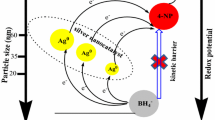
Abstract
Today, plant extracts based on synthetic procedures have drawn consideration over conventional methods like physical and chemical procedures to synthesize nanomaterials. Green synthesis of nanomaterials has become an area of interest because of numerous advantages such as non-hazardous, economical, and feasible methods with a variety of applications in biomedicine, nanotechnology and nano-optoelectronics and as catalysts for various organic transformations. In this research, silver nanoparticles were deposited on the surface of nano-silica spheres by an in-situ reduction of Ag+ ions using an aqueous extract of Thymus kotschyanus aerial parts as a natural reducing and a capping agent. The result recorded from ultraviolet–visible (UV–Vis) spectrometer, Fourier transform infrared spectroscopy (FT-IR), thermal gravimetric analysis, scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM–EDS) and X-ray powder diffraction supports the biosynthesis and characterization of Ag/SiO2 nanoparticles. The results indicated that the average size of Ag/SiO2 nanoparticles is 25–60 nm. The Ag/SiO2 nanoparticles act as an environmentally friendly heterogeneous catalyst in the synthesis of spirooxindoles via the three-component condensation reaction of isatins, activated methylene reagents, and 1,3-dicarbonyl compounds in aqueous media, and the desired products were obtained with yields ranging from 90 to 98%. The catalyst can be recovered easily and used repetitively without significant loss of catalytic activity.
Graphical abstract









Similar content being viewed by others
References
J.S. Wilkes, Green Chem. 4, 73 (2002)
P. Anastas, N. Eghbali, Chem. Soc. Rev. 39, 301 (2010)
X.Y. Li, Y. Liu, X.L. Chen, X.Y. Lu, X.X. Liang, S.S. Zhu, C.W. Wei, L.B. Qu, B. Yu, Green Chem. 22, 4445 (2020)
Z. Sadri, F.K. Behbahani, COS 17, 324 (2020)
K.D. Dwivedi, B. Borah, L.R. Chowhan, Front. Chem. 7, 944 (2020)
K. Verma, Y.K. Tailor, S. Khandelwal, M. Agarwal, E. Rushell, Y. Kumari, K. Awasthi, M. Kumar, RSC Adv. 8, 30430 (2018)
L. Dai, K. Mao, Z. Pan, L. Rong, Res. Chem. Intermed. 45, 769 (2019)
V.M. Muzalevskiy, K.V. Belyaeva, B.A. Trofimov, V.G. Nenajdenko, Green Chem. 21, 6353 (2019)
J. Feng, S. Wang, J. Feng, Q. Li, J. Yue, G. Yue, P. Zou, G. Wang, New J. Chem. 44, 11937 (2020)
Y.C. Wang, J.L. Wang, K.S. Burgess, J.W. Zhang, Q.M. Zheng, Y.D. Pu, L.J. Yan, X.B. Chen, RSC Adv. 8, 5702 (2018)
C.J. Li, T.H. Chan, Organic reactions in aqueous media (Wiley, New York, 1997)
H.C. Hailes, Org. Process. Res. Dev. 11, 114 (2007)
K. Zhang, J.M. Suh, J.W. Choi, H.W. Jang, M. Shokouhimehr, R.S. Varma, ACS Omega 4, 483 (2019)
D. Raja, A. Philips, P. Palani, W.Y. Lin, S. Devikala, G.C. Senadi, J. Org. Chem. 8, 11531 (2020)
T.N. Ansari, J.B. Jasinski, D.K. Leahy, S. Handa, J. Am. Chem. Soc. 1, 308 (2021)
O. Gherasim, R.A. Puiu, A.C. Bîrcă, A.C. Burdușel, A.M. Grumezescu, Nanomaterials 10, 2318 (2020)
K.V. Alex, P.T. Pavai, R. Rugmini, M.S. Prasad, K. Kamakshi, K.C. Sekhar, ACS Omega 5, 13123 (2020)
P. Mathur, S. Jha, S. Ramteke, N.K. Jain, Artif. Cells. Nanomed Biotechnol. 46, 115 (2018)
M. Bhagat, R. Anand, R. Datt, V. Gupta, S. Arya, J. Inorg. Organomet. Polym. Mater. 29, 1039 (2019)
L. Huang, Y. Sun, S. Mahmud, H. Liu, J. Inorg. Organomet. Polym. Mater. 30, 1653 (2020)
W. Ali, V. Shabani, M. Linke, S. Sayin, B. Gebert, S. Altinpinar, M. Hildebrandt, J.S. Gutmann, T. Mayer-Gall, RSC Adv. 9, 4553 (2019)
L. Zhu, H. Dai, S. Zhang, D. Hu, Q. Zhou, M. Zou, J. Adkins, J. Zheng, Anal. Lett. 52, 2868 (2019)
X.Y. Dong, Z.W. Gao, K.F. Yang, W.Q. Zhang, L.W. Xu, Catal. Sci. Technol. 5, 2554 (2015)
R.B. Nasir Baig, R.S. Varma, ACS Sustain. Chem. Eng. 1, 805 (2013)
R. Baharfar, R. Azimi, Synth. Commun. 44, 89 (2014)
M. Cobos, I. De-La-Pinta, G. Quindós, M.J. Fernández, M.D. Fernández, Nanomaterials 10, 376 (2020)
R. Poreddy, E.J. García-Suárez, A. Riisager, S. Kegnæs, Dalton. Trans. 43, 4255 (2014)
J.X. Zhao, R.N. Shi, Y.H. Quan, J.J. Liu, J. Wang, Y.L. Pei, X.H. Wang, Z. Li, J. Ren, Chem. Eng. Sci. 207, 1060 (2019)
X. Pei, Y. Li, Y. Deng, L. Lu, W. Li, R. Shi, A. Lei, L. Zhang, Carbohydr. Polym. 251, 117020 (2021)
Z. Zhang, Y. Jiang, M. Chi, Z. Yang, C. Wang, X. Lu, RSC Adv. 5, 94456 (2015)
V.G. Pol, D.N. Srivastava, O. Palchik, V. Palchik, M.A. Slifkin, A.M. Weiss, A. Gedanken, Langmuir 18, 3352 (2002)
Z. Jiang, C. Liu, J. Phys. Chem. B 107, 12411 (2003)
Y. Kobayashi, V. Salgueiriño-Maceira, L.M. Lizmarzán, Chem. Mater. 13, 1630 (2001)
W. Chen, J. Zhang, Scr. Mater. 49, 321 (2003)
H. Ramadoss, D. Saravanan, S.P. Sudhan, S.S. Mansoor, Der. Pharm. Lett. 8, 25 (2016)
K. Ding, Y. Lu, Z. Nikolovska-Coleska, S. Qiu, Y. Ding, W. Gao, J. Stuckey, K. Krajewski, P.P. Roller, Y. Tomita, D.A. Parrish, J.R. Deschamps, S. Wang, J. Am. Chem. Soc. 127, 10130 (2005)
S. Mathusalini, T. Arasakumar, K. Lakshmi, C.H. Lin, P.S. Mohan, M.G. Ramnath, R. Thirugnanasampandan, New J. Chem. 40, 5164 (2016)
A.H. Abdel-Rahman, E.M. Keshk, M.A. Hanna, S.M. El-Bady, Bioorg. Med. Chem. 12, 2483 (2004)
G. Kumari, M. Modi, S.K. Gupta, R.K. Singh, Eur. J. Med. Chem. 46, 1181 (2011)
V.V. Vintonyak, K. Warburg, H. Kruse, S. Grimme, K. Hübel, D. Rauh, H. Waldmann, Angew. Chem. Int. Ed. Engl. 49, 5902 (2010)
S.T. Hilton, T.C.T. Ho, G. Pljevaljcic, K. Jones, Org. Lett. 2, 2639 (2000)
S.P. Baran, R.M. Richter, J. Am. Chem. Soc. 127, 15394 (2005)
M.Y. Chang, C.L. Pai, Y.H. Kung, Tetrahedron Lett. 46, 8463 (2005)
J. Ma, S.M. Hecht, Chem. Commun. 1190 (2004)
S. Edmondson, S.J. Danishefsky, L. Sepplorenzinol, N. Rosen, J. Am. Chem. Soc. 121, 2147 (1999)
Y. Wang, A.A. Cobo, A.K. Franz, Org. Chem. Front. 8, 4315 (2021)
G.J. Mei, F. Shi, Chem. Commun. 54, 6607 (2018)
L.J. Yan, Y.C. Wang, Chem. Select. 1, 6948 (2016)
D. Cheng, Y. Ishihara, B. Tan, C.F. Barbas, ACS Catal. 4, 743 (2014)
L. Hong, R. Wang, Adv. Synth. Catal. 355, 1023 (2013)
X.N. Zhang, Y.X. Li, Z.H. Zhang, Tetrahedron 67, 7426 (2011)
A. Kamal, K.S. Babu, M.V.P.S.V. Vardhan, S.M.A. Hussaini, R. Mahesh, S.P. Shaik, A. Alarifi, Bioorg. Med. Chem. Lett. 25, 2199 (2015)
L. Moradi, Z. Ataei, Green. Chem. Lett. Rev. 10, 380 (2017)
B. Karmakar, A. Nayak, J. Banerji, Tetrahedron Lett. 53, 5004 (2012)
M. Kidwai, A. Jahan, N.K. Mishra, Appl. Catal. A: General. 425–426, 35 (2012)
G.D. Wang, X.N. Zhang, Z.H. Zhang, J. Heterocycl. Chem. 50, 61 (2013)
S. Pradhan, B.G. Mishra, Mol. Catal. 446, 58 (2018)
G.M. Ziarani, A. Badiei, S. Mousavi, N. Lashgari, A. Shahbazi, Chin. J. Catal. 33, 1832 (2012)
Y. Li, H. Chen, C. Shi, D. Shi, S. Ji, J. Comb. Chem. 12, 231 (2010)
F.A. Tameh, J.S. Ghomi, M.M. Hashemi, H.S. Alavi, RSC. Adv. 6, 74802 (2016)
F. Kamali, F. Shirini, J. Mol. Struct. 1227, 129654 (2021)
M.N. Chen, J.Q. Di, J.M. Li, L.P. Mo, Z.H. Zhang, Tetrahedron 76, 131059 (2020)
B. Sadeghi, M. Ghasem-Pirbaluti, P. Farokhi Nezhad, R. Abbasi Nezhad, Res. Chem. Intermed. 41, 4047 (2015)
K. Niknam, P. Abolpour, Monatsh. Chem. 146, 683 (2015)
B. Sadeghi, Z. Lasemi, R. Azimi, Orient. J. Chem. 31, 1175 (2015)
M. Zhang, Q.Y. Fu, G. Gao, H.Y. He, Y. Zhang, Y.S. Wu, Z.H. Zhang, A.C.S. Sustain, Chem. Eng. 5, 6175 (2017)
Z. Lasemi, M. Tajbakhsh, H. Alinezhad, F. Mehrparvar, Res. Chem. Intermed. 46, 3667 (2020)
R. Azimi, Z. Lasemi, A. Bagheri Hashkavayi, Lett. Org. Chem. 17, 268 (2020)
R. Azimi, R. Baharfar, Can. J. Chem. 92, 1163 (2014)
R. Baharfar, R. Azimi, M. Mohseni, J. Food. Sci. Technol. 52, 6777 (2015)
E.C. Njagi, H. Huang, L. Stafford, H. Genuino, H.M. Galindo, J.B. Collins, G.E. Hoag, S.L. Suib, Langmuir 27, 264 (2011)
M. Dabiri, M. Bahramnejad, M. Baghbanzadeh, Tetrahedron 65, 9443 (2009)
S. Zhu, S.I. Ji, Y. Zhang, Tetrahedron 63, 9365 (2007)
K. Rad-Moghadam, L. Youseftabar-Miri, Tetrahedron 67, 5693 (2011)
R.G. Chaudhary, J.A. Tanna, N.V. Gandhare, A.R. Rai, H.D. Juneja, Adv. Mater. Lett. 6, 990 (2015)
G.S. Hari, Y.R. Lee, Synthesis 2010, 453 (2010)
G. Shanthi, G. Subbulakshmi, P.T. Perumal, Tetrahedron 63, 2057 (2007)
Acknowledgements
We are grateful to the Research Institute of Forests and Rangelands, Agricultural Research of Iran for partial support of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Azimi, R., Lasemi, Z. Environmentally friendly synthesis of Ag/SiO2 nanoparticles using Thymus kotschyanus extract and its application as a green catalyst for synthesis of spirooxindoles. Res Chem Intermed 48, 1615–1630 (2022). https://doi.org/10.1007/s11164-022-04667-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04667-z