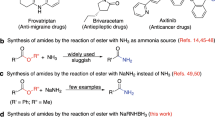
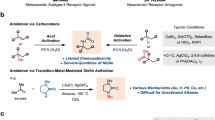
Abstract
Aryl amides and amide derivatives are important functionality in drug and drug discovery, as many drugs have it as an integral part. However, the synthesis of amide requires the condensation or coupling of amines which is a tedious or costly approach. Herein we disclosed two easy and convenient methods for directly synthesizing aryl amide using aryl nitrile in a short time with good to excellent yield via base-driven partial hydrolysis.



Similar content being viewed by others
Data availability
Electronic supplementary information (ESI) available for all spectroscopic data is available free of charge via the Internet at https://www.springer.com/journal/11164.
References
M. Ahmadi, L. Moradi, M. Sadeghzadeh, Res. Chem. Intermed. 44, 7873 (2018)
B.R. Penfold, J.C.B. White, Acta Crystallogr. 12, 130 (1959)
S. Aitipamula, P.S. Chow, R.B.H. Tan, Cryst. Growth Des. 10, 2229 (2010)
J. Angst, M. Stabl, Psychopharmacology 106, S109 (1992)
H. Kale, R. Fass, Pocket Guide to Gastrointestinal Drugs, vol. 1 (2014)
C.S. Conner, Drug Intell. Clin. Pharm. 18, 137 (1984)
T. Swamy, P. Raviteja, B.V. Subba Reddy, V. Ravinder, ChemistrySelect 2, 7612 (2017)
R.R. Naredla, D.A. Klumpp, Tetrahedron Lett. 53, 4779 (2012)
D. G. S. Andayani, S. R. Anggraeni, E. Liviawaty, R. M. Chrisentia, Y. Srikandace, in IOP Conference Series: Earth and Environmental Science (IOP Publishing, 2018), p. 12005
L. Lu, Z. Qi, T. Wang, X. Zhang, K. Zhang, K. Wang, Y. Cheng, Y. Xiao, Z. Li, S. Jiang, A.C.S. Med, Chem. Lett. 13, 586 (2022)
Z. Liu, A. Jiang, Y. Wang, P. Xu, Q. Zhang, Y. Wang, S. He, N. Wang, H. Jin, B. Zhang, Bioorg. Med. Chem. Lett. 67, 128759 (2022)
J. Rouanet, M. Quintana, P. Auzeloux, F. Cachin, F. Degoul, Pharmacol. Ther. 224, 107829 (2021)
E. Valeur, M. Bradley, Chem. Soc. Rev. 38, 606 (2009)
K. Aftab, K. Aslam, S. Kousar, M.J.U.H. Nadeem, Pak. J. Pharm. Sci. 29, 423 (2016)
P.H. Williams, J. Am. Chem. Soc. 26, 659 (1961)
J. Kuwabara, Y. Sawada, M. Yoshimatsu, Synlett 29, 2061 (2018)
H. Chen, W. Dai, Y. Chen, Q. Xu, J. Chen, L. Yu, Y. Zhao, M. Ye, Y. Pan, Green Chem. 16, 2136 (2014)
B. Yu, X.J. Shi, J.L. Ren, X.N. Sun, P.P. Qi, Y. Fang, X.W. Ye, M.M. Wang, J.W. Wang, E. Zhang, D.Q. Yu, H.M. Liu, Eur. J. Med. Chem. 66, 171 (2013)
M.I. Moraes, C. Iglesias, I.S. Teixeira, H.M.S. Milagre, S.R. Giordano, C.D.F. Milagre, Results Chem. 5, 100760 (2023)
A. Mousavi-Mashhadi, S.A. Shiri, J. Iran Chem. Soc. 19, 4523 (2022)
J.N. Moorthy, N. Singhal, J. Org. Chem. 70, 1926 (2005)
D.D. Sanz Sharley, J.M.J. Williams, Tetrahedron Lett. 58, 4090 (2017)
S. Yadav, R. Gupta, Inorg. Chem. 61, 15463 (2022)
B. Guo, J.G. De Vries, Chem. Sci. 10, 10647 (2019)
X. Ma, D. Liu, X. Wan, J. Zhao, Synth. Commun. 53, 503 (2023)
S.S. Choudhury, S. Mahapatra, H.S. Biswal, Green Chem. 24, 4981 (2022)
Funding
G.L.K. is grateful to SERB- Department of Science & Technology (DST) Govt of India for research funding (Grant no. EEQ/2021/000114). S.K. and R.A. acknowledge the Department of Pharmaceuticals, Ministry of Chemicals, and Fertilizers for the financial assistance of the fellowship.
Author information
Authors and Affiliations
Contributions
S.K. carried out the research work. R.A. helped in the collection of spectroscopic data. G.L.K. hypothesized the concept and carried out the writing of the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, S., Ambatwar, R. & Khatik, G.L. Easy and efficient direct conversion of aryl nitrile into aryl amide. Res Chem Intermed 49, 3951–3962 (2023). https://doi.org/10.1007/s11164-023-05062-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05062-y