Abstract
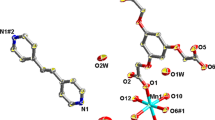
The synthesized condition by means of controlling pH values of the reaction mixtures has been explored. Three Mn2+, Cd2+ ion coordination polymers with the fully deprotonated cis, cis, cis-1,2,3,4-cyclopentanetetracarboxylic acid (H4L) ligand were synthesized and characterized, sometimes incorporating different auxiliary ligand: {[Mn(H2O)6][Mn3(L)2(H2O)2]}·3H2O (1), [Cd2L(H2O)4] (2) and [Cd2Lphen)2(H2O)2]·3H2O (3) (phen = 1,10-phenanthroline). With an initial of pH values of 6.0, 4.0 and 9.0 of the reaction mixtures, we get the compounds 1, 2 and 3, respectively. In crystal of 1, two crystallographic different Mn2+ ions (Mn1 and Mn2) form a negatively charged coordination polymeric chain, which contains a centrosymmetrically linear trinuclear Mn2+ cluster (Mn3L2) subunit; another crystallographically independent Mn2+ ions (Mn3) coordinated by six water molecules act as counter ions to link the neighboring coordination polymeric chains via intermolecular H-bond interactions. The Mn2+ ions in 1 were completely replaced by Cd2+ ions to give 2 and 3, respectively. Complex 2 shows 3D distributions of Cd2+ ions while Complex 3 displays 2D networks contributed by intermolecular H-bond interactions. Three complexes exhibit distinct thermal decomposition mechanisms, and the deprotonated cis, cis, cis-1,2,3,4-cyclopentan-etetracarboxylic acid ligands decompose in 420–700 °C to give the residue MnO + C in 1 and CdO in 2 and in 3. Complex 1 shows a complicated magnetic behavior of coexistence of antiferromagnetic exchange interactions between neighboring Mn2+ ions.
Graphical abstract













Similar content being viewed by others
References
G.R.A. Wyllie, W.R. Scheidt, Chem. Rev. 102, 1067 (2002)
F.R. Keene, Coord. Chem. Rev. 166, 121 (1997)
N.J. Castellanos, H. Martinez, F. Martinez, K. Leus, P.V. Voort, Res. Chem. Intermed. 47, 4227 (2021)
L. Perez-Garcia, D.B. Amabilino, Chem. Soc. Rev. 36, 941 (2007)
S.S. Rad, H. Saeidiroshan, B.M. Eshkevari, M.A. Ghasemzadeh, Res. Chem. Intermed. 47, 2143 (2021)
S. Nagahama, T. Tanaka, A. Matsumoto, Angew. Chem. 116, 3899 (2004)
S. Noro, S. Kitagawa, M. Kondo, K. Seki, Angew. Chem. Int. Ed. 39, 2081 (2000)
M.J. Zaworotko, Angew. Chem. Int. Ed. 39, 2113 (2000)
B. Li, R.J. Wei, J. Tao, R.B. Huang, L.S. Zheng, Z.P. Zheng, J. Am. Chem. Soc. 132, 1558 (2010)
H.C. Zhou, S. Kitagawa, Chem. Soc. Rev. 43, 5415 (2014)
S.T. Zheng, G.Y. Yang, Dalton Trans. 39, 700 (2010)
A.C. Kathalikkattil, Y. Gu, J.F. Kurisingal, H. Lee, H. Kim, Y. Choe, D.W. Park, Res. Chem. Intermed. 47, 3979 (2021)
D.K. Wang, Z.H. Li, Res. Chem. Intermed. 43, 5169 (2017)
M. Ding, R.W. Flaig, H.L. Jiang, O.M. Yaghi, Chem. Soc. Rev. 48, 2783 (2019)
R.Q. Zou, H. Sakurai, S. Han, R.Q. Zhong, Q. Xu, J. Am. Chem. Soc. 129, 8402 (2007)
R. Kitaura, S. Kitagawa, Y. Kubota, T.C. Kobayashi, K. Kindo, Y. Mita, A. Matsuo, M. Kobayashi, H.C. Chang, T.C. Ozawa, M. Suzuki, M. Sakata, M. Takata, Science 298, 2358 (2002)
H. Furukawa, K.E. Cordova, M. O’Keefe, O.M. Yaghi, Science 341, 1230444 (2013)
X.L. Li, K. Chen, Y. Liu, Z.X. Wang, T.W. Wang, J.L. Zuo, Y.Z. Li, Y. Wang, J.S. Zhu, J.M. Liu, Y. Song, X.Z. You, Angew. Chem. Int. Ed. 46, 6820 (2007)
J.Y. Hu, J.A. Zhao, Q.Q. Guo, H.W. Hou, Y.T. Fan, Inorg. Chem. 49, 3679 (2010)
H. Hou, Y. Wei, Y. Song, L. Mi, M. Tang, L. Li, Y. Fan, Angew. Chem. Int. Ed. 44, 6067 (2005)
W. Chen, H.M. Yuan, J.Y. Wang, Z.Y. Liu, J.J. Xu, M. Yang, J.S. Chen, J. Am. Chem. Soc. 125, 9266 (2003)
J. Yan, J. Gao, D.L. Long, H.N. Miras, L. Cronin, J. Am. Chem. Soc. 132, 11410 (2010)
A.K. Srivastava, G. Panda, Chem. Eur. J. 14, 4675 (2008)
A. Mlodzianowska, L. Latos-Grazynski, L. Szterenberg, Inorg. Chem. 47, 6364 (2008)
S. Ma, D. Sun, J.M. Simmons, C.D. Collier, D. Yuan, H.C. Zhou, J. Am. Chem. Soc. 130, 1012 (2008)
B.H. Ye, M.L. Tong, X.M. Chen, Coord. Chem. Rev. 249, 545 (2005)
K.P. Rao, A. Thirumurugan, C.N.R. Rao, Chem. Eur. J. 13, 3193 (2007)
X.Y. Wang, S.C. Sevov, Chem. Mater. 19, 3763 (2007)
L.F. Ma, L.Y. Wang, F. Zhong, Synth. React. Inorg. Metal-Org. Chem. 38, 455 (2008)
L. Zhang, J. Zhang, Z.J. Li, Y.Y. Qin, Q.P. Lin, Y.G. Yao, Chem. Eur. J. 15, 989 (2009)
J. Luo, F. Jiang, R. Wang, M. Hong, Inorg. Chem. Commun. 7, 638 (2004)
B.S. Randhawa, M. Kaur, J. Therm. Anal. Calorim. 89, 251 (2007)
J. Wang, L.L. Zheng, C.J. Li, Y.Z. Zheng, M.L. Tong, Cryst. Growth Des. 6, 357 (2006)
J. Wang, S. Hu, M.L. Tong, Eur. J. Inorg. Chem. 10, 2069 (2006)
J. Wang, Z.J. Lin, Y.C. Ou, Y. Shen, R. Herchel, M.L. Tong, Chem. Eur. J. 14, 7218 (2008)
L.F. Huang, C.C. Ji, Z.Z. Lu, X.Q. Yao, J.S. Hu, H.G. Zheng, Dalton Trans. 40, 3183 (2011)
C.S. Liu, E.C. Sañudo, M. Hu, L.M. Zhou, L.Q. Guo, S.T. Ma, L.J. Gao, S.M. Fang, CrystEngComm 12, 853 (2010)
SAINT, Version 6.02a; Bruker AXS Inc.: Madison, W1, 2002.
G. M. Sheldrick, SADABS, Program for Bruker Area Detector Absorption Correction, University of Göttingen, Göttingen, Germany, (1997)
G. M. Sheldrick, SHELXS-97, Program for Crystal Structure Solution, University of Göttingen, Göttingen, Germany, (1997)
G. M. Sheldrick, SHELXL-97, Program for Crystal Structure Refinement, University of Göttingen, Göttingen, Germany, (1997)
K.L. Zhang, J.B. Yuan, L.J. Yuan, J.T. Sun, Chinese. J. Inorg. Chem. 15, 549 (1999)
X.M. Ren, T. Akutagawa, S. Noro, S. Nishihara, T. Nakamura, Y. Yoshida, K. Inoue, J. Phys. Chem. B 109, 7671 (2006)
O. Kahn, Molecular Magnetism (Wiley-VCH, New York, 1993)
T.C. Higgs, K. Spartalian, C.J.O. Connor, B.F. Matzanke, C.J. Carrano, Inorg. Chem. 37, 2263 (1998)
Acknowledgements
This work supported by Grants from the Natural Science Research Project of Anhui Provincial Department of Education (KJ2012B178, KJ2012ZD11, KJ2021A145), Science Research Project of Huangshan University (2012xkjq010) and the National Undergraduate Training Program for Innovation and Entrepreneurship (202010375004, 202010375015).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, LF., Qi, ZP. & Li, CJ. Three coordination polymers of manganese(II), cadmium(II) with a flexible ligand of cis, cis, cis-1,2,3,4-cyclopentanetetracarboxylic acid: crystal structures, thermal decomposition mechanisms and magnetic properties. Res Chem Intermed 48, 3867–3882 (2022). https://doi.org/10.1007/s11164-022-04784-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04784-9