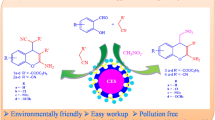
Abstract
A sustainable, green and efficient process for the synthesis of 2-nitro alcohol derivatives from different substituted aromatic aldehydes with nitroalkane by stirring at ambient temperature with high product yield is reported. Adoption of very mild reaction conditions, use of Calcined Eggshell (CES) as natural catalyst and simple workup are expected to contribute to the development of environmentally benign synthetic method for Henry (nitroaldol) reaction. CES is ecologically safe, inexpensive, and attractive heterogeneous base catalyst obtained from renewable resources, thus opening a new perspective for this process.
Graphical abstract









Similar content being viewed by others
References
T. Suami, K.I. Tadono, A. Suga, Y. Ueno, J. Carbohydr. Chem. 3, 429 (1984)
E.J. Corey, F.Y. Zhang, Angew. Chem. Int. Ed. 38, 1931 (1999)
Y. Sohtome, N. Takemura, T. Iguchi, Y. Hashimoto, K. Nagasawa, Synlett. 2006(1), 144 (2006)
R.E. Looper, R.M. Williams, Angew. Chem. Int. Ed. 43, 2930 (2004)
I. Kudyba, J. Raczko, J. Jurczak, J. Org. Chem. 69, 2844 (2004)
I. Kudyba, J. Raczko, J. Jurczak, Tet. Lett. 44, 8685 (2003)
B.M. Trost, V.S.C. Yeh, H. Ito, N. Bremeyer, Org. Lett. 4, 2621 (2002)
H. Sasai, N. Itoh, T. Suzuki, M. Shibasaki, Tet. Lett. 34, 855 (1993)
T.M. Williams, H.S. Mosher, Tet. Lett. 26, 6269 (1985)
P.I. Dalko, L. Moisan, Angew. Chem. Int. Ed. 40, 3726 (2001)
L. Henry, Bull. Soc. Chim. Fr. 13, 999 (1895)
J. Hubner, J. Liebscher, M. Patzel, Tetrahedron 58, 10485 (2002)
B.M. Vanderbilt, H.B. Hass, Ind. Eng. Chem. 32, 34 (1940)
H. B. Hass, B. M. Vanderbilt, U. S. Patent, 2139120 (1938)
F.J. Villani, F.F. Nord, J. Am. Chem. Soc. 69, 2608 (1947)
C.B. Gairaud, G.R. Lappin, J. Org. Chem. 18, 1 (1953)
H.B. Hass, A.G. Susie, R.L. Heider, J. Org. Chem. 15, 8 (1950)
M.J. Astle, F.P. Abbott, J. Org. Chem. 21, 1228 (1956)
F.G. Cirujano, R. Luque, A. Dhakshinamoorthy, Molecules 26, 1445 (2021)
S. Niju, K.M.M.S. Begum, N. Anantharaman, Environ. Prog. Sustain Energy 34, 248 (2015)
S. Niju, K.M. Meera, S. Begum, N. Anantharaman, J Saudi Chem Soc 18, 702 (2014)
G. Joshi, D.S. Rawat, B.Y. Lamba, K.K. Bisht, P. Kumar, N. Kumar, S. Kumar, Energy Convers. Manag. 96, 258 (2015)
M.B. Deshmukh, S.S. Patil, S.D. Jadhav, P.B. Pawar, Syn. Comm. 42, 1177 (2012)
S.S. Patil, S.D. Jadhav, M.B. Deshmukh, J. Chem. Sci. 125, 851 (2013)
S.S. Patil, S.D. Jadhav, U.P. Patil, Arch. Appl. Sci. Res. 4(2), 1074 (2012)
S.S. Patil, S.D. Jadhav, M.B. Deshmukh, Ind J. Chem. Sec. B 52B, 1172 (2013)
S.T. Morbale, S.D. Jadhav, M.B. Deshmukh, S.S. Patil, RSC Adv. 5, 84610 (2015)
S.T. Morbale, S.K. Shinde, S.D. Jadhav, M.B. Deshmukh, S.S. Patil, Der Pharm. Lett. 7(12), 169 (2015)
R.C. Patil, U.P. Patil, A.A. Jagdale, S.K. Shinde, S.S. Patil, Res. Chem. Intermed. 46, 3527 (2020)
R.C. Patil, S.K. Shinde, U.P. Patil, A.T. Birajdar, S.S. Patil, Res. Chem. Intermed. 47, 1675 (2021)
R. C. Patil, A. A. Jagdale, U. P. Patil, J. S. Ghodake, S. S. Mali, C. K. Hong, S. S. Patil Catal Lett (2021)
R.C. Patil, S.A. Damate, D.N. Zambare, S.S. Patil, New J. Chem. 45, 9152 (2021)
W.J. Stadelman, Encyclopedia of Food Science and Technology, 2nd edn. (Wiley, New York, 2000)
K. Rajkumari, D. Das, G. Pathak, L. Rokhum, New J. Chem. 43, 2134 (2019)
R.C. Tang, Z. Guan, Y.H. He, W. Zhu, J. Mol. Catal. B Enzym. 63, 62 (2010)
N. Surneni, N.C. Barua, B. Saikia, Tetrahedron Lett. 57, 2814 (2016)
M. Jagdale, A. Naikwade, R.S. Salunkhe, M. Rajmane, G.S. Rashinkar, New J. Chem. 42, 10993 (2018)
A. Karmakar, S. Hazra, M.F.C.G. Silva, A.J.L. Pombeiro, New J. Chem. 38, 4837 (2014)
X. Yu, B. Perez, Z. Zhang, R. Gao, Z. Guo, Green Chem. 18, 2753 (2016)
Y. Xi, R.J. Davis, J. Mol. Catal. A Chem. 341, 22 (2011)
Acknowledgements
Authors Rupesh C. Patil and Ashutosh A. Jagdale are grateful to SARTHI, Pune for the award of CMSRF-2019 and CMNRF-2019 fellowship, respectively. [CIN-U74999PN2018NPL177394, dated 11th Sept. 2019]. Authors are also thankful to Indian Institute of Chemical Technology (IICT), Hyderabad for NMR analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jadhav, S.D., Patil, R.C., Jagdale, A.A. et al. Revisit to Henry reaction by non conventional heterogeneous and efficient catalyst for nitroalcohol synthesis. Res Chem Intermed 48, 593–606 (2022). https://doi.org/10.1007/s11164-021-04608-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04608-2