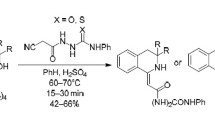
Abstract
Cyclothiomethylation of phenyl hydrazine with CH2O and H2S in a ratio of 1: 3: 2 in an acidic medium (HCl) afforded previously unknown 3-phenyl-1,3,4-thiadiazolidine (35% yield) and N-phenyl(perhydro-1,3,5-dithiazin-5-yl)amine (35% yield). The analogous reaction in an alkaline medium (BuONa) produced N-phenyl(perhydro-1,3-thiazetidin-3-yl)amine (22% yield). The reaction of 1,2-diphenyl hydrazine with CH2O and H2S in an alkaline medium gave 1,2,4,5-tetraphenylhexahydro-1,2,4,5-tetrazine and previously unknown 3,4-diphenyl-1,3,4-thiadiazolidine and 5,6-diphenyltetrahydro-1,3,5,6-dithiadiazepine in 39 and 22% yields, respectively. Cyclothiomethylation of benzyl hydrazine afforded previously unknown bis[(6-benzyl-4,2,6-thiadiazolidin-2-yl)methyl] sulfide (60% yield) and N-benzyl(perhydro-1,3,5-dithiazin-5-yl)amine (19% yield). The reaction of tosyl hydrazine produced 3-[(p-tolyl)sulfonyl]-1,3,4-thiadiazolidine, N-(perhydro-1,3,5-dithiazin-5-yl)-p-tolylsulfonamide, and 3,7-bis(p-tolylsulfonylamino)-1,5-dithia-3,7-diazacyclooctane in 21, 38, and 41% yields, respectively.
Similar content being viewed by others
References
S. R. Khafizova, V. R. Akhmetova, T. V. Tyumkina, L. M. Khalilov, R. V. Kunakova, and U. M. Dzhemilev, Izv. Akad. Nauk, Ser. Khim., 2004, 1652 [Russ. Chem. Bull., Int. Ed., 2004, 53, 1717].
M. R. Stillings, A. P. Welbourn, and D. J. Walter, Med. Chem., 1986, 29, 2280.
M. Kidwai, N. Negi, and S. R. Chaudhary, Acta Pharma, 1995, 45, 511.
US Pat. 4,246,126 (1981); Chem. Abstrs, 1981, 94, 142505h.
Heterocyclic Compounds, Ed. R. C. Elderfield, Wiley, New York, 1961.
S. P. Gromov, S. N. Dmitrieva, A. I. Vedernikov, and M. V. Churakova, Izv. Akad. Nauk, Ser. Khim., 2004, 1362 [Russ. Chem. Bull., Int. Ed., 2004, 53, 1417].
R. V. Kunakova, S. R. Khafizova, Yu. S. Dal’nova, R. S. Aleev, L. M. Khalilov, and U. M. Dzhemilev, Neftekhimiya, 2002, 42, 382 [Russ. Petroleum Chem., 2002, 42, 347 (Engl. Transl.)].
S. R. Khafizova, V. R. Akhmetova, R. V. Kunakova, and U. M. Dzhemilev, Izv. Akad. Nauk, Ser. Khim., 2003, 1722 [Russ. Chem. Bull., Int. Ed., 2003, 52, 1817].
S. R. Khafizova, V. R. Akhmetova, L. F. Korzhova, T. V. Khakimova, G. R. Nadyrgulova, R. V. Kunakova, E. A. Kruglov, and U. M. Dzhemilev, Izv. Akad. Nauk, Ser. Khim., 2005, 423 [Russ. Chem. Bull., Int. Ed., 2005, 54, 432].
V. R. Akhmetova, G. R. Nadyrgulova, S. R. Khafizova, R. R. Khairullina, E. A. Paramonov, R. V. Kunakova, and U. M. Dzhemilev, Zh. Org. Khim., 2006, 42, 151 [Russ. J. Org. Chem., 2006, 42 (Engl. Transl.)].
A. P. Grekov and V. Ya. Veselov, Fizicheskaya khimiya gidrazina [Physical Chemistry of Hydrazine], Naukova Dumka, Kiev, 1979, 264 pp. (in Russian).
Comprehensive Organic Chemistry, Eds S. D. Barton and W. D. Ollis, Pergamon Press, Oxford-New York-Toronto-Sydney-Paris-Frankfurt, 1979.
R. S. Aleev, Yu. S. Dal’nova, Yu. I. Popova, R. M. Masagutov, and S. R. Rafikov, Dokl. Akad. Nauk SSSR, 1988, 303, 873 [Dokl. Chem., 1988 (Engl. Transl.)].
B. Rassow, J. Prakt. Chem., 1901, 64, 136.
Weygand-Hilgetag, Organisch-Chemische Experimentierkunst, Leipzig, 1964.
R. G. Kostyanovskii, P. Rademakher, Yu. I. El’natanov, G. K. Kadorkina, G. A. Nikiforov, I. I. Chervin, S. V. Usachev, and V. R. Kostyanovskii, Izv. Akad. Nauk, Ser. Khim., 1997, 1346 [Russ. Chem. Bull., 1997, 46, 1291 (Engl. Transl.)].
J. E. Anderson and J. M. Lehn, J. Am. Chem. Soc., 1967, 89, 81.
G. V. Shustov, A. Yu. Shibaev, Yu. V. Puzanov, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., 1988, 1869 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1988, 37, 1671 (Engl. Transl.)].
V. J. Baker, A. R. Katritzky, and J.-P. Majoral, J. Chem. Soc., Perkin Trans. 2, 1975, 1191.
H. Günter, NMR Spectroscopy, An Introduction, Wiley, Chichester, 1980.
B. V. Ioffe and V. S. Stopskii, Dokl. Akad. Nauk SSSR, 1967, 175, 1064 [Dokl. Chem., 1967 (Engl. Transl.)].
Houben-Weyl, Methoden der organischen Chemie, Verlag Georg Tieme, Stuttgart, 1953.
Analiz nefti i nefteproduktov [Analysis of Petroleum and Petroleum Products], Ed. B. M. Rybak, Gostoptekhizdat, Moscow, 1962, 5, 887 pp. (in Russian).
G. Cadenas-Pliego, M. J. Rosales-Hoz, R. Contreras, and A. Flores-Parra, Tetrahedron Asymmetry, 1994, 5, 633.
R. M. Kuznetsov, A. S. Balueva, I. A. Litvinov, A. T. Gubaidullin, G. N. Nikonov, A. A. Karasik, and O. G. Sinyashin, Izv. Akad. Nauk, Ser. Khim., 2002, 142 [Russ. Chem. Bull., Int. Ed., 2002, 51, 151].
B. A. Arbuzov, O. A. Erastov, G. N. Nikonov, I. A. Litvinov, D. S. Yufit, and Yu. T. Struchkov, Dokl. Akad. Nauk SSSR, 1981, 257, 127 [Dokl. Chem., 1981 (Engl. Transl.)].
N. D. Cheronis, Micro and Semimicro Methods, Intersci. Publ., New York, 1954.
Spravochnik khimika [Chemist’s Handbook], Ed. B. P. Nikol’skii, Khimiya, Moscow-Leningrad, 1964, 3, 1008 pp. (in Russian).
G. M. Sheldrick, SHELXTL PLUS, PC Version, System of Computer Programs for the Determination of Crystal Structure from X-ray Diffraction Data, Rev. 502, Siemens Analytical X-Ray Instruments, Inc., Germany, 1994.
Author information
Authors and Affiliations
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 1758–1767, October, 2006.
Rights and permissions
About this article
Cite this article
Akhmetova, V.R., Nadyrgulova, G.R., Tyumkina, T.V. et al. Cyclothiomethylation of aryl hydrazines with formaldehyde and hydrogen sulfide. Russ Chem Bull 55, 1824–1834 (2006). https://doi.org/10.1007/s11172-006-0493-7
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11172-006-0493-7