Abstract
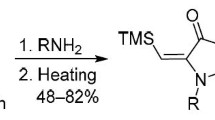
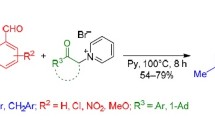
Substituted 5-aryl-3-ethylidene-3H-pyrrol-2-ones were synthesized by the reaction of the corresponding 4-aryl-4-oxobutanoic acids with ketones in the presence of aminating agents. The conditions of this reaction were developed with the use of both the conventional condensation technique and microwave activation. The structures of the reaction products were con-firmed by elemental analysis, IR spectroscopy, and 1H NMR spectroscopy.
Similar content being viewed by others
References
M. M. Abd-Alla and A. Essawy, Ind. J. Chem., 1977, 15, 523.
M. M. Abd-Alla, E. A. Soliman, and M. W. Osman, Rev. Roum. Chim, 1980, 25, 415.
A. Yu. Yegorova, Izv. Akad. Nauk, Ser. Khim., 2002, 172 [Russ. Chem. Bull., Int. Ed., 2002, 51, 183].
El-Abbady, M. A. Omara, and N. G. Kandil, Rev. Roum. Chim., 1974, 19, 79.
Y. Bocchi and G. P. Gardini, Tetrahedron Lett., 1971, 211.
M. M. Abd-Alla, E. A. Soliman, and A. A. Hamed, Egypt. J. Chem., 1980, 23, 93.
A. Yu. Yegorova, V. A. Sedavkina, and Z. Yu. Timofeeva, Khim. Geterotsikl. Soedin., 2001, 602 [Chem. Heterocycl. Compd., 2001 (Engl. Transl.)].
B. V. Timokhin, V. A. Baranskii, and G. D. Eliseeva, Usp. Khim., 1999, 68, 80 [Russ. Chem. Rev., 1999, 68 (Engl. Transl.)].
N. N. Romanova, P. V. Kudan, and A. G. Gravis, Khim. Geterotsikl. Soedin., 2000, 1308 [Chem. Heterocycl. Compd., 2000 (Engl. Transl.)].
L. F. Tietze and T. Eicher, Reaktionen und Synthesen im organisch-chemischen Praktikum und Forschungslaboratorium, Georg Thieme Verlag, Stuttgart-New York, 1991.
Author information
Authors and Affiliations
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 4, pp. 693–696, April, 2006.
Rights and permissions
About this article
Cite this article
Yegorova, A.Y., Kamneva, I.E. & Shtykova, L.S. Effective synthesis of 5-aryl-3-ethylidene-3H-pyrrol-2-ones. Russ Chem Bull 55, 718–721 (2006). https://doi.org/10.1007/s11172-006-0319-7
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11172-006-0319-7