugashov, G. G. Furin, P. V. Petrovskii, V. B. Shur, Russ. Chem. Bull. (Int. Ed.), 2001, 50, 1673 [Izv. Akad. Nauk, Ser. Khim., 2001, 1595].
J. B. King, M. R. Haneline, M. Tsunoda, F. P. Gabbaï, J. Am. Chem. Soc., 2002, 124, 9350.
R. A. Walton. Quart. Revs., 1965, 19, 126.
K. F. Purcell, R. S. Drago, J. Am. Chem. Soc., 1966, 88, 919.
C. Reichardt, Solvents and Solvent Effects in Organic Chemistry, Weinheim, 1988.
B. Cordero, V. Gómez, A. E. Platero-Prats, M. Revés, J. Echeverría, E. Cremades, F. Barragán, S. Alvarez, Dalton Trans., 2008, 2832.
X. Yang, Z. Zheng, C. B. Knobler, M. F. Hawthorne, J. Am. Chem. Soc., 1993, 115, 193.
A. A. Zinn, Z. Zheng, C. B. Knobler, M. F. Hawthorne, J. Am. Chem. Soc., 1996, 118, 70.
I. A. Tikhonova, F. M. Dolgushin, A. I. Yanovsky, Z. A. Starikova, P. V. Petrovskii, G. G. Furin, V. B. Shur, J. Organomet. Chem., 2000, 613, 60.
Yu. K. Grishin, Yu. A. Strelenko, L. A. Margulis, Yu. A. Ustynyuk, L. S. Golovchenko, A. S. Peregudov, D. N. Kravtsov, Dokl. Akad. Nauk SSSR, 1979, 249, 892 [Dokl. Chem. (Engl. Transl.), 1979].
SMART V5.051 and SAINT V5.00, Area Detector Control and Integration Software, Bruker AXS Inc., Madison, WI-53719, USA, 1998.
G. M. Sheldrick, SADABS, Bruker AXS Inc., Madison, WI-53719, USA, 1997.
G. M. Sheldrick, Acta Cryst., 2008, A64, 112.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the Academician of the Russian Academy of Sciences I. P. Beletskaya on the occasion of her birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 3, pp. 0710–0715, March, 2013.
Rights and permissions
About this article
Cite this article
Tikhonova, I.A., Yakovenko, A.A., Tugashov, K.I. et al. Coordination chemistry of anticrowns. Complexation of cyclic trimeric perfluoro-o-phenylenemercury (o-C6F4Hg)3 with compounds containing an active methylene group. Russ Chem Bull 62, 710–715 (2013). https://doi.org/10.1007/s11172-013-0097-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-013-0097-y