Abstract
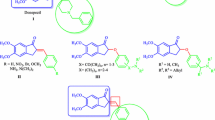
The analog of the alkaloid flustrabromine containing no Br atom in position 6 was obtained in three steps. The key step involves reductive prenylation of 3-R-indole with triprenylborane.
Similar content being viewed by others
References
Yu. N. Bubnov, E. A. Shagova, S. V. Evchenko, A. V. Ignatenko, and I. D. Gridnev, Izv. Akad. Nauk SSSR, Ser. Khim., 1991, 2644 [Bull. Russ. Acad. Sci., Div. Chem. Sci., 1991, 40, 2315 (Engl. Transl.)].
Yu. N. Bubnov, Pure Appl. Chem., 1994, 66, 235; Yu. N. Bubnov, Izv. Akad. Nauk, Ser. Khim., 1995, 1203 [Russ. Chem. Bull., 1995, 44, 1156 (Engl. Transl.)]; Yu. N. Bubnov, in Advances in Boron Chemistry, Ed. W. Siebert, The Royal Society of Chemistry, 1997, p. 123; Yu. N. Bubnov, E. V. Klimkina, I. V. Zhun, F. V. Pastukhov, and I. V. Yampolski, in Contemporary Boron Chemistry, Eds. M. Davidson, A. K. Hughes, T. B. Marder, and K. Wade, The Royal Society of Chemistry, Thomas Graham House, Cambridge, 2000, p. 446.
Yu. N. Bubnov, E. A. Shagova, S. V. Evchenko, and A. V. Ignatenko, Izv. Akad. Nauk SSSR, Ser. Khim., 1994, 693 [Russ. Chem. Bull., 1994, 43, 645 (Engl. Transl.)]; Yu. N. Bubnov, E. V. Klimkina, A. V. Ignatenko, and I. D. Gridnev, Tetrahedron Lett., 1996, 37, 1317; Yu. N. Bubnov, E. V. Klimkina, A. V. Ignatenko, and I. D. Gridnev, Tetrahedron Lett., 1997, 38, 4631.
Yu. N. Bubnov, E. E. Demina, A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1997, 627 [Russ. Chem. Bull., 1997, 46, 606 (Engl. Transl.)]; Yu. N. Bubnov, E. E. Demina, M. O. Dekaprilevich, and Yu. T. Struchkov, Izv. Akad. Nauk, Ser. Khim., 1997, 1413 [Russ. Chem. Bull., 1997, 46, 1356 (Engl. Transl.)].
Yu. N. Bubnov, E. V. Klimkina, L. I. Lavrinovich, A. Yu. Zykov, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1999, 1718 [Russ. Chem. Bull., 1997, 48, 1696 (Engl. Transl.)]; Yu. N. Bubnov, A. Yu. Zykov, L. I. Lavrinovich, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1993, 1329 [Russ. Chem. Bull., 1993, 42, 1271 (Engl. Transl.)].
Yu. N. Bubnov, L. I. Lavrinovich, A. Yu. Zykov, E. V. Klimkina, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1993, 1327 [Russ. Chem. Bull., 1993, 42, 1269 (Engl. Transl.)].
Yu. N. Bubnov, M. E. Gurskii, and T. V. Potapova, Izv. Akad. Nauk, Ser. Khim., 1996, 2807 [Russ. Chem. Bull., 1996, 45, 2665 (Engl. Transl.)].
Yu. N. Bubnov, S. V. Evchenko, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1992, 2815 [Bull. Russ. Acad. Sci., Div. Chem. Sci., 1992, 41, 2239 (Engl. Transl.)].
Yu. N. Bubnov, S. V. Evchenko, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1993, 1325 [Russ. Chem. Bull., 1993, 42, 1268 (Engl. Transl.)]; Yu. N. Bubnov, E. V. Klimkina, and A. V. Ignatenko, Izv. Akad. Nauk, Ser. Khim., 1998, 1206 [Russ. Chem. Bull., 1998, 47, 1175 (Engl. Transl.)].
Yu. N. Bubnov, I. V. Zhun, E. V. Klimkina, A. V. Ignatenko, and Z. A. Starikova, Eur. J. Org. Chem., 2000, 3323.
P. Wulf, J. S. Carle, and C. Christophersen, J. Chem. Soc. Perkin Trans. 1, 1981, 2895; C. Christophersen, Acta Chem. Scand., 1981, B39, 517.
A. Alemany, E. Fernandez Alvarez, O. Nieto Lopez, and M. E. Rubio Herraez, Bull. Soc. Chim. Fr., 1974, 12, 2883.
A. P. Terent’ev, M. N. Preobrazhenskaya, A. S. Bobkov, and G. M. Sorokina, Zh. Org. Khim., 1959, 29, 2541 [J. Gen. Chem. USSR, 1959, 29 (Engl. Transl.)].
J. M. Schkeryants, J. C. G. Woo, P. P. Siliphaivanh, K. M. Depew, and S. J. Danishefsky, J. Am. Chem. Soc., 1999, 121, 11964.
B. M. Mikhailov, V. N. Smirnov, and O. D. Ryazanova, Dokl. Akad. Nauk, 1972, 204, 612 [Dokl. Chem., 1972, 204 (Engl. Transl.)].
Author information
Authors and Affiliations
Additional information
__________
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 2126–2128, October, 2004.
Rights and permissions
About this article
Cite this article
Zhun, I.V., Ignatenko, A.V. Use of allylic boranes in organic syntheses. Synthesis of an analog of the alkaloid flustrabromine. Russ Chem Bull 53, 2221–2223 (2004). https://doi.org/10.1007/s11172-005-0103-0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11172-005-0103-0