Abstract
This study reported the fabrication of TiO2-decorated Fe2O3QDs/g-C3N4 ternary Z-scheme photocatalyst via low-temperature calcination followed by a nonaqueous route with tunable particle size and strong interfacial contact. The subsequent Fe2O3QDs/g-C3N4 and TiO2/Fe2O3QDs/g-C3N4 were investigated in terms of structure, morphology, optical properties, and surface chemical composition analysis via transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), energy dispersive x-ray spectroscopy (EDX), UV–visible spectroscopy, ESR and photoluminescence spectroscopy (PL). The crystalline parameters of the samples were investigated by X-ray diffraction (XRD) The Williamson-Hall method and geometrical phase analysis of HRTEM micrographs were employed to investigate lattice defects. Under visible light, the photocatalytic capabilities of as-fabricated TiO2/Fe2O3QDs/g-C3N4 were examined by degrading Rhodamine B (RhB), and an enhancement in photocatalytic efficacy was found. TiO2 works as a primary photosensitizer, providing extra photoinduced electrons influenced by oxygen vacancies, while Fe2O3 acts as a "bridge" for electron transport from the TiO2 moiety to the g-C3N4 thereby establishing an indirect charge transport pathway based on the Z-scheme. Radical scavenging tests were conducted to further explore the cause of increased activity and degradation mechanisms. Designing materials with oxygen vacancies and optimized structures can lead to improved solar energy conversion capabilities, particularly regarding contaminant removal. The proposed technique might be a viable option for the removal of rhodamine b compounds and for remedying freshwater reservoirs.
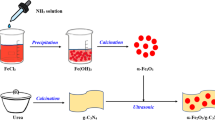
Graphical Abstract
TiO2 /Fe2O3QDs/g-C3N4Ternary Z-Scheme Photocatalysts! A facile strategy is exploited to modulate the crystallinity and surface properties of TiO2. TiO2 is incorporated into Fe2O3QDs/g-C3N4 through solvothermal treatment, which results in the formation of surface flaws and lattice-oxygen activated regions as well as the transformation of the Fe2O3QDs/g-C3N4 heterojunction into an indirect z-scheme system by rational modulation of the band alignment of two systems. The electron–hole pair is efficiently separated, conserving the electron reduction capability and the electron oxidation capability of the hole. The adapted structure and mesocrystalline nature resulted in a 7.3-fold improvement in the photocatalytic performance of TFCN over pure g-C3N4.





















Similar content being viewed by others
Availability of data and materials
The present research results have not been published before. Data and Materials are all in the main text, figures, and tables.
References
C. Bingkun, P. Narayan, Z. Haizheng, J. Phys. Chem. Lett. 9, 435 (2018)
A. Umar, M.S. Akhtar, M.S. Al-Assiri, A.E. Al Salami, S.H. Kim, Ceram Int. 44, 5017 (2018)
L. Huang, H. Xu, Y. Li, H. Li, X. Cheng, J. Xia, Y. Xu, Dalton Trans. 42, 8606 (2013)
S. Kamal, S. Balu, S. Palanisamy, K. Uma, V. Velusamy, Results Phys. 12, 1238 (2019)
W. Guo, K. Fan, J. Zhang, C. Xu, Appl. Surf. Sci. 447, 125 (2018)
S.G. Kumar, L.G. Devi, J. Phys. Chem. 115, 13211 (2011)
J. Ke, M. Adnan Younis, Y. Kong, H. Zhou, J. Liu, L. Lei, A Nano Micro Lett. 10, 6 (2018)
S. Nayak, K. Parida, Chem Asian J. 16(16), 2211 (2021)
S. Nayak, G. Swain, K. Parida, ACS Appl. Mater. Interf. 11(23), 20923 (2019)
S. Nayak, K.M. Parida, ACS Omega 3(7), 7324 (2018)
S. Nayak, K. Parida, Catalysts 11(9), 1072 (2021)
S. Nayak, K. Parida, Inorgan. Chem. Front. 7(20), 3805 (2020)
V. Rives, M.A. Ulibarri, Coord. Chem. Rev. 181(1), 61 (1999)
S. Nayak, K.M. Parida, Sci. Rep. 9(1), 2458 (2019)
S. Nayak, K. Parida, Sci. Rep. 12(1), 9264 (2022)
S. Nayak, L. Mohapatra, K. Parida, J. Mater. Chem. A 3(36), 18622 (2015)
K. Maeda, X. Wang, Y. Nishihara, D. Lu, M. Antonietti, K. Domen, J. Phys. Chem. C 113(12), 4940 (2009)
A.B. Jorge, D.J. Martin, M.T. Dhanoa, A.S. Rahman, N. Makwana, J. Tang, P.F. McMillan, J. Phys. Chem. C 117(14), 7178 (2013)
J.L. Gunjakar, I.Y. Kim, J.M. Lee, N.-S. Lee, S.-J. Hwang, Energy Environ. Sci. 6, 1008 (2013)
Y. Jiang, Y. Song, Y. Li, W. Tian, Z. Pan, P. Yang, Y. Li, Q. Gu, L. Hu, ACS Appl. Mater. Interf. 9, 37645 (2017)
S. Nayak, K.M. Parida, Int. J. Hydrog. Energy 41(46), 21166 (2016)
Y.S. Xu, W.D. Zhang, Appl. Catal. B 140, 306 (2013)
C. Zhao, L. Liu, G. Rao, H. Zhao, L. Wang, J. Xu et al., Catal. Sci. Technol. 5, 3288e95 (2015)
C. Chen, P. Gunawan, R. Xu, J. Mater. Chem. 21, 121825 (2011)
S. Nayak, A.C. Pradhan, K.M. Parida, Inorg. Chem. 57(14), 8646 (2018)
Y. Lei, C.S. Chen, Y.J. Tu, Y.H. Huang, H. Zhang, Environ. Sci. Technol. 49, 6838 (2015). https://doi.org/10.1021/acs.est.5b00623
J. Wang, C. Li, J. Cong, Z. Liu, H. Zhang, M. Liang, J. Gao, S. Wang, J. Yao, J. Solid State Chem. 238, 246 (2016). https://doi.org/10.1016/j.jssc.2016.03.042
R. Li, Y. Jia, N. Bu, J. Wu, Q. Zhen, J. Alloys. Compd. 643, 88 (2015). https://doi.org/10.1016/j.jallcom.2015.03.266
X. Chao, H. An, G. Yang, Catal Today 317, 99 (2018). https://doi.org/10.1016/j.cattod.2018.01.023
H. Gao, Z. Yang, W. Xu, S. Zhang, J. Li, Small 14, 1801353 (2018)
C.X. Guo, H.B. Yang, Z.M. Sheng, Z.S. Lu, Q.L. Song, C.M. Li, Angew. Chem. Int. Ed 49, 3014 (2010)
I. Nikitskiy, S. Goossens, D. Kufer, T. Lasanta, G. Navickaite, F.H.L. Koppens, G. Konstantatos, Nat. Commun. 7, 11954 (2016)
L. Fu, X. Xiao, A. Wang, J. Phys. Chem. Solids 122, 104 (2018)
C. Xue, X. Yan, H. An, H. Li, J. Wei, G. Yang, Appl. Catal. B 222, 157 (2018). https://doi.org/10.1016/j.apcatb.2017.10.008
X. Liu, A. Jin, Y. Jia, J. Jiang, N. Hu, X. Chen, RSC Adv. 5, 92033 (2015). https://doi.org/10.1039/c5ra18466e
S. Pany, K.M. Parida, Phys. Chem. Chem. Phys. 17(12), 8070 (2015)
J. Xu, G. Wang, J. Fan, B. Liu, S. Cao, J. Yu, J. Power Sources 15(274), 77 (2015)
L. Zhou, L. Wang, Y. Liu, J. Lei, J. Zhang, Phys. Chem. Chem. Phys. 17, 17406 (2015)
C. Wang, Z.X. Deng, G. Zhang, S. Fan, Y. Li, Powder Technol. 125, 39 (2002)
M. Niederberger, M.H. Bartl, J.D. Stucky, J. Am. Chem. Soc. 124, 13642 (2002)
M. Niederberger, G. Garnweitne, Chem. Eur. J. 12, 7282 (2006)
G.V. Jensen, M. Bremholm, N. Lock, G.R. Deen, Chem. Mater. 22, 6044 (2010)
M. Niederberger, G. Garnweitner, F. Krumeich, R. Nesper, H. Cölfen, M. Antonietti, Chem. Mater. 16, 1202 (2004)
J. Zhu, J. Yang, Z.F. Bian, J. Ren, Y.M. Liu, Y. Cao, H.X. Li, H.Y. He, K.N. Fan, Appl. Catal. B: Environ. 76, 82 (2007)
X.J. She, J.J. Wu, H. Xu, J. Zhong, Y. Wang, Y.H. Song, K.Q. Nie, Y. Liu, Y.C. Yang, M.T.F. Rodrigues, R. Vajtai, J. Lou, D.L. Du, H.M. Li, P.M. Ajayan, Adv. Energy matrix. 7, 1700025 (2017)
C. Xue, H. An, X. Yan, J. Li, B. Yang, J. Wei, G. Yang, Nano Energy 39, 513 (2017). https://doi.org/10.1016/j.nanoen.2017.07.030
A. Ganguly, S. Sharma, P. Papakonstantinou, J. Hamilton, J. Phys. Chem. C 115, 17009 (2011)
A. Mishra, A. Mehta, S. Kainth, S. Basu, J. Alloy. Comp. 764, 406 (2018)
D. Sha, J. Wang, N. Ye, Y. Dai, J. Ren, M. Chen, X. Yan, Mater. Technol. 32(7), 451 (2017)
S. Martha, A. Nashim, K.M. Parida, J. Mater. Chem. A 1, 7816 (2013)
W. Ming, Y. Jun-Min, Z. Xue- Wei, Z. Ming, Appl. Surf. Sci. 354, 196 (2015)
J. Hou, S. Jiao, H. Zhu, R.V. Kumar, CrystEngComm 13, 4735 (2011). https://doi.org/10.1039/c1ce05409k
L. Bo, J. Li, B. Xu, X. Yan, B. Yang, J. Wei, G. Yang, Appl. Catal. B 243, 94 (2019). https://doi.org/10.1016/j.apcatb.2018.10.029
I. Nakamura, N. Negishi, S. Kutsuna, T. Ihara, S. Sugihara, K. Takeuchi, J. Mol. Catal. A Chem. 161, 205 (2000)
Y. Yao, C. Xu, S. Yu, D. Zhang, S. Wang, Ind. Eng. Chem. Res. 52, 130226103026006 (2013). https://doi.org/10.1021/ie303220x
T. Ihara, M. Miyoshi, Y. Iriyama, O. Matsumoto, S. Sugihara, Appl. Catal. B Environ. 42, 403 (2003)
B. Zhang, Q. Wang, I. Zhuang, S. Guan, B. Li, J. Photochem. Photobiol. A 362, 1 (2018)
I. Ibrahim, G.V. Belessiotis, M. Antoniadou, A. Kaltzoglou, E. Sakellis, F. Katsaros, P. Falaras, Results Eng. 14, 100470 (2022)
C. Zhou, N.F. Ye, X.H. Yan, J.J. Wang, J.M. Pan, D.F. Wang, X.N. Cheng, J. Materiomics 4(3), 238 (2018)
X. Bi, S. Yu, E. Liu, L. Liu, K. Zhang, J. Zang, Y. Zhao, Colloids Surf. A 603, 125193 (2020)
W. Liao, M. Murugananthan, Y. Zhang, Phys. Chem. Chem. Phys. 17, 8877 (2015)
Funding
This work was financially supported by the Natural Science Foundation of China funds (N.S.F.C.) under Grants No. 10972025, 21876008.
Author information
Authors and Affiliations
Contributions
Shahid Iqbal wrote the main manuscript text and prepared figures under the supervision of prof. Jianjun Liu. While all the authors reviewed and formally analysed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
This declaration is “not applicable”.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iqbal, S., Liu, J., Ma, H. et al. Development of TiO2 decorated Fe2O3QDs/g-C3N4 Ternary Z-scheme photocatalyst involving the investigation of phase analysis via strain mapping and its photocatalytic performance under visible light illumination. Res Chem Intermed 49, 3327–3362 (2023). https://doi.org/10.1007/s11164-023-04987-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-04987-8