Abstract
Fe3O4@L-Arginine and Fe3O4@L-Histidine nanoparticles (NPs) are synthesized and explored as catalysts for the sequential Knoevenagel condensation and Michael addition reactions (KMS). The reaction parameters like the amount of catalyst, temperature, and solvent systems are optimized for both of the catalysts for the Knoevenagel condensation reaction. It was observed that the developed catalyst systems work well under solvent-free conditions at 80 °C. Excellent to high yield was achieved in the corresponding Knoevenagel adducts (up to 97%), in turn, delivered good yield in Michael products (up to 70%). Surprisingly, Fe3O4@L-Hist NPs did not show any catalytic activity for the sequential Michael addition. The probable reasons for the high activities of Fe3O4@L-Arg NPs for KMS have been discussed. The catalyst was also recovered and reused for 5 cycles with equal performance in terms of activity and selectivity. The optimized protocol for sequential Knoevenagel condensation and Michael addition reaction can result in high isolated yields with saving in the cost of solvent and falls under the domain of green chemistry.









Similar content being viewed by others
Data availability
All data analyzed in this study are included in this article.
References
M.P. van der Helm, B. Klemm, R. Eelkema, Nat. Rev. Chem. 3, 491 (2019)
S.E. John, S. Gulati, N. Shankaraiah, Org. Chem. Front. 8, 4237 (2021)
E. Heuson, F. Dumeigni, Catal Sci Technol. 10, 7082 (2020)
S. Khaef, A. Rostami, V. Khakyzadeh, M.A. Zolfigol, A.A. Taherpour, M. Yarie, Mol. Catal. 484, 110772 (2020)
A. Khazaei, A.R. Moosavi-Zare, F. Gholami, V. Khakyzadeh, Appl. Organometal. Chem. 30, 691 (2016)
V. Khakyzadeh, H. Rezaei-Vahidian, S. Sediqi, S.B. Azimi, R. Karimi-Nami, Chem. Methodol. 5, 324 (2021)
B. List, Chem. Rev. 107, 5413 (2007)
B. List, Angew. Chem. Int. Ed. 49, 1730 (2010)
P.I. Dalko, L. Moisan, Angew. Chem. Int. Ed. 43, 5138 (2004)
D. Elhamifar, S. Kazempoor, B. Karimi, Catal. Sci. Technol. 6, 4318 (2016)
L. Zhu, N. Lei, Z. Miao, C. Sheng, C. Zhuang, J. Yao, W. Zhang, Chin. J. Chem. 30, 139 (2012)
Y. Wang, Z. Shang, T. Wu, J. Fan, X. Chen, J. Mol. Catal. A. Chem. 253, 212 (2006)
A. Rahmati, K. Vakili, Amino Acids 39, 911 (2010)
J. Han, Y. Xu, Y. Su, X. She, X. Pan, Catal. Commun. 9, 2077 (2008)
P.S. Rao, R.V. Venkataratnam, Tetrahedron Lett. 32, 5821 (1991)
B. Green, R.I. Crane, I.S. Khaidem, R.S. Leighton, S.S. Newazand, T.E. Smyser, J. Org. Chem. 50, 640 (1985)
R.M. Kumbhare, M. Sridhar, Catal. Commun. 9, 403 (2008)
D.S. Bose, A.V. Narsaiah, J. Chem. Res. 1, 36 (2001)
M. Trilla, R. Pleixats, M.W.C. Man, C. Bied, Green Chem. 11, 1815 (2009)
K. Motokura, M. Tada, Y. Iwasawa, Chem. Asian J. 3, 1230 (2008)
R. Vaid, M. Gupta, Montash chem.146, 645 (2015)
M. Kolahdoozan, R.J. Kalbasi, Z.S. Shahzeidi, F. Zamani, J. Chem. 496837 (2013)
S. Kantevari, R. Bantu, L. Nagarapu, J. Mol. Catal. A: Chem 269, 53 (2007)
V.S.R.R. Pullabhotla, A. Rahman, S.B. Jonnalagadda, Catal. Commun. 10, 365 (2009)
P.M. Price, J. H. Clark, D. J. Macquarrie, J. Chem. Soc., Dalton Trans. 101 (2000)
J. Yu, L. Zhang, B. Cheng, Y. Su, J. Phys. Chem. 111, 10582 (2007)
A. Corma, S. Iborra, I. Rodriguez, F. Sanchez, J. Catal. 211, 208 (2002)
M.B. Gawande, R.V. Jayaram, Catal. Commun. 7, 931 (2006)
S. Saravanamurugan, M. Palanichamy, M. Hartmann, V. Murugesan, Appl. Catal. A. 298, 8 (2006)
A. Corma, R.M. Martin-Aranda, Appl. Catal. A 105, 271 (1993)
T.I. Reddy, R.S. Verma, Tetrahedron Lett. 38, 1721 (1997)
U.D. Joshi, P.N. Joshi, S.S. Tamhankar, V.V. Joshi, C.V. Rode, V.P. Shiralkar, Appl. Catal. A. 239, 209 (2003)
A. Corma, V. Fornes, R.M. Martin-Aranda, H. Garcia, J. Primo, Appl. Catal. 59, 237 (1990)
J. N. Appaturi, R. Ratti, B. L. Phoon, S. M. Batagarawa, I. Ud Din, M. Selvaraj, R. J. Ramalingam, Dalton Trans. 50, 4445 (2021)
K. Komura, Y. Mishima, M. Koketsu, Appl. Catal. A: General 445, 128 (2012)
E. Rodrigo, B.G. Alcubilla, R. Sainz, J.L.G. Fierro, R. Ferritto, M.B. Cid, Chem. Commun. 50, 6270 (2014)
H. Ishitani, K. Kanai, Y. Saito, T. Tsubogo, S. Kobayashi, Eur. J. Org. Chem. 6491 (2017)
R. Jenkins, R.L. Snyder, Introduction to X-ray Powder Diffractometry (John Wiley & Sons, NY, 1996)
E. Shah, H.P. Soni, RSC Adv. 3, 17453 (2013)
E. Shah E, P. Upadhyay, M. Singh, M. S. Mansuri, R. Begum, N. Sheth, H. P. Soni, New J. Chem. 40, 9507 (2016)
M. Kakihana, T. Nagumo, O. Makoto, H. Kakihana, J. Phys. Chem. 91, 6128 (1987)
P.S. Subramanian, P.C. Dave, V.P. Boricha, D. Srinivas, Polyhedron 17, 443 (1998)
G.B. Deacon, R.J. Philips, Coord. Chem. Rev. 33, 227 (1980)
T.F. Pauwels, W. Lippens, P.W. Smet, G.G. Herman, A.M. Goeminne, Polyhedron 18, 1029 (1999)
A. Barth, Prog. Biophys. Mol. Biol. 74, 141 (2000)
K.S.W. Sing, D.H. Everett, R.A.W. Haul, L. Moscou, R.A. Pierotti, J. Rouquerol, T. Siemieniewska, Pure Appl. Chem. 57, 603 (1985)
J. Chomoucka, J. Drbohlavova, D. Huska, V. Adam, R. Kizek, J. Hubalek, Pharmacol. Res. 62, 144 (2010)
L. Li, Y. Yang, J. Ding, J.M. Xue, Chem. Mater. 22, 3183 (2010)
H.L. Swanson, C. Guo, M. Cao, Phys. Chem. Chem. Phys. 22, 20349 (1998)
R.A. Sheldon, Green Chem. 19, 18 (2017)
D. Meng Y. Qiao, X. Wang, W. Wen, S. Zhao, RSC Adv. 8, 30180 (2018)
D. Patel, R. Vithalani, C.K. Modi, New J. Chem. 44, 2868 (2020)
G. Wang, Z. Ding, L. Meng, G. Yan, Z. Chen, J. Hu, Appl. Organomet. Chem. 34, e5907 (2020)
I. delHeirro, Y. Pérez, M. Fazardo, Mol. Catal. 450, 112 (2018)
Acknowledgements
The authors thank DST-FIST for providing NMR and BET surface area measurement facilities in the Department of Chemistry, Faculty of Science, The M. S. University of Baroda.
Funding
This work was financially supported by Science and Engineering Research Board (SERB), New Delhi under a sponsored scheme (No. EMR/2016/007638).
Author information
Authors and Affiliations
Contributions
BS contributed to methodology, validation, data curation, and formal analysis. HS contributed to writing—the original draft, conceptualization, validation, investigation, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Supplementary information
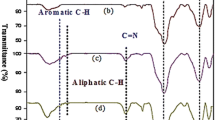
The data that supports the findings of this study are available in the supplementary material of this article. HRSEM images, FTIR, UV–Vis and fluorescence, 1H NMR, 13C NMR spectra, and HRMS data of all the products are provided in Supplementary Materials.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sanghavi, B., Soni, H.P. Fe3O4@L-arginine and Fe3O4@L-histidine nanoparticles for one-pot solvent-free sequential Knoevenagel–Michael addition reactions. Res Chem Intermed 49, 3963–3985 (2023). https://doi.org/10.1007/s11164-023-05017-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05017-3